Abstract
Background or Purpose
The prognosis of m ixed cardiomyopathy (CMP) in patients with implanted cardioverter-defibrillators (ICDs) has not been investigated. We aim to study the demographic, clinical, device therapies and survival characteristics of mixed CMP in a cohort of patients implanted with a defibrillator.
Methods
The term mixed CMP was used to categorise patients with impaired left ventricular ejection fraction attributed to documented non-ischemic triggers with concomitant moderate coronary artery disease. This is a single center observational cohort of 526 patients with a mean follow-up of 8.7 ± 3.5 years.
Results
There were 42.5% patients with ischemic cardiomyopathy (ICM), 26.9% with non-ischemic cardiomyopathy (NICM) and 30.6% with mixed CMP. Mixed CMP, compared to NICM, was associated with higher mean age (69.1 ± 9.6 years), atrial fibrillation (55.3%) and greater incidence of comorbidities. The proportion of patients with mixed CMP receiving device shocks was 23.6%, compared to 18.4% in NICM and 27% in ICM. The VT cycle length recorded in mixed CMP (281.6 ± 43.1 ms) was comparable with ICM (282.5 ± 44 ms; p = 0.9) and lesser than NICM (297.7 ± 48.7 ms; p = 0.1). All-cause mortality in mixed CMP (21.1%) was similar to ICM (20.1%; p = 0.8) and higher than NICM (15.6%; p = 0.2). The Kaplan–Meier curves revealed hazards of 1.57 (95% CI: 0.91, 2.68) for mixed CMP compared to NICM.
Conclusion
In a cohort of patients with ICD, the group with mixed CMP represents a phenotype predominantly comprised of the elderly with a higher incidence of comorbidities. Mixed CMP resembles ICM in terms of number of device shocks and VT cycle length. Trends of long-term prognosis of patients with mixed CMP are worse than NICM and similar to ICM.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Significant progress has been made with the tools in diagnosis and management of heart failure. One of these advances is the prevention of sudden cardiac death (SCD) with implantable cardioverter-defibrillators (ICD) [1]. Regardless, the long-term mortality rates in heart failure patients, even with ICD, continue to remain as high as 50% at 10 years [2, 3]. These trends are worse in ischemic (ICM) than in nonischemic (NICM) forms of cardiomyopathy (CMP) [4]. It may not be right to simplify the burden of coronary artery disease (CAD) in patients with cardiomyopathies as a binary component of epicardial stenosis of more than or less than 75%, and thus attribute the heart failure to ischemic or nonischemic aetiologies [5]. There is ongoing research on ways to detect ischemia in cardiomyopathies [6]. The studies on the prognosis of concomitant CAD in dilated cardiomyopathies (DCM) are few, and these studies have reported the prognosis of the association of CAD in only idiopathic DCM [7-9]. Thus, the effect of moderate CAD coexisting with DCM with definite non-ischemic triggers is largely unexplored. The resultant phenotype of ‘mixed cardiomyopathy’ might identify clinical and outcome characteristics that are distinct from ICM or NICM and may impact on clinical management. This phenotype is gaining attention of late and the prognosis in terms of increased ventricular arrhythmia burden seems to parallel ICM [10, 11]. We aim to study the demographic, clinical, device therapies and survival characteristics of mixed CMP in a cohort of patients implanted with a defibrillator.
2 Methods
The Canberra Hospital (TCH) device registry is a prospectively maintained database of implanted cardiac devices. The demographic and clinical data is being recorded at scheduled clinic visits and the device data is being interrogated through scheduled or unscheduled clinic visits and remote monitoring of the devices. In this study, consecutive patients receiving an ICD between January 2005 and June 2019 who had regular interrogation (clinical or remote transmission) of the implanted ICD in the follow-up and with an invasive coronary angiogram to rule out coronary artery disease were included. The identity of these patients was linked with the National Death Index (NDI) obtained from the Australian Institute of Health and Welfare (AIHW) to confirm the survival status and cause of death. The following patients were excluded from the study: incomplete clinical or device data; no survival data; in-hospital or immediate post-procedure (< 30 days) deaths; channelopathies.
The study complies with the Declaration of Helsinki and was approved by the Human Research Ethics Committee (2019/LRE/0127) and the AIHW Ethics Committee (EO2020/1/1102). The primary objective of the study was to analyse the characteristics of the demographic variables, clinical variables, device therapies and survival data of patients receiving an ICD in patients with mixed CMP in comparison with ICM and NICM. The secondary objectives were to analyse the characteristics of clinical, device therapies and mortality in non-survivors in the total cohort and to identify the significant predictors of mortality in the total cohort.
2.1 Data collection
Demographic and clinical variables including history of diabetes mellitus, hypertension, chronic kidney disease (CKD), lung disease, malignancy, alcohol/ drug abuse, renal functions and echocardiographic findings including type and severity of valve pathologies were recorded. The left ventricular ejection fraction (LVEF) at implant and at the last follow-up was recorded. History of CAD, myocardial infarction (MI), percutaneous coronary intervention (PCI) history of bypass surgery, valve replacement; documented atrial and ventricular arrhythmias; list of anti-arrhythmic and heart failure medications; symptoms of syncope or sudden cardiac arrest (SCA); history of radiofrequency ablation (RFA) for VT in relation to the time of the ICD implant was collected. The following device characteristics were collected: information on clinical interrogation during a scheduled clinic visit or remote transmission, type of ICD, the programming zones of the ICD, date of first and second therapy from the device, verification of the type of tachyarrhythmia and the type of therapies delivered verified with the stored intracardiac electrograms (EGMs), change in the programming parameters, ventricular tachyarrhythmia (VT) storms, minimum cycle length of the recorded VT (1st and 2nd episode was taken into account), date and number of generator changes, therapies after generator change. The survival characteristics were collected from the NDI.
2.2 Study definitions
-
1.
Coronary artery disease (CAD) was defined by the presence of stenosis \(\ge\) 50% in atleast one of three major epicardial vessels or \(\ge\) 30% in the left main vessel. Lesions on coronary angiography (CAG) were graded visually by two cardiologists on the following ordinal scale: 0 to < 50%, \(\ge\) 50 to < 75, \(\ge\) 75% and 100%. The interobserver agreement for both grading of stenosis and location of CAD was calculated. The final consensus was reached upon by mutual agreement.
-
2.
ICM was defined as those patients with impaired LVEF in whom there was a history of MI, evidence of prior MI in form of q-waves in ECG or regional wall motion abnormalities in echocardiogram or \(\ge\) 75% coronary artery stenosis in one of the major epicardial vessels or \(\ge\) 50% coronary artery stenosis in the left main coronary artery as evidenced in a diagnostic coronary angiogram (CAG) [12].
-
3.
NICM was defined as those patients with depressed LV systolic function (< 50%) in whom moderate to severe CAD (\(\ge\) 50% stenosis in one of the epicardial coronary vessels) was ruled out by a CAG and with no history suggestive of MI. After corroborative evidence from electrocardiography, echocardiography, cardiac MRI, PET scan and genomic assessment, the aetiopathogenesis of NICM was assigned and included the following; post-myocarditis sequelae, arrhythmogenic right ventricular cardiomyopathy, sarcoidosis, hypertrophic cardiomyopathy, infiltrative cardiomyopathy (amyloidosis, hemochromatosis), non-compaction (dilated and low LVEF associated with features of non-compaction documented by echocardiogram or cardiac MRI), valvular heart disease (severe valvular stenosis/ regurgitation leading to dilatation of heart and low LVEF), alcohol-related (documented alcohol abuse or dependence leading to deterioration in LVEF), congenital heart disease (including post-operative patients with persisting heart defects or new onset valvular diseases), tachy-cardiomyopathy, and chemotherapy-related cardiomyopathy. Patients with no known aetiology other than those stated above, but with LVEF ≤ 35% were classified as idiopathic dilated cardiomyopathy (DCM).
-
4.
The term mixed CMP was used in this study to categorise patients with depressed LV systolic function (< 50%), a documented non-ischemic aetiology and with moderate CAD (\(\ge\) 50% and < 75% stenosis) in one or more of left anterior descending artery (LAD), left circumflex artery (LCX), right coronary artery (RCA), or 30–50% stenosis involving the left main coronary artery.
-
5.
Minimum cycle length of VT was calculated based on the least measured near-field EGM intervals in the available intracardiac traces. The average of first 10 intervals was considered in case of unstable intervals. Additional study definitions are incorporated in the supplement.
2.3 Statistical analysis
Categorical variables are summarised as percentages. Normally distributed continuous data is expressed as mean ± SD, and non-normally distributed data is expressed as median with interquartile range of 25th and 70th percentiles. For comparing variables, we used a χ2-test (categorical variables), a t-test (normally distributed continuous variables) and a Mann–Whitney U test (non-normal continuous variables). The kappa statistics were used to calculate the inter-observer variability in the extent and location of CAD detected in the coronary angiograms. Cumulative hazard and the survival curves following ICD intervention were analysed with the Kaplan–Meier survival analysis method and the statistical comparison using the log-rank test. The Cox proportional hazards regression models were used to determine the predictors of survival. The coefficients were expressed as hazard ratios with 95% confidence intervals. p value < 0.05 was considered statistically significant.
3 Results
In this study, 526 patients were followed up for a mean period of 8.7 ± 3.5 years. The total cohort comprised of 224 patients of ICM (42.5%), 141 patients of NICM (26.9%) and 161 patients of mixed CMP (30.6%) (Fig. 1).
3.1 Demographic and clinical characteristics (Table 1)
The mean age of patients with mixed CMP (69.1 ± 9.6 years) was higher compared to both ICM (66.3 ± 10.9 years; p = 0.008) and NICM (54.4 ± 14.5 years; p < 0.001). The mean LVEF in patients with mixed CMP (32.9 ± 8.6%) was comparable to patients with ICM (32.7 ± 8.3%; p = 0.8) and lower compared to patients with NICM (40.9 ± 14.2%; p < 0.001). The proportion of male gender was 82% in mixed CMP, 92% in ICM and 66.7% in NICM. Patients with mixed CMP, in comparison with ICM, had lesser proportions of diabetes mellitus (33.5% vs 44.8%; p = 0.03), higher proportions of alcohol abuse (22.4% vs 8%; p < 0.001) and malignancy (30.4% vs 2.7%; p < 0.001), and comparable proportions of hypertension, chronic lung diseases and chronic kidney diseases (Table 1; Supplemental Table 1). Patients with mixed CMP, in comparison with NICM, had higher proportions of diabetes mellitus (33.5% vs 13.5%; p < 0.001), systemic hypertension (62.1% vs 36.2%; p < 0.001), chronic lung disease (13% vs 2.1%; p < 0.001), chronic kidney disease (22.4% vs 7.1%; p < 0.001), malignancy (30.4% vs 11.3%; p < 0.001) and comparable proportions of alcohol abuse (22.4% vs 17%; p = 0.2).
The distribution of moderate CAD in patients with mixed CMP was LM/LAD (22.4%), LCX/RCA (1.8%), double vessel disease (56.6%) and triple vessel disease (18.6%). The level of agreement was strong (kappa = 0.81 for grading of stenosis and 0.83 for location of CAD) between the two cardiologists. The coexisting nonischemic aetiologies in the patients of mixed CMP were post myocarditis sequelae (32.9%), chemotherapy-related (24.2%), tachycardiomyopathy (19.3%), alcohol-related (16.1%) and hypertrophic cardiomyopathy (7.5%). The nonischemic aetiologies in the patients of NICM were idiopathic (23%), ARVC (11%), restrictive CMP (22.7%), valvular heart diseases (12.8%), inflammatory (10.6%), chemotherapy-related (5%), tachycardiomyopathy (6.4%), alcohol-related (7.1%) and congenital heart diseases (1.4%).
The proportion of patients receiving ICD for secondary prevention in mixed CMP was 44.1% compared to 56.3% in ICM (p = 0.02) and 38.3% in NICM (p = 0.3). While history of sudden cardiac arrest was comparable amongst all the 3 groups (23.6% in mixed CMP, 20.5% in ICM and 16.3% in NICM), incidence of atrial fibrillation was higher in mixed CMP (55.3%) compared to ICM (28.6%; p < 0.001) and NICM (30.5%; p < 0.001). While usage of beta blockers was comparable amongst all the 3 groups (> 95%), amiodarone usage was highest in ICM (38%). With respect to the distribution of type of ICD implant, patients with mixed CMP had higher proportions of CRT-d (29.2%) compared to patients with ICM (18.4%; p = 0.04) and NICM (18.4%; p = 0.03).
3.2 Analysis of device therapies (Table 1; Supplemental Table 2)
The proportion of patients with mixed CMP receiving device therapies (34.2%) and device shocks (23.6%) was intermediate between ICM (device therapies 41.1%; device shocks 27.2%) and NICM (device therapies 29.1%; device shocks 18.4%). These differences were not significant between mixed CMP and the other groups. Among the patients receiving device shocks, the distribution of appropriate and inappropriate shocks was comparable between all the 3 groups. The minimum VT cycle length recorded in patients with mixed CMP (281.6 ± 43.1 ms) was comparable to that in ICM (282.5 ± 44 ms; p = 0.9) in ICM and lesser than in NICM (297.7 ± 48.7 ms; p = 0.1).
3.2.1 Survival characteristics
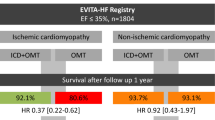
The all-cause mortality in patients in mixed CMP (21.1%) was similar to that observed in ICM (20.1%; p = 0.8) and higher than in NICM (15.6%; p = 0.2). Time-adjusted survival estimated using the Kaplan-Meir curves revealed hazards of 1.57 (95% CI: 0.91, 2.68; p = 0.1) for mixed CMP compared to NICM (Fig. 2). The mean age at death in patients with mixed CMP (79 ± 8 years) was significantly higher than in ICM (73 ± 12 years; p = 0.01) and NICM (66 ± 14 years; p < 0.001). Analysis of the cause of death revealed higher proportion of non-cardiac deaths in patients with mixed CMP (52.9%), compared to ICM (26.7%; p = 0.04) and NICM (18.2%; p = 0.02). The distribution of heart failure related deaths and sudden cardiac deaths was similar between all the 3 groups (Table 2).
The Cox regression analysis (Table 3) revealed the following significant predictors of mortality age (HR: 1.04; 95% C.I: 1.02–1.06), LVEF (HR: 0.96; 95% C.I: 0.93–0.99), CKD (HR: 2.9; 95% C.I: 1.9–4.5), NYHA class (HR: 1.7; 95% C.I:1.1–2.4) and CAD (HR: 1.9; 95% C.I: 1.1–3.2). This model accounted for various confounding variables including age, gender, clinical variables, presence or absence of moderate–severe CAD and documented nonischemic triggers. Compared to the survivors in the whole cohort, the non-survivors had significantly (p < 0.05) higher mean age (69.1 ± 11.8y vs 62.7 ± 13y), lower LVEF (29.7 ± 6.6% vs 36.2 ± 11.3%), higher NYHA class III (51.5% vs 19.8%), lower GFR (65.8 ± 27.8 vs 85.5 ± 25.2), and significantly higher incidences of comorbidities—hypertension (64.4% vs 52.9%), chronic kidney disease (46.5% vs 10%), malignancy (23.8% vs 11%). The distribution of ICM, NICM and mixed CMP was similar (Supplemental Table 3). The proportion of patients receiving therapies was significantly higher in the non-survivors compared to the survivors (50% vs 32.3%, p = 0.001). Among the patients receiving device therapies, significantly higher proportion of patients received shocks in non-survivors compared to survivors (79.6% vs 63.7%, p = 0.04).
4 Discussion
The salient features of our study are as follows: (1) the phenotype of mixed CMP, when compared to NICM, is associated with higher mean age and higher incidence of comorbidities; (2) ventricular arrhythmias in mixed CMP resembles ICM in terms of number of device shocks and VT cycle length; and (3) trends of long-term prognosis of patients with mixed CMP is worse than NICM and similar to ICM.
4.1 Extent of CAD in dilated cardiomyopathy
When accounted for moderate CAD, our study reveals that at least 53% of the NICM cohort, with known nonischemic triggers, would be reclassified as mixed CMP. This cohort accounts to 30.6% of the total cohort of cardiomyopathies in our study. Cardiomyopathies with overlapping ischemic and nonischemic aetiologies are not uncommon in clinical practice [11]. In a histopathological study on hearts excised at transplantation in patients diagnosed with idiopathic DCM, coronary atherosclerosis was diagnosed in 65.5% of the hearts with 43.6% showing moderate to severe lesions [13].
In our study, nearly 77% of the mixed CMP patients had moderate CAD in more than one epicardial vessel and the majority had double vessel involvement. Concomitant CAD in DCM has been studied previously; however, they have been largely on idiopathic DCM. In addition, the results of prognosis reported in these studies are contradictory. In a study on idiopathic DCM patients, CAD burden had significant correlation with major adverse cardiovascular events [8]. Yet another large-scale study in over 12,000 heart failure patients had also shown that the prognosis in nonobstructive CAD (< 70% stenosis) is worse than in heart failure with no CAD [9]. However, a few other studies did not show differences in survival between idiopathic DCM with moderate CAD and no CAD [4, 7]. Our study is different from the above studies in that it reveals poor prognosis in patients with implanted defibrillators and CMP secondary to definite nonischemic triggers and with concomitant CAD (\(\ge\) 50% to < 75% stenosis). This subset has been largely excluded from the previous studies of DCM with coexisting CAD.
4.2 The phenotype of mixed CMP
We found mixed CMP more common in the elderly and male patients when compared to both ICM and NICM. Also, the clinical phenotype in mixed CMP seems to represent a subset of patients with higher incidences of comorbidities, especially hypertension, chronic kidney diseases, atrial fibrillation and malignancies, when compared to NICM. It is perceivable that these risk factors would also explain a relatively higher burden of CAD found in the group with mixed CMP compared to NICM [14, 15]. This finding is also consistent with the studies on idiopathic DCM with coexisting CAD [7-9]. While the proportion of device therapies and device shocks in mixed CMP falls in an intermediate category between ICM and NICM, the recorded minimum VT cycle length is comparable to patients with ICM. In a very recent study, albeit in a small cohort of 24 patients with mixed CMP undergoing catheter ablation for ventricular arrhythmias, it was shown that this subset had a higher incidence of ventricular arrhythmias and all-cause mortality than both ICM and NICM [10]. Our study reveals all-cause mortality rates of nearly 20% in both the ICM and mixed CMP cohorts. As the mean age and incidences of coexisting illnesses especially chronic kidney diseases and malignancies are higher in the cohort of mixed CMP, it is not surprising that most of the deaths in this cohort are non-cardiac, unlike the predominantly cardiac deaths in ICM and NICM. The mixed CMP group revealed higher hazards of all-cause mortality when compared to NICM (HR: 1.57; 95% CI: 0.91–2.68; p = 0.1). In a larger study of 2254 heart failure patients with nonobstructive CAD, when compared to 2656 heart failure patients with no CAD, there was an increased hazard of cardiovascular death (HR: 1.82; 95% CI: 1.27–2.62; p < 0.001) and all-cause mortality (HR: 1.18; 95% CI: 1.05–1.33; p < 0.005) [9].
4.3 Possible pathogenesis in mixed CMP (Central Illustration- Fig. 3)
While epicardial CAD is only one determinant of myocardial ischemia, there are multiple contributing factors: (1) supply–demand mismatch due to the low coronary perfusion pressures in the setting of severe myocardial dysfunction, (2) coronary microvascular dysfunction secondary to atherosclerosis, (3) impaired myocardial metabolic control due to the underlying CMP [16]. Coronary perfusion indices like flow reserves and microvascular resistance have been shown to be associated with poor prognosis in heart failure independent of ischemic or nonischemic classification [6, 17, 18]. Electro anatomical mapping studies have highlighted the mixed pathophysiological substrate in this subset of mixed CMP [10, 19, 20]. Such mixed pathological substrates have also been documented in small-scale studies with LGE-CMRi as well as with perfusion-CMRi [8, 21, 22]. While these can be plausible explanations for the bad prognosis in mixed CMP, there could be several other contributing factors as well like age and coexisting illnesses.
4.4 Limitations
This is a retrospective study focussing on characterising the phenotype of mixed CMP, and hence the causal relationship between moderate CAD and depressed systolic function could not be sought. Whether or not myocardial revascularisation would benefit these patients in the presence of a demonstrable myocardial ischemia, has to be explored prospectively. Also, scoring of the extent of CAD and its burden with indices or variables like focal or diffuse involvement and location is likely to throw more light into the incremental effect of each variable on the perfusion abnormality [4, 8]. A larger sample size could have established statistical significance to the observed higher trends of mortality in mixed CMP compared to NICM. Finally, though this is the first study to address the phenotype of mixed CMP in patients implanted with defibrillators, and hence arbitrary definitions were employed for the categorisation of mixed CMP.
5 Conclusion
Our study characterises the mixed phenotype of dilated cardiomyopathies who have established nonischemic triggers and concomitant moderate CAD, in a cohort who had received an ICD. The prognosis in patients with mixed CMP, with regards to device therapies and all-cause mortality, resembles ICM. The prognosis in patients with mixed CMP is poorer than NICM in terms of significantly higher burden of comorbidities, poorer LV functions and trend towards higher proportions of device shocks and higher mortality. The higher mortality seems to be driven by higher incidences of non-cardiac deaths, thus representing a sicker subset than NICM. Large-scale studies focusing on this phenotype need to assess the mediators of poorer prognosis due to the underlying pathophysiological substrate and the associated coexisting illnesses.
Data availability
The data that support the findings of the study are available on request from the corresponding author.
Abbreviations
- CAD:
-
Coronary artery disease
- CAG:
-
Coronary angiogram
- CKD:
-
Chronic kidney disease
- CMP:
-
Cardiomyopathy
- CMRi:
-
Cardiac magnetic resonance imaging
- DCM:
-
Dilated cardiomyopathy
- EGM:
-
Electrogram
- HR:
-
Hazard ratio
- ICD:
-
Implantable cardioverter-defibrillator
- ICM:
-
Ischemic cardiomyopathy
- LAD:
-
Left anterior descending artery
- LCX:
-
Left circumflex artery
- LGE:
-
Late gadolinium enhacement
- LV:
-
Left ventricle
- LVEF:
-
Left ventricular ejection fraction
- MI:
-
Myocardial infarction
- MRI:
-
Magnetic resonance imaging
- NICM:
-
Nonischemic cardiomyopathy
- NYHA:
-
New York heart association
- NDI:
-
National death index
- PCI:
-
Percutaneous coronary intervention
- PET:
-
Positron emission tomography
- RCA:
-
Right coronary artery
- SCD:
-
Sudden cardiac death
- SCA:
-
Sudden cardiac arrest
- VT:
-
Ventricular tachyarrhythmia
References
Pathak RK, Sanders P, Deo R. Primary prevention implantable cardioverter-defibrillator and opportunities for sudden cardiac death risk assessment in non-ischaemic cardiomyopathy. Eur Heart J. 2018;39:2859–66.
Poole JE, Olshansky B, Mark DB, et al. Long-term outcomes of implantable cardioverter-defibrillator therapy in the SCD-HeFT. J Am Coll Cardiol. 2020;76:405–15.
Raja DC, Samarawickrema I, Das S, et al. Long-term mortality in heart failure with mid-range ejection fraction: systematic review and meta-analysis. ESC Heart Fail. 2022;9:4088–99.
Bart BA, Shaw LK, McCants CB, et al. Clinical determinants of mortality in patients with angiographically diagnosed ischemic or nonischemic cardiomyopathy. J Am Coll Cardiol. 1997;30:1002–8.
Felker GM, Shaw LK, O’Connor CM. A standardized definition of ischemic cardiomyopathy for use in clinical research. J Am Coll Cardiol. 2002;39:210–8.
Majmudar MD, Murthy VL, Shah RV, et al. Quantification of coronary flow reserve in patients with ischaemic and non-ischaemic cardiomyopathy and its association with clinical outcomes. Eur Heart J Cardiovasc Imaging. 2015;16:900–9.
Frankenstein L, Hees H, Taeger T, et al. Clinical characteristics, morbidity, and prognostic value of concomitant coronary artery disease in idiopathic dilated cardiomyopathy. Clin Res Cardiol. 2013;102:771–80.
Canu M, Margerit L, Mekhdoul I et al. Prognosis of coronary atherosclerotic burden in non-ischemic dilated cardiomyopathies. J Clin Med 2021;10
Braga JR, Austin PC, Ross HJ, Tu JV, Lee DS. Importance of nonobstructive coronary artery disease in the prognosis of patients with heart failure. JACC Heart Fail. 2019;7:493–501.
Bennett RG, Campbell T, Kotake Y, Turnbull S, Kumar S. Clinical, electroanatomic and electrophysiologic characterization, and outcomes of catheter ablation for ventricular tachycardia in patients with a mixed cardiomyopathy. Circ Arrhythm Electrophysiol. 2022;15:e010476.
Madias JE. Ischemic, nonischemic, and probably “mixed” dilated cardiomyopathies: what’s in a definition? Int J Cardiol. 2014;175:565–6.
Bardy GH, Lee KL, Mark DB, et al. Amiodarone or an implantable cardioverter–defibrillator for congestive heart failure. N Engl J Med. 2005;352:225–37.
Repetto A, Dal Bello B, Pasotti M, et al. Coronary atherosclerosis in end-stage idiopathic dilated cardiomyopathy: an innocent bystander? Eur Heart J. 2005;26:1519–27.
Albakri A. Ischemic cardiomyopathy: a review of literature on clinical status and meta-analysis of diagnostic and clinical management. Bio Eng Med 2018;3
Das D, Asher A, Ghosh AK. Cancer and coronary artery disease: common associations, diagnosis and management challenges. Curr Treat Options Oncol. 2019;20:46.
Goodwill AG, Dick GM, Kiel AM, Tune JD. Regulation of coronary blood flow. Compr Physiol. 2017;7:321–82.
Yong AS, Ho M, Shah MG, Ng MK, Fearon WF. Coronary microcirculatory resistance is independent of epicardial stenosis. Circ Cardiovasc Interv. 2012;5(103–8):S1-2.
Paterson I, Mielniczuk LM, O’Meara E, So A, White JA. Imaging heart failure: current and future applications. Can J Cardiol. 2013;29:317–28.
Aldhoon B, Tzou WS, Riley MP, et al. Nonischemic cardiomyopathy substrate and ventricular tachycardia in the setting of coronary artery disease. Heart Rhythm. 2013;10:1622–7.
Hutchinson MD, Gerstenfeld EP, Desjardins B, et al. Endocardial unipolar voltage mapping to detect epicardial ventricular tachycardia substrate in patients with nonischemic left ventricular cardiomyopathy. Circ Arrhythm Electrophysiol. 2011;4:49–55.
Gulsin GS, Shetye A, Khoo J, et al. Does stress perfusion imaging improve the diagnostic accuracy of late gadolinium enhanced cardiac magnetic resonance for establishing the etiology of heart failure? BMC Cardiovasc Disord. 2017;17:98.
Raja DC, Samarawickrema I, Srinivasan JR et al. Correlation of myocardial strain by CMR-feature tracking with substrate abnormalities detected by electro-anatomical mapping in patients with nonischemic cardiomyopathy. J Interv Card Electrophysiol 2023
Acknowledgements
Dr Sanders is supported by a Practitioner Fellowship from the National Health and Medical Research Council of Australia and by the National Heart Foundation of Australia. We acknowledge the support extended by the cardiac physiologists at the Canberra Health Services and the Canberra Heart Rhythm for the collection of the data and maintenance of the CHR ICD registry.
Funding
Open Access funding enabled and organized by CAUL and its Member Institutions
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics approval
The study was conducted in accordance with the Declaration of Helsinki. The study was approved by the institutional Human Research Ethics Committee (2019/LRE/0127) and the AIHW Ethics Committee (EO2020/1/1102).
Conflict of interest
Dr Sanders reports having served on the advisory board of Medtronic, Abbott Medical, Boston Scientific, CathRx and PaceMate. Dr Sanders reports that the University of Adelaide has received on his behalf lecture and/or consulting fees from Medtronic, Abbott Medical and Boston Scientific. Dr Sanders reports that the University of Adelaide has received on his behalf research funding from Medtronic, Abbott Medical, Boston Scientific, BD and Microport. All other authors have no disclosures. Dr Pathak reports having served on the advisory board of Medtronic, Abbott Medical, and Boston Scientific. Dr Pathak reports that Canberra Heart Rhythm Foundation has received on his behalf lecture and/or consulting fees from Medtronic, Abbott Medical, Boston Scientific and Biotronik.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Raja, D.C., Samarawickrema, I., Menon, S.K. et al. Characteristics of the phenotype of mixed cardiomyopathy in patients with implantable cardioverter-defibrillators. J Interv Card Electrophysiol 67, 129–137 (2024). https://doi.org/10.1007/s10840-023-01577-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10840-023-01577-x