Abstract
Purpose
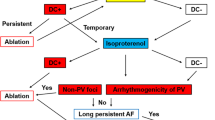
The CARTOFINDER mapping system analyzes activation patterns using unipolar potentials during atrial fibrillation (AF), where isoproterenol (ISP) is conventionally used to induce non-pulmonary vein (PV) foci and confirm PV arrhythmogenicity. In 20 patients with persistent AF who underwent ablation at our hospital, arrhythmogenic foci were evaluated using both these methods.
Methods
Before pulmonary vein isolation (PVI), PV and left atrium (LA) were analyzed during AF using CARTOFINDER, and the isolation line was determined based on the results. After PVI, ISP was loaded after return of sinus rhythm and confirmation of the presence of arrhythmogenic foci. The activation site in LA was ablated at the discretion of the surgeon.
Results
Focal activation sites detected by CARTOFINDER correlated with the arrhythmogenic foci induced by ISP in the PVs. The results also showed that a greater number of focal activation sites in the PVs correlated to an increased response to ISP administration. In one patient, it was observed that the focal activation site identified in the PV also coincided with the site of the origin of automaticity induced by ISP after PVI.
Conclusion
CARTOFINDER and ISP both reliably determined the presence of arrhythmogenic foci in PV, in patients with persistent AF. Knowledge of the nature of arrhythmogenic foci in non-PV is considered to be a topic for future studies, and further data collection is required.




Similar content being viewed by others
Data availability
Not applicable.
Code availability
Not applicable.
References
Haïssaguerre M, Jaïs P, Shah DC, et al. Spontaneous initiation of atrial fibrillation by ectopic beats originating in the pulmonary veins. N Engl J Med. 1998;339:659–66. https://doi.org/10.1056/NEJM199809033391003.
Kumagai K, Ogawa M, Noguchi H, Yasuda T, Nakashima H, Saku K. Electrophysiologic properties of pulmonary veins assessed using a multielectrode basket catheter. J Am Coll Cardiol. 2004;43:2281–9. https://doi.org/10.1016/j.jacc.2004.01.051.
Verma A, Jiang CY, Betts TR, et al. Approaches to catheter ablation for persistent atrial fibrillation. N Engl J Med. 2015;372:1812–22. https://doi.org/10.1056/NEJMoa1408288.
Bhargava M, Di Biase L, Mohanty P, et al. Impact of type of atrial fibrillation and repeat catheter ablation on long-term freedom from atrial fibrillation: results from a multicenter study. Heart Rhythm. 2009;6:1403–12. https://doi.org/10.1016/j.hrthm.2009.06.014.
Hayashi K, An Y, Nagashima M, et al. Importance of non-pulmonary vein foci in catheter ablation for paroxysmal atrial fibrillation. Heart Rhythm. 2015;12:1918–24. https://doi.org/10.1016/j.hrthm.2015.05.003.
Santangeli P, Zado ES, Hutchinson MD, et al. Prevalence and distribution of focal triggers in persistent and longstanding persistent atrial fibrillation. Heart Rhythm. 2016;13:374–82. https://doi.org/10.1016/j.hrthm.2015.10.023.
Hung Y, Lo LW, Lin YJ, et al. Characteristics and long-term catheter ablation outcome in longstanding persistent atrial fibrillation patients with non-pulmonary vein triggers. Int J Cardiol. 2017;241:205–11. https://doi.org/10.1016/j.ijcard.2017.04.050.
Chang HY, Lo LW, Lin YJ, et al. Long-term outcome of catheter ablation in patients with atrial fibrillation originating from non-pulmonary vein ectopy. J Cardiovasc Electrophysiol. 2013;24:250–8. https://doi.org/10.1111/jce.12036.
Zhao Y, Di Biase L, Trivedi C, et al. Importance of non-pulmonary vein triggers ablation to achieve long-term freedom from paroxysmal atrial fibrillation in patients with low ejection fraction. Heart Rhythm. 2016;13:141–9. https://doi.org/10.1016/j.hrthm.2015.08.029.
Verma A, Sarkozy A, Skanes A, et al. Characterization and significance of localized source identified by a novel automated algorithm during mapping of human persistent atrial fibrillation. J Cardiovasc Electrophysiol. 2018;29:1480–8. https://doi.org/10.1111/jce.13742.
Honarbakhsh S, Schilling RJ, Providencia R, et al. Automated detection of repetitive focal activations in persistent atrial fibrillation: validation of a novel detection algorithm and application through panoramic and sequential mapping. J Cardiovasc Electrophysiol. 2019;30:58–66. https://doi.org/10.1111/jce.13752.
Sakamoto Y, Osanai H, Hiramatsu S, et al. Efficacy of isoproterenol in the evaluation of dormant conduction and arrhythmogenic foci identification in atrial fibrillation ablation. BMC Cardiovasc Disord. 2020;20:397. https://doi.org/10.1186/s12872-020-01685-w.
Takahashi Y, Yamashita S, Suzuki M, Yabe K, Hirao K. Efficacy of catheter ablation of focal sources in persistent atrial fibrillation. J Cardiovasc Electrophysiol. 2018;29:559–65. https://doi.org/10.1111/jce.13415.
Calvo D, Rubin J, Pérez D, Moris C. Ablation of rotor domains effectively modulates dynamics of human: long-standing persistent atrial fibrillation. Circ Arrhythm Electrophysiol. 2017;10:e005740. https://doi.org/10.1161/CIRCEP.117.005740.
Takahashi Y, Akiyoshi K, Sekigawa M, et al. Endocardial contact mapping of the left atrial appendage in persistent atrial fibrillation. J Cardiovasc Electrophysiol. 2020;31:112–8. https://doi.org/10.1111/jce.14278.
Takahashi Y, Iwai S, Yamashita S, et al. Novel mapping technique for localization of focal and reentrant activation during atrial fibrillation. J Cardiovasc Electrophysiol. 2017;28:375–82. https://doi.org/10.1111/jce.13163.
Honarbakhsh S, Schilling RJ, Dhillon G, et al. A novel mapping system for panoramic mapping of the left atrium: application to detect and characterize localized sources maintaining atrial fibrillation. JACC Clin Electrophysiol. 2018;4:124–34. https://doi.org/10.1016/j.jacep.2017.09.177.
Daoud EG, Zeidan Z, Hummel JD, et al. Identification of repetitive activation patterns using novel computational analysis of multielectrode recordings during atrial fibrillation and flutter in humans. JACC Clin Electrophysiol. 2017;3:207–16. https://doi.org/10.1016/j.jacep.2016.08.001.
Acknowledgements
We gratefully acknowledge the contributions of the co-medical staff working on our ablation team.
Author information
Authors and Affiliations
Contributions
Yusuke Sakamoto: conception and design of study, acquisition of data, analysis and interpretation of data, and manuscript drafting
Hiroyuki Osanai: conception and design of study, technical help, writing and editing assistance
Shotaro Hiramatsu: technical help, writing and editing assistance
Shun Kondo: technical help, writing and editing assistance
Takahiro Kanbara: technical help, writing and editing assistance
Yoshihito Nakashima: technical help, writing and editing assistance
Hiroshi Asano: technical help, writing and editing assistance
Masayoshi Ajioka: technical help, writing and editing assistance
Corresponding author
Ethics declarations
Ethics approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. The study was approved by the Bioethics Committee of the Tosei General Hospital (No. 954).
Consent to participate
All patients provided written informed consent.
Consent for publication
All patients provided written informed consent.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sakamoto, Y., Osanai, H., Hiramatsu, S. et al. Evaluation of arrhythmogenic foci using a novel automated detection algorithm and isoproterenol in persistent atrial fibrillation. J Interv Card Electrophysiol 65, 63–71 (2022). https://doi.org/10.1007/s10840-022-01124-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10840-022-01124-0