Abstract
Purpose
In order to increase the responder rate to CRT, stimulation of the left ventricular (LV) from multiple sites has been suggested as a promising alternative to standard biventricular pacing (BIV). The aim of the study was to compare, in a group of candidates for CRT, the effects of different pacing configurations—BIV, triple ventricular (TRIV) by means of two LV leads, multipoint (MPP), and multipoint plus a second LV lead (MPP + TRIV) pacing—on both hemodynamics and QRS duration.
Methods
Fifteen patients (13 male) with permanent AF (mean age 76 ± 7 years; left ventricular ejection fraction 33 ± 7%; 7 with ischemic cardiomyopathy; mean QRS duration 178 ± 25 ms) were selected as candidates for CRT. Two LV leads were positioned in two different branches of the coronary sinus. Acute hemodynamic response was evaluated by means of a RADI pressure wire as the variation in LVdp/dtmax.
Results
Per patient, 2.7 ± 0.7 veins and 5.2 ± 1.9 pacing sites were evaluated. From baseline values of 998 ± 186 mmHg/s, BIV, TRIV, MPP, and MPP-TRIV pacing increased LVdp/dtmax to 1200 ± 281 mmHg/s, 1226 ± 284 mmHg/s, 1274 ± 303 mmHg, and 1289 ± 298 mmHg, respectively (p < 0.001). Bonferroni post-hoc analysis showed significantly higher values during all pacing configurations in comparison with the baseline; moreover, higher values were recorded during MPP and MPP + TRIV than at the baseline or during BIV and also during MPP + TRIV than during TRIV. Mean QRS width decreased from 178 ± 25 ms at the baseline to 171 ± 21, 167 ± 20, 168 ± 20, and 164 ± 15 ms, during BIV, TRIV, MPP, and MPP-TRIV, respectively (p < 0.001).
Conclusions
In patients with AF, the acute response to CRT improves as the size of the early activated LV region increases.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
A selected group of patients with systolic heart failure (HF) and prolonged QRS may benefit from Cardiac Resynchronization Therapy (CRT) [1, 2]. However, one third of patients referred for this therapy do not show a favorable long-term outcome [3]. One of the reasons for the lack of response is the suboptimal position of the left ventricular (LV) pacing lead [4]. As described in our previous paper [5], we usually target the most electrically delayed site in order to achieve the optimal resynchronization. Novel techniques suggested by some authors are based on the concept that stimulating a larger area of the LV may improve the success rate of CRT. Multisite LV pacing can be carried out with several LV leads in separate coronary sinus (CS) veins [6, 7] or by means of a single multipolar lead capable of delivering multiple stimuli within one CS vein [8,9,10,11]. Our study aimed to compare the acute effects of four pacing configurations in a selected group of patients in permanent atrial fibrillation (AF): biventricular (BIV), multiple site pacing by means of two LV leads (triple ventricular, TRIV), multipoint pacing (MPP) through a single quadripolar lead, and the combination of TRIV and MPP.
2 Methods
Fifteen consecutive patients with permanent AF and indications for the implantation of a CRT-device, in accordance with the European Society of Cardiology/European Heart Rhythm Association (ESC/EHRA) guidelines [12], were enrolled in a prospective study.
In our center, the dual LV site technique is part of common practice in AF patients who are candidates for CRT. We usually optimize the best pacing site during the CRT implantation procedure by means of Q-LV and hemodynamic measurements. The implanting method was described in our previous manuscript [13]. Briefly, the right ventricular (RV) lead was implanted in the mid-septum, according to our standard implantation procedure. Cannulation of the CS and sub-cannulation of all suitable collateral veins were performed by means of a telescopic approach.
Pacing sites were classified as anterior, antero-lateral, lateral, postero-lateral, and posterior segments (in the left anterior oblique view) and as basal, mid, and apical ventricular segments (in the right anterior oblique view), in accordance with the scheme established by Singh et al. [14].
Suitable pacing sites were systematically screened by measuring the local electrical delay (Q-LV) during intrinsic activation of the LV, by means of a BARD Labsystem Pro EP V2.4a (C.R. Bard Inc., Lowell, MA), and the LVdp/dtmax by means of a Certus Pressure Wire and PhysioMon software (St. Jude Medical Systems AB, Uppsala, Sweden) during all pacing configurations and during intrinsic rhythm. LVdP/dtmax at baseline and during the different pacing protocols was calculated over an interval of 15 s; premature ventricular contractions were eliminated electronically. A period of 30 s was allowed to elapse after any change in pacing settings or lead position to allow hemodynamic stabilization. To minimize the impact of respiration and physiological variation, each 15-s LVdP/dtmax value was measured during three separate recordings for each test configuration [15].
In accordance with the guidelines, before CRT implantation, all patients underwent optimization of medical therapy, which included the up-titration of beta-blocker treatment. At the time of implantation, the mean heart rate of the whole population was 59 ± 7 (range 52–71 bpm). With the intention to obtain 100% of ventricular capture, the pacing protocol was set to 10 beats above the basal heart rate (mean pacing rate was 70 ± 4, range 65–80). Twelve-lead QRS morphology was examined beat by beat in the EP system in every pacing setting by an expert electrophysiologist to confirm continuous capture and avoid fusion beats. The first step was to place the quadripolar lead (Quartet™ 1458Q, St. Jude Medical) in every single vein available for cannulation. As per protocol, in every vein we collected data on the Q-LV interval measured from the proximal dipole, defined as LV1 (electrodes M3-P4, inter-dipole length 17 mm), and from the distal dipole, defined as LV2 (electrodes D1-M2 inter-dipole length 20 mm); we also assessed the hemodynamic effect by measuring the LVdp/dtmax at every site. A second bipolar lead, defined as LV3 (Quickflex MicroTM 1258T, St. Jude Medical), was then implanted, usually in an anterior CS vein, and all the measurements were repeated in order to obtain all the possible combinations of pacing between the two leads. We started with BIV pacing with the proximal or distal dipole of the quadripolar lead (RV + LV1 or RV +LV2, depending on the best Q-LV), then TRIV (RV + LV1 + LV3 or RV + LV2 + LV3, depending on the best Q-LV), then MPP (RV + LV1 + LV2) and finally TRIV plus MPP (RV + LV1 + LV2 + LV3) in every vein (an explanatory example of all the data collected is displayed in Fig. 1). To explain our procedure more clearly, if patients had only two available veins, the quadripolar lead was implanted in the one with the longest Q-LV; if 3 or more veins were available, the quadripolar lead was tested in all veins, including those with non-optimal Q-LV. We did not collect data on veins that were visualized but not cannulated. MPP stimulation was performed by means of simultaneous pacing from the two dipoles of the quadripolar lead.
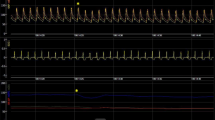
Example of the measurements taken on each patient—this patient was a 73-year-old male with permanent AF, NYHA class III, LBBB, ischemic CMP, and basal QRS 150 ms. a Coronary sinus venous angiography in RAO 20° showing an anterior vein, an antero-lateral vein, and a postero-lateral vein. b Final lead positions; the quadripolar lead is positioned in a postero-lateral vein and the bipolar lead is positioned in an antero-lateral vein. Q-LV measurements at each site are reported. c Final lead positions in LAO view. d Effects on LVdp/dtmax and on QRS width (e) during different pacing protocols. The values at the top of panels d and e indicate the best measurements. The dots in the graphs represent all the data collected for each pacing configuration. Abbreviations are in the text
The sequence of site testing was not randomized in order to reduce as much as possible the procedural time.
The definitive position of the quadripolar lead was the optimal pacing site, defined as the most delayed site in terms of Q-LV, while the second bipolar lead was positioned in a different CS vein, usually in the most anatomically remote vein from the quadripolar lead. We evaluated the effect of the different pacing configurations on hemodynamics and QRS duration, as schematically illustrated in Table 1 and Fig. 2.
Pacing configurations (BIV = RV ± LV1 or BIV = RV ± LV2 depending on the best Q-LV; TRIV = RV ± LV1 ± LV3 or TRIV = RV ± LV2 ± LV3 depending on the best Q-LV; MPP = RV + LV1 + LV2; MPP + TRIV = RV + LV1 + LV2 + LV3)—black dots represent active dipoles, light gray dots represent passive dipoles, and gray circles suggest activation from active dipoles. BIV biventricular pacing (RV pacing plus LV pacing from one of the two dipoles of the quadripolar lead, LV1), TRIV tri-ventricular pacing (RV pacing plus LV1 plus a second LV bipolar pacing lead, LV3), MPP multipoint pacing (RV pacing plus pacing from both dipoles of the quadripolar lead, LV1 and LV2), MPP + TRIV tri-ventricular plus multipoint pacing (pacing from RV, LV1, LV2, and LV3)
In all configurations, pacing was performed by means of a triple-chamber pacing system analyzer (Merlin EX3100 PSA, St. Jude Medical). TRIV was obtained by connecting the bipolar LV lead to the atrial channel of the PSA, and by programming the atrio-ventricular delay to the minimum available value of 25 ms. The RV-LV delay was always set at 0 ms. During MPP, the LV1 and LV2 of the quadripolar lead were paced simultaneously from the LV output of the pacing system analyzer by means of a custom-made epsilon-shaped adapter. TRIV+MPP was obtained by combining the previous configurations.
The two LV leads were implanted definitively, as per protocol in our center. The bipolar LV lead was connected to the atrial port of the device and programmed with the shortest available AV delay (25 ms).
2.1 Statistical analysis
Our aim was to evaluate the improvement in ventricular function, as estimated by the change in LV-dP/dtmax and QRS-width from the basal measurement, obtained by switching the cardiac pacing protocol from single-site BIV, TRIV, MPP to MPP + TRIV pacing in the same subject in a one-arm (intra-patient) study. The instrumental data collected were tabulated along with topographic and protocol information and patients’ clinical characteristics.
We used the “repeated measures analysis of variance” to estimate variations in within-subject measurements of LVdp/dtmax and QRS-width; the Greenhouse-Geisser adjustment was applied to degrees of freedom, and equal weights were attributed to measurements. The Bonferroni correction was used for pairwise planned comparisons between the pacing protocols.
The analyses were conducted on “all measurements” collected from patients (78 measurements).
3 Results
The implantation of two LV leads was successfully performed in all patients. No procedure-related complications were reported. The characteristics of the patient population are shown in Table 2.
The mean procedural time was 174 ± 27 min, and the total fluoroscopy time was 36 ± 8 min. On average, 2.7 ± 0.7 veins per patient were cannulated and 5.2 ± 1.9 pacing sites were evaluated.
The final positions of the LV pacing leads are reported in Table 3.
Conventional BIV pacing resulted in a 202-mmHg/s increase in LVdp/dtmax (from 998 ± 186 to 1200 ± 281 mmHg/s). TRIV pacing caused a small, non-significant, further increase to 1226 ± 284 mmHg/s, whereas MPP and MPP + TRIV increased LVdp/dtmax significantly above the BIV pacing level (to 1274 ± 303 and 1289 ± 298 mmHg/s, respectively; repeated measures ANOVA; p < 0.001 followed by Bonferroni post-hoc analysis). Figure 3 displays the LVdp/dtmax intra-patient response of each of the 15 patients in the cases of the best and worst measurements for every pacing modality. Figure 4 displays the gain in within-subject measurements.
Gain in LVdp/dtmax yielded by the different pacing modes, during biventricular (BIV), triple ventricular (TRIV), multipoint (MPP), and MPP + TRIV pacing protocols vs baseline (considering all pacing sites, 78 series of measurements in the 15 patients)—Bonferroni post-hoc analysis showed significant differences from basal in all pacing configurations; significant differences were observed between BIV and MPP, between BIV and MPP + TRIV, and between TRIV and MPP + TRIV
The effects of the different pacing protocols on QRS width are reported in Fig. 5. TRIV, MPP, and their combination reduced QRS duration significantly in comparison with the baseline.
Effects on QRS width during biventricular (BIV), triple ventricular (TRIV) by means of two LV leads, multipoint (MPP), and multipoint plus second LV lead (MPP-TRIV) pacing protocols vs baseline (considering all pacing sites, 78 measurements)—Bonferroni post-hoc analysis showed significant differences from the baseline in all pacing configurations
Percentage increase in LVdp/dtmax and percentage reduction in QRS width during the different pacing protocols are summarized in Table 4.
p values for difference combinations related to gain in LVdP/dtmax and QRS width during pacing protocols are reported in Tables 5 and 6, respectively.
4 Discussion
Our data indicate that, in CRT patients with AF, pacing from a wider area in the LV acutely increases contractility more than standard BIV does. Most importantly, MPP increases contractility at least as much as TRIV, while it has the advantage of requiring only one LV lead. Combining MPP + TRIV can increase contractility even further, indicating that increasing the area of early activation on the LV increases the hemodynamic effect.
4.1 Increasing the early activated area in the LV
The present study showed a two-step increase in the acute hemodynamic effect of CRT, beyond that due to conventional BIV pacing: a first increase yielded by TRIV or MPP alone and a further increase yielded by combining the two approaches. Because each of the two approaches aims to increase the size of the early activated region, these data support the idea that increasing this region is beneficial for resynchronization; this is also evidenced by a stepwise reduction in QRS duration. Several studies support the idea that QRS narrowing by CRT is a strong determinant of both echocardiographic and clinical response to CRT [16, 17].
While TRIV pacing can be considered to create an early activated region with a largely circumferential orientation (between two contributory venous zones), MPP creates a more baso-apically oriented zone. It is interesting that the orientation of the early activated zone does not seem to matter, as both approaches result in similar hemodynamic and electrophysiological benefit in comparison with conventional BIV pacing.
Finally, the observation that the combination of TRIV and MPP further improves CRT response is a further argument in favor of the idea that increasing the early activated region on the LV increases the effect of CRT. This may raise the question of what the optimal size of this early activated region may be. However, animal studies indicate that the additional hemodynamic benefit of pacing more than four sites may be small [18].
4.2 Multisite vs. multipoint pacing
The idea of pacing from multiple sites has already been expounded upon by various authors [6, 7, 19], who used two leads in two different veins of the CS tree (TRIV). This technique has been demonstrated to be feasible and has yielded promising results from a clinical point of view, providing improvement in ejection fraction (EF) and ventricular remodeling [6]. Other authors have demonstrated a favorable effect of triple-site pacing on NYHA class in comparison with standard dual-site pacing. In a study by Rogers et al. [20], triple-site pacing was obtained by applying dual-site pacing to either the right or left ventricle and proved significantly superior to bipolar pacing in terms of the 6-min walking test, Minnesota living with HF score, ventricular remodeling, and EF.
In a study by Behar [21], 19 patients were paced at two different LV sites by means of two LV leads connected to a bifurcating adapter. While this technique yielded beneficial hemodynamic results on implantation, it also proved problematic in patients in sinus rhythm. Indeed, the two LV leads need to be connected to the device through an adapter and are stimulated from one source, while they may have different impedances. On the other hand, TRIV pacing in patients with AF, as in the study by Leclercq [6] and in our study, is more feasible because the second LV lead is connected to the atrial port. However, this solution is limited by the minimum AV interval, which is between 25 and 30 ms in the majority of commercially available devices.
The introduction of quadripolar technology has enabled a larger area to be paced from a single multipolar lead, and initial experiences documented significant improvements in hemodynamic [8, 9] and mechanical dyssynchrony [10, 11]. Our previous experience [22] extended the concept of the benefit of MPP, in that acute hemodynamic improvement was obtained in every single vein; indeed, on studying 29 patients with an average of 3.2 veins and 6.3 pacing sites per patient, we observed an improvement in LVdp/dtmax at all sites, including the best and worst positions, which suggests that this pacing modality is widely applicable. Our acute results were confirmed in a subsequent study in which MPP, optimized on implantation as previously described, proved significantly superior in terms of remodeling and clinical status to conventional BIV without optimization [23]. Pappone also demonstrated the positive effects of MPP at 3-month and 1-year follow-up examinations [24, 25]. Finally, a large multicenter Italian experience confirmed the favorable effects of MPP in comparison with standard BIV [26]. Recently, the IDE study met the pre-specified hypothesis of non-inferiority of MPP to standard BIV in terms of safety and effectiveness. Moreover, among patients randomized to MPP, those paced from anatomically distant poles displayed a significantly higher rate of response than those paced from close poles [27].
The question of whether acute improvement in LVdP/dtmax predicts long-term clinical benefit needs more evidence. However, our previous experience [23] indicates that, on implantation, acute optimization by means of LVdp/dtmax and electrical delay correlates positively with clinical and remodeling improvement at one-year follow-up. Previously, Duckett found that a 10% increase in LVdp/dtmax on pacing predicted LV reverse remodeling at 6-month follow-up [28].
The present study provides the first head-to-head comparison between the two modalities of pacing a larger area, multisite (TRIV) and multipoint pacing (MPP). The finding that MPP yields at least the same hemodynamic benefit as TRIV pacing suggests that MPP may be preferred to TRIV pacing, as the implantation technique of MPP technology is as simple as that of a bipolar LV lead and avoids placement of a second lead, with its accompanying increased risk of dislodgement.
4.3 Limitations
This was an acute, single-center, non-randomized study; the patient sample size was limited, and the pacing protocol configurations were conditioned by the limitations of the currently available technology. Moreover, as outlined in the methods section, due to the long protocol procedure and in order to reduce as much as possible the duration of the implant, we did not randomize the pacing protocol series. As result, the lack of a randomization of the protocol might have influenced the hemodynamics by introducing linear effects. Furthermore, the acute hemodynamic results may not lead to long-term clinical benefit. The implications of this study should be confirmed in a larger, randomized, prospective, multicenter, long-term study.
5 Conclusions
Pacing from a wider area, as in TRIV and MPP, increases electrophysiological and hemodynamic benefits in comparison with standard BIV pacing, and a further improvement is achieved by TRIV + MPP. This suggests that a larger early activated region increases the efficacy of CRT. In accordance with the concept that “less is more,” it appears that MPP, which requires only one lead, is probably the most convenient technique in terms of risk/benefit ratio.
References
Bristol MR, Saxon LA, Boehmer J, Krueger S, Kass DA, De Marco T, et al. Cardiac-resynchronization therapy with or without an implantable defibrillator in advanced chronic heart failure. N Engl J Med. 2004;350(21):2140–50.
Cleland JG, Daubert JC, Erdmann E, Freemantle N, Gras D, Kappenberger L, et al. The effect of cardiac resynchronization on morbidity and mortality in heart failure. N Engl J Med. 2005;352(15):1539–49.
McAlister FA, Ezekowitz J, Hooton N, Vandermeer B, Spooner C, Dryden DM, et al. Cardiac resynchronization therapy for patients with left ventricular systolic dysfunction: a systematic review. JAMA. 2007;297(22):2502–14.
Mullens W, Grimm RA, Verga T, Dresing T, Starling RC, Wilkoff BL, et al. Insights from a cardiac resynchronization optimization clinic as part of a heart failure disease management program. J Am Coll Cardiol. 2009;53:765–73.
Zanon F, Baracca E, Pastore G, Fraccaro C, Roncon L, Aggio S, et al. Determination of the longest intrapatient left ventricular electrical delay may predict acute hemodynamic improvement in patients after cardiac resynchronization therapy. Circ Arrhythm Electrophysiol. 2014;7:377–83.
Leclercq C, Gadler F, Kranig W, Ellery S, Gras D, Lazarus A, et al. A randomized comparison of triple-site versus dual-site ventricular stimulation in patients with congestive heart failure. J Am Coll Cardiol. 2008;51(15):1455–62.
Lenarczyk R, Kowalski O, Sredniawa B, Pruszkowska-Skrzep P, Mazurek M, Jędrzejczyk-Patej E, et al. Implantation feasibility, procedure-related adverse events and lead performance during 1-year follow-up in patients undergoing triple-site cardiac resynchronization therapy: a substudy of TRUST CRT randomized trial. J Cardiovasc Electrophysiol. 2012;23(11):1228–36.
Pappone C, Ćalović Ž, Vicedomini G, Cuko A, McSpadden LC, Ryu K, et al. Multipoint left ventricular pacing improves acute hemodynamic response assessed with pressure-volume loops in cardiac resynchronization therapy patients. Heart Rhythm. 2014;11(3):394–401.
Thibault B, Dubuc M, Khairy P, Guerra PG, Macle L, Rivard L, et al. Acute haemodynamic comparison of multisite and biventricular pacing with a quadripolar left ventricular lead. Europace. 2013;15(7):984–91.
Osca J, Alonso P, Cano O, Andrés A, Miro V, Tello MJ, et al. The use of multisite left ventricular pacing via quadripolar lead improves acute haemodynamics and mechanical dyssynchrony assessed by radial strain speckle tracking: initial results. Europace. 2016;18(4):560–7.
Rinaldi CA, Kranig W, Leclercq C, Kacet S, Betts T, Bordachar P, et al. Acute effects of multisite left ventricular pacing on mechanical dyssynchrony in patients receiving cardiac resynchronization therapy. J Card Fail. 2013 Nov;19(11):731–8.
European Society of Cardiology (ESC); European Heart Rhythm Association (EHRA), Brignole M, Auricchio A, Baron-Esquivias G, et al. 2013 ESC guidelines on cardiac pacing and cardiac resynchronization therapy: the task force on cardiac pacing and resynchronization therapy of the European Society of Cardiology (ESC). Developed in collaboration with the European Heart Rhythm Association (EHRA). Europace. 2013;15(8):1070–118.
Zanon F, Baracca E, Pastore G, Aggio S, Rigatelli G, Dondina C, et al. Implantation of left ventricular leads using a telescopic catheter system. Pacing Clin Electrophysiol. 2006;29(11):1266–72.
Singh JP, Klein HU, Huang DT, Reek S, Kuniss M, Quesada A, et al. Left ventricular lead position and clinical outcome in the multicenter automatic defibrillator implantation trial-cardiac resynchronization therapy (MADIT-CRT) trial. Circulation. 2011 Mar 22;123(11):1159–66.
Thibault B, Dubuc M, Karst E, Andrade J, Dyrda K, Guerra PG. Design of an acute dP/dt hemodynamic measurement protocol to isolate cardiac effect of pacing. J Card Fail. 2014;20:365–72.
Lecoq G, Leclercq C, Leray E, Crocq C, Alonso C, de Place C, et al. Clinical and electrocardiographic predictors of a positive response to cardiac resynchronization therapy in advanced heart failure. Eur Heart J. 2005;26(11):1094–100.
Korantzopoulos P, Zhang Z, Li G, Fragakis N, Liu T. Meta-analysis of the usefulness of change in QRS width to predict response to cardiac resynchronization therapy. Am J Cardiol. 2016;118(9):1368–73.
Ploux S, Strik M, van Hunnik A, van Middendorp L, Kuiper M, Prinzen FW. Acute electrical and hemodynamic effects of multisite left ventricular pacing for cardiac resynchronization therapy in the dyssynchronous canine heart. Heart Rhythm. 2014;11(1):119–25.
Pappone C, Rosanio S, Oreto G, Tocchi M, Gulletta S, Salvati A, et al. Cardiac pacing in heart failure patients with left bundle branch block: impact of pacing site for optimizing left ventricular resynchronization. Ital Heart J. 2000;1(7):464–9.
Rogers DP, Lambiase PD, Lowe MD, Chow AW. A randomized double-blind crossover trial of triventricular versus biventricular pacing in heart failure. Eur J Heart Fail. 2012;14(5):495–505.
Behar JM, Bostock J, Ginks M, Jackson T, Sohal M, Claridge S, et al. Limitations of chronic delivery of multi-vein left ventricular stimulation for cardiac resynchronization therapy. J Interv Card Electrophysiol. 2015;42(2):135–42.
Zanon F, Baracca E, Pastore G, Marcantoni L, Fraccaro C, Lanza D, et al. Multipoint pacing by a left ventricular quadripolar lead improves the acute hemodynamic response to CRT compared with conventional biventricular pacing at any site. Heart Rhythm. 2015;12(5):975–81.
Zanon F, Marcantoni L, Baracca E, Pastore G, Lanza D, Fraccaro C, et al. Optimization of left ventricular pacing site plus multipoint pacing improves remodeling and clinical response to cardiac resynchronization therapy at 1 year. Heart Rhythm. 2016;13(8):1644–51.
Pappone C, Ćalović Ž, Vicedomini G, Cuko A, McSpadden LC, Ryu K, et al. Multipoint left ventricular pacing in a single coronary sinus branch improves mid-term echocardiographic and clinical response to cardiac resynchronization therapy. J Cardiovasc Electrophysiol. 2015;26(1):58–63.
Pappone C, Ćalović Ž, Vicedomini G, Cuko A, McSpadden LC, Ryu K, et al. Improving cardiac resynchronization therapy response with multipoint left ventricular pacing: twelve-month follow-up study. Heart Rhythm. 2015;12(6):1250–8.
Forleo GB, Santini L, Giammaria M, Potenza D, Curnis A, Calabrese V, et al. Multipoint pacing via a quadripolar left-ventricular lead: preliminary results from the Italian registry on multipoint left-ventricular pacing in cardiac resynchronization therapy (IRON-MPP). Europace. 2017;19(7):1170–7.
Niazi I, Baker J, Corbisiero R, Love C, Martin D, Sheppard R, et al. Safety and efficacy of multipoint pacing in cardiac resynchronization therapy. JACC: Clin Electrophysiol. 2017;492:1510–8. https://doi.org/10.1016/j.jacep.2017.06.022.
Duckett SG, Ginks M, Shetty AK, Bostock J, Gill JS, Hamid S, et al. Invasive acute hemodynamic response to guide left ventricular lead implantation predicts chronic remodeling in patients undergoing cardiac resynchronization therapy. J Am Coll Cardiol. 2011;58:1128–36.
Acknowledgments
We thank Paola Raffagnato, Antonella Tiribello, and Graziano Boaretto (S. Maria Della Misericordia Hospital, Rovigo, Italy) for their technical support in collecting data.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Dr. Zanon has some conflicts of interests, having received modest speaker fees from Boston Scientific, Medtronic, St. Jude Medical, and Livanova. Dr. Prinzen receives research grants from Medtronic, MSD, EBR Systems, and Proteus Biomedical. Domenico Pacetta is an employee of St. Jude Medical Italy. The other authors report no disclosures.
Ethical approval
The local ethics board approved the study.
Informed consent
All patients provided written informed consent.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Zanon, F., Marcantoni, L., Baracca, E. et al. Hemodynamic comparison of different multisites and multipoint pacing strategies in cardiac resynchronization therapies. J Interv Card Electrophysiol 53, 31–39 (2018). https://doi.org/10.1007/s10840-018-0362-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10840-018-0362-y