Abstract
Purpose
Mitral annular flutter (MAF) is a common arrhythmia after atrial fibrillation ablation. We sought to compare the efficacy and safety of catheter ablation utilizing either a left atrial anterior wall (LAAW) line or a lateral mitral isthmus (LMI) line.
Methods
We performed a systematic review for all studies that compared LAAW versus LMI lines. Risk ratio (RR) and mean difference (MD) 95% confidence intervals were measured for dichotomous and continuous variables, respectively.
Results
Four studies with a total of 594 patients were included, one of which was a randomized control trial. In the LMI ablation group, 40% of patients required CS ablation. There were no significant differences in bidirectional block (RR 1.26; 95% CI, 0.94–1.69) or ablation time (MD −1.5; 95% CI, −6.11–3.11), but LAAW ablation was associated with longer ablation line length (MD 11.42; 95% CI, 10.69–12.14) and longer LAA activation delay (MD 67.68; 95% CI, 33.47–101.89.14) when compared to LMI. There was no significant difference in pericardial effusions (RR 0.36; 95% CI, 0.39–20.75) between groups and more patients were maintained sinus rhythm (RR 1.19; 95% CI, 1.03–1.37, p = 0.02) who underwent LAAW compared to LMI.
Conclusion
Ablation of mitral annular flutter with a LAAW line compared to a LMI line showed no difference in rates of acute bidirectional block, ablation time, or pericardial effusion. However, LAAW ablation required a longer ablation line length, resulted in greater LAA activation delayed and was associated with more sinus rhythm maintenance, with the added advantage of avoiding ablation in the CS.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
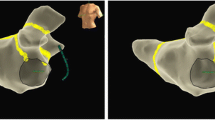
Mitral annular flutter (MAF) is the most common left atrial macro-reentrant atrial arrhythmia following catheter ablation of atrial fibrillation (AF) [1]. MAF is often resistant to both rate-controlling and antiarrhythmic drugs, thus necessitating catheter ablation for treatment [2]. The two most common approaches for ablation of peri-mitral flutter include a left atrial anterior wall (LAAW) line and a lateral mitral isthmus (LMI) line. While the LAAW line is drawn between the anterior mitral isthmus and right superior pulmonary vein (or occasionally the left superior pulmonary vein or roof line), the LMI line is drawn between the left lower pulmonary vein and LMI. Both approaches have been shown to be effective, but the LMI line often requires additional coronary sinus (CS) ablation to achieve bidirectional block [3,4,5]. The purpose of our current study was to perform a systematic review of the literature and meta-analysis to compare the efficacy and safety of both approaches.
2 Methods
We searched PubMed, clinicaltrials.gov, Medline, Google Scholar, and the Cochrane Central Register of Clinical Trials (Cochrane Library, Issue 09, 2017). This was assessed up to May 2020. No language restriction was applied. The reference list of all eligible studies was also reviewed. Search terms included (Mitral Annular Flutter OR Atrial Fibrillation) and (Mitral Isthmus Ablation or Anterior Mitral Ablation) and (Catheter Ablation).
Studies were selected by two independent reviewers. The PRISMA statement for reporting systemic reviews and meta-analyses was applied to the methods for this study [6]. The studies had to fulfill the following criteria to be considered in the analysis: (1) Studies had to have compared outcomes in patients who underwent ablation with LAAW versus LMI lesion sets; (2) Studies had to have compared and reported rates of achieving bidirectional block, ablation times, ablation line length, LAA activation delay, rates of pericardial effusions, and/or maintenance of sinus rhythm; (3) Studies must have been published in a peer-reviewed scientific journal.
We aimed to compare the efficacy and safety between LAAW and LMI lines. Two authors (F.L. and O.M.A.) independently performed literature search and extracted data from eligible studies. Outcomes were extracted from original manuscripts and supplementary data. Information was gathered using standardized protocol and reporting forms. Disagreements were resolved by consensus. Two reviewers (F.L. and O.M.A.) independently assessed the quality items and discrepancies were resolved by consensus or involvement of a third reviewer (J.C.H), if necessary.
Two authors (F.L. and O.M.A.) independently assessed the risk of bias of the included trials using standard criteria defined in the Cochrane Handbook for Systematic Reviews of Interventions. Discrepancies were resolved by discussion or adjudication by a third author (J.C.H.).
Data was summarized across treatment arms using the Mantel-Haenszel risk ratio (RR), inverse variance mean difference (MD). The Mantel-Haenszel methods are the fixed-effect methods used when event rates are low or study size is small, as the estimates of the standard errors of the effect estimates that are used in the inverse variance methods may be poor. Heterogeneity of effects was evaluated using the Higgins I-squared (I2) statistic. Random effects models for analyses were used with high heterogeneity (defined as I2 > 25%); otherwise, fixed effects models of DerSimonian and Laird were used. Funnel plot analysis was used to address publication bias. The statistical analysis was performed by the Review Manager (RevMan) Version 5.3. Copenhagen: The Nordic Cochrane Centre, The Cochrane Collaboration, 2014. Descriptive statistics are presented as means and standard deviations (SD) for continuous variables or number of cases (n) and percentages (%) for dichotomous and categorical variables.
3 Results
3.1 Study selection
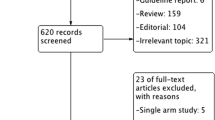
The initial search resulted in 1384 abstracts, of which 725 were duplications and 627 were excluded based on titles and abstracts (Fig. 1). We included four studies in our final analysis, including one prospective randomized control trial [7], two prospective nonrandomized studies [8, 9], and one retrospective study [10].
3.2 Study characteristics
Baseline demographics of patients included in the four studies are summarized in Table 1. We included a total of 594 patients. Patients were predominately male and many had failed anti-arrhythmic medications. Overall, 318 patients (54%) underwent LAAW ablation and 276 patients (46%) had LMI ablation. Study characteristics are shown in Table 2. A mitral isthmus line was drawn for documented MAF in 25% of patients in the study by Heumer et al. and 93% of patients in the study by Maheshwari et al. [8, 10]. The remainder of patients had a mitral isthmus line drawn empirically [7, 9]. In all studies, patients underwent pulmonary vein isolation (PVI), cavo-tricuspid isthmus ablation, and left atrial (LA) roof line ablation, following varied protocols. LAAW ablation was performed by connecting the right superior pulmonary vein (PV) to the mitral annulus in two studies [7, 10], left superior PV to the anterior mitral annulus in one study [8], and LA roof line to the mitral annulus in one study [9]. LMI was performed by ablating from the left inferior PV to the posterolateral mitral annulus. Two studies reported use of a contact force catheter [9, 10] and steerable sheaths [8, 10], whereas the other two studies did not report whether or not these items were used.
3.3 Quality assessment
The risk of bias is summarized in Figs. 2 and 3. Three studies were considered to be at “high risk” for selection bias due to non-randomization [8,9,10]. All studies were considered to be at “unclear risk” for performance bias as blinding methodology was not reported. One study was considered to have “unclear risk” for attrition bias given the data on attrition after randomization of cohorts is unavailable [7].
3.4 Study endpoints
Study endpoints between the LAAW and LMI groups are summarized in Figs. 4 and 5. There were no significant differences in achievement of bidirectional block (RR 1.26; 95% CI, 0.94–1.69, p = 0.11) between LAAW (79%) and LMI (62%) groups. Of the 170 patients who achieved bidirectional block with a LMI line, 107 required additional ablation in the coronary sinus. One study proceeded with the alternative ablation strategy when the initial attempt failed to achieve conduction block (i.e., 5 patients who failed to achieve conduction block in the LAAW group underwent LMI ablation of which 4 were successful, and of 7 patients in the LMI group who failed to achieve conduction block, 3 underwent LAAW ablation but none were successful) [10]. Although there was no difference in ablation time in LAAW and LMI groups (20.6 ± 7.7 versus 22.1 ± 11.4 min) (MD −1.5; 95% CI, −6.11–3.11, p = 0.52), LAAW ablation was associated with longer ablation line length (37.7 ± 3.9 versus 26.3 ± 3.9 mm) (MD 11.42; 95% CI, 10.69–12.14, p < 0.01). LAAW ablation was associated with significant LAA activation delay (156.4 ± 35.9 versus 88.7 ± 31.0 ms) (MD 67.68; 95% CI, 33.47–101.89, p < 0.01), a trend toward fewer pericardial effusions (0.9 versus 3.6%) (RR 0.36; 95% CI, 0.12–1.12, p = 0.08), and a higher proportion of patients maintained in sinus rhythm at follow-up (66.6 versus 56.3%) (RR 1.19; 95% CI, 1.03–1.37, p = 0.02).
4 Discussion
This is the first systematic review and meta-analysis of studies comparing procedural and peri-procedural outcomes between LAAW and LMI ablation lines for treatment of MAF. The results of this meta-analysis show that there are no significant differences in rates of bidirectional block, ablation time, and risk of pericardial effusion between ablation approaches. However, LAAW ablation necessitates a longer ablation line length and results in delayed LAA activation, while improving maintenance of sinus rhythm during follow-up and foregoing the need for ablation of the CS.
While there are two approaches to mitral isthmus line ablations, the LMI line was first described and has been the conventional approach [4]. However, ablation using this line often necessitates ablation within the CS, which may result in complications such as coronary spasm or occlusion, perforation of the CS, and pericardial tamponade [3, 11,12,13]. Furthermore, achieving conduction block with an LMI line may be challenging given the increased myocardial thickness, convective cooling from the CS and left circumflex artery, and epicardial connections [14, 15]. A superolateral mitral isthmus line, where the left-sided pulmonary veins are connected with the mitral annulus along the posterior base of the left atrial appendage, may be a potential alternative. This method targets the mitral isthmus at its thinnest portion where bridging of an endocardial linear lesion by muscular sleeves encircling the CS is unlikely. While a high acute success rate of bidirectional block using endocardial ablation only has been demonstrated (98.2 versus 87.7% with the traditional LMI line, p = 0.06), it may be associated with a higher incidence of pericardial tamponade (5.2 versus 0% with the traditional LMI line, p = 0.24) [16].
The LAAW line was described subsequently in an attempt to overcome these limitations. Conduction block may be easier to achieve in the low-voltage area of the LAAW; the rigid structure of the posterior wall of the aorta facilitates good contact pressure; ablation across the septo-atrial bundle reduces left atrial critical mass and may block multi-loop reentry around the mitral valve annulus. In addition, complex fractionated atrial electrograms are usually localized to the antero-septum or base of the left atrial appendage and may be disrupted by a LAAW ablation line [17,18,19], Furthermore, although the LAAW line is longer, it encounters less endocardial obstacles such as diverticula and pouches, which means that it usually requires a lower amount of radiofrequency energy to create a transmural lesion [20, 21]. However, LAAW line ablation has its own limitations. LAAW ablation results in significant conduction delay to the left atrial appendage and may result in intra-atrial or atrio-ventricular dyssynchrony [8, 9]. While cardiac MRI follow-up of 29 patients did not show significant hemodynamic derangements, there is still lack of data on the possible consequences on left atrial appendage flow velocity [9]. Inadvertent disconnection of the left atrial appendage may result from disruption of Bachmann’s bundle and has led to a concern about exposing the patient to a higher risk of thrombus formation and systemic embolization (e.g., stroke) [7, 22, 23], but evidence thus far has been conflicting [7, 24]. Furthermore, failure to achieve durable conduction block, which occurs in a significant percentage of patients [3, 9, 25,26,27,28], can result in areas of slow conduction along the line that can become substrate for macroreentry [29, 30]. Difficulties in attaining complete conduction block at the mitral isthmus have been attributed to a lack of contact pressure and catheter stability, myocardial thickness, and possible epicardial sleeves at the CS [26, 28, 31]. There is also the potential to injure the sinus nodal artery that runs along the left atrial roof which can result in sinus node dysfunction [32].
While this study did not show any difference in the safety of LAAW and LMI lines immediately post-procedure, long-term follow-up is lacking. As previously mentioned, the stroke risk associated with conduction delay from a LAAW ablation is not known. Matsuo et al. looked at 50 patients over an average follow-up of 19 ± 4 months undergoing ablation of AF that had MAF which was present during AF ablation (24/50) or during follow-up (26/50). The incidence of MAF during the index ablation was significantly higher in patients who required ablation of the mitral isthmus as part of the stepwise approach to terminate persistent AF than in those who did not (23 (9/39) versus 8% (5/59), p = 0.04). Following the procedure, MAF was more frequent in patients with prior MI ablation than in those without (41 versus 15%, p = 0.01) [25]. Anousheh et al. showed that the risk of MAF was four times higher if block was not achieved during the first procedure. Although this study had an acute rate of mitral isthmus line block of 83%, four of seven patients had MAF at an average follow-up of 18 ± 5 months due to recovery of conduction across the MI line [33]. Wong et al. found resumption of conduction across the mitral isthmus line to be present in 44% of redo procedures, with long-term maintenance of bidirectional block in 58% of patients who underwent a repeat ablation. After a mean follow-up of 20 ± 9 months, 73% of patients remained free from atrial flutter or tachycardia [12]. Taken together, these data show a large minority of patients develop MAF after a mitral isthmus line when followed out to as far as 20 months.
5 Study limitations
This study has several important limitations that should be acknowledged. First, the studies included in the meta-analysis enrolled heterogeneous populations with variations in study design and ablation protocols, which may limit the generalizability of the results. Second, the decision to perform LAAW or LMI ablation was not standardized but rather based on proceduralist preference in three of the studies. Third, normal values for pre-ablation LAA activation delay were not always defined and it is thus unknown how the effects of endogenous scarring and prior pulmonary vein isolation may play a role in choice of LAAW or LMI lines. Fourth, there was notable heterogeneity in the use of ECGs, Holter monitors, event monitors, loop recorders, or device interrogation at various time intervals, which could have resulted in differential assessment of arrhythmia recurrence rates among studies. Despite these limitations, our study represents the first meta-analysis comparing LAAW and LMI and offers valuable data on the outcomes of these two ablation techniques.
6 Conclusion
Ablation of MAF with LAAW line compared to a LMI line showed no difference in rates of bidirectional block, ablation time, and pericardial effusion. However, LAAW ablation necessitates longer ablation line length and results in delayed LAA activation, while improving maintenance of sinus rhythm in follow-up and foregoing the need for ablation of the CS. These findings suggest that the technique used for MAF ablation should be individualized based on operator experience, patient anatomy, and substrate location.
Abbreviations
- CI:
-
Confidence interval
- CS:
-
Coronary sinus
- LAA:
-
Left atrial appendage
- LAAW:
-
Left atrial anterior wall
- LMI:
-
Lateral mitral isthmus
- MAF:
-
Mitral annular flutter
- MD:
-
Mean difference
- RR:
-
Risk ratio
References
Chae S, Oral H, Good E, Dey S, Wimmer A, Crawford T, et al. Atrial tachycardia after circumferential pulmonary vein ablation of atrial fibrillation: mechanistic insights, results of catheter ablation, and risk factors for recurrence. J Am Coll Cardiol. 2007;50:1781–7.
Gerstenfeld EP, Callans DJ, Dixit S, Russo AM, Nayak H, Lin D, et al. Mechanisms of organized left atrial tachycardias occurring after pulmonary vein isolation. Circulation. 2004;110:1351–7.
Jais P, Hocini M, Hsu LF, Sanders P, Scavee C, Weerasooriya R, et al. Technique and results of linear ablation at the mitral isthmus. Circulation. 2004;110:2996–3002.
Tzeis S, Luik A, Jilek C, Schmitt C, Estner HL, Wu J, et al. The modified anterior line: an alternative linear lesion in perimitral flutter. J Cardiovasc Electrophysiol. 2010;21:665–70.
Makanjee B, Klein GJ, Derval N, Skanes AC. An anterior ablation line is preferred for perimitral flutter after heart transplant. J Cardiovasc Electrophysiol. 2010;21:574–6.
Liberati A, Altman DG, Tetzlaff J, Mulrow C, Gotzsche PC, Ioannidis JP, et al. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. Ann Intern Med. 2009;151:W65–94.
Zhang J, Tang C, Zhang Y, Su XI. Electrophysiologic and clinical consequences of left atrial anterior wall linear ablation in patients with persistent atrial fibrillation. J Cardiovasc Electrophysiol. 2015;26:934–43.
Huemer M, Wutzler A, Parwani AS, Attanasio P, Matsuda H, Blaschke F, et al. Comparison of the anterior and posterior mitral isthmus ablation lines in patients with perimitral annulus flutter or persistent atrial fibrillation. J Interv Card Electrophysiol. 2015;44:119–29.
Pak HN, Oh YS, Lim HE, Kim YH, Hwang C. Comparison of voltage map-guided left atrial anterior wall ablation versus left lateral mitral isthmus ablation in patients with persistent atrial fibrillation. Heart Rhythm. 2011;8:199–206.
Maheshwari A, Shirai Y, Hyman MC, Arkles JS, Santangeli P, Schaller RD, et al. Septal versus lateral mitral isthmus ablation for treatment of mitral annular flutter. JACC Clin Electrophysiol. 2019;5:1292–9.
Makimoto H, Zhang Q, Tilz RR, Wissner E, Cuneo A, Kuck KH, et al. Aborted sudden cardiac death due to radiofrequency ablation within the coronary sinus and subsequent total occlusion of the circumflex artery. J Cardiovasc Electrophysiol. 2013;24:929–32.
Wong KC, Qureshi N, Jones M, Rajappan K, Bashir Y, Betts TR. Mitral isthmus ablation using steerable sheath and high ablation power: a single center experience. J Cardiovasc Electrophysiol. 2012;23:1193–200.
Yeo KK, Davenport J, Raff G, Laird JR. Life-threatening coronary sinus thrombosis following catheter ablation: case report and review of literature. Cardiovasc Revasc Med. 2010;11:262 e1-5.
Abi-Saleh B, Skouri H, Cantillon DJ, Fowler J, Wazni O, Tchou P, et al. Efficacy of ablation at the anteroseptal line for the treatment of perimitral flutter. J Arrhythm. 2015;31:359–63.
Cho Y, Lee W, Park EA, Oh IY, Choi EK, Seo JW, et al. The anatomical characteristics of three different endocardial lines in the left atrium: evaluation by computed tomography prior to mitral isthmus block attempt. Europace. 2012;14:1104–11.
Maurer T, Metzner A, Ho SY, Wohlmuth P, Reissmann B, Heeger C, et al. Catheter ablation of the superolateral mitral isthmus line: a novel approach to reduce the need for epicardial ablation. Circ Arrhythm Electrophysiol. 2017;10.
Ho SY, Anderson RH, Sanchez-Quintana D. Atrial structure and fibres: morphologic bases of atrial conduction. Cardiovasc Res. 2002;54:325–36.
Park JH, Pak HN, Kim SK, Jang JK, Choi JI, Lim HE, et al. Electrophysiologic characteristics of complex fractionated atrial electrograms in patients with atrial fibrillation. J Cardiovasc Electrophysiol. 2009;20:266–72.
Solheim E, Off MK, Hoff PI, Schuster P, Ohm OJ, Chen J. Characteristics and distribution of complex fractionated atrial electrograms in patients with paroxysmal and persistent atrial fibrillation. J Interv Card Electrophysiol. 2010;28:87–93.
Becker AE. Left atrial isthmus: anatomic aspects relevant for linear catheter ablation procedures in humans. J Cardiovasc Electrophysiol. 2004;15:809–12.
Chiang SJ, Tsao HM, Wu MH, Tai CT, Chang SL, Wongcharoen W, et al. Anatomic characteristics of the left atrial isthmus in patients with atrial fibrillation: lessons from computed tomographic images. J Cardiovasc Electrophysiol. 2006;17:1274–8.
Chan CP, Wong WS, Pumprueg S, Veerareddy S, Billakanty S, Ellis C, et al. Inadvertent electrical isolation of the left atrial appendage during catheter ablation of persistent atrial fibrillation. Heart Rhythm. 2010;7:173–80.
Rillig A, Tilz RR, Lin T, Fink T, Heeger CH, Arya A, et al. Unexpectedly high incidence of stroke and left atrial appendage thrombus formation after electrical isolation of the left atrial appendage for the treatment of atrial tachyarrhythmias. Circ Arrhythm Electrophysiol. 2016;9:e003461.
Sanders P, Jais P, Hocini M, Hsu LF, Scavee C, Sacher F, et al. Electrophysiologic and clinical consequences of linear catheter ablation to transect the anterior left atrium in patients with atrial fibrillation. Heart Rhythm. 2004;1:176–84.
Matsuo S, Wright M, Knecht S, Nault I, Lellouche N, Lim KT, et al. Peri-mitral atrial flutter in patients with atrial fibrillation ablation. Heart Rhythm. 2010;7:2–8.
Matsuo S, Yamane T, Date T, Hioki M, Narui R, Ito K, et al. Completion of mitral isthmus ablation using a steerable sheath: prospective randomized comparison with a nonsteerable sheath. J Cardiovasc Electrophysiol. 2011;22:1331–8.
Willems S, Klemm H, Rostock T, Brandstrup B, Ventura R, Steven D, et al. Substrate modification combined with pulmonary vein isolation improves outcome of catheter ablation in patients with persistent atrial fibrillation: a prospective randomized comparison. Eur Heart J. 2006;27:2871–8.
Yokokawa M, Sundaram B, Garg A, Stojanovska J, Oral H, Morady F, et al. Impact of mitral isthmus anatomy on the likelihood of achieving linear block in patients undergoing catheter ablation of persistent atrial fibrillation. Heart Rhythm. 2011;8:1404–10.
Knecht S, Hocini M, Wright M, Lellouche N, O’Neill MD, Matsuo S, et al. Left atrial linear lesions are required for successful treatment of persistent atrial fibrillation. Eur Heart J. 2008;29:2359–66.
Sawhney N, Anand K, Robertson CE, Wurdeman T, Anousheh R, Feld GK. Recovery of mitral isthmus conduction leads to the development of macro-reentrant tachycardia after left atrial linear ablation for atrial fibrillation. Circ Arrhythm Electrophysiol. 2011;4:832–7.
Beinart R, Abbara S, Blum A, Ferencik M, Heist K, Ruskin J, et al. Left atrial wall thickness variability measured by CT scans in patients undergoing pulmonary vein isolation. J Cardiovasc Electrophysiol. 2011;22:1232–6.
Barra S, Gopalan D, Baran J, Fynn S, Heck P, Agarwal S. Acute and sub-acute sinus node dysfunction following pulmonary vein isolation: a case series. Eur Heart J Case Rep. 2018;2:ytx020.
Anousheh R, Sawhney NS, Panutich M, Tate C, Chen WC, Feld GK. Effect of mitral isthmus block on development of atrial tachycardia following ablation for atrial fibrillation. Pacing Clin Electrophysiol. 2010;33:460–8.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Dr. Hsu reports receiving honoraria from Medtronic, Abbott, Boston Scientific, Biotronik, Janssen Pharmaceuticals, and Bristol-Myers Squibb, equity in Acutus Medical, and research grants from Biotronik and Biosense-Webster.
Dr. Ho reports receiving a research grant from Abbott, equity in Vektor Medical, and fellowship support from Medtronic, Abbott, Boston Scientific, and Biotronik.
Dr. Feld reports receiving fellowship training stipend support from Medtronic, St. Jude, Biotronik, Biosense Webster, Boston Scientific, and research grants from Circa Scientific, and Cardix Therapeutics.
Code availability
Stata 13.1 was used for statistical analysis.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Aldaas, O.M., Lupercio, F., Lin, A.Y. et al. Ablation of mitral annular flutter ablation utilizing a left atrial anterior line versus a lateral mitral isthmus line: a systematic review and meta-analysis. J Interv Card Electrophysiol 63, 87–95 (2022). https://doi.org/10.1007/s10840-021-00943-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10840-021-00943-x