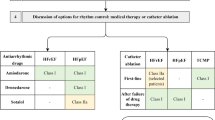
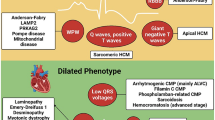
Abstract
Purpose
The effect of the heart failure substrate on the initiation of ventricular fibrillation (VF) and its resulting mechanism is not known. The objective of this study was to determine the effects of substrate on VF initiation and its spatiotemporal organization in the heart failure model.
Methods
Optical action potentials were recorded from LV wedge preparations either from structurally normal hearts (control, n = 11) or from congestive heart failure (CHF; n = 7), at the epicardial surface, endocardial surface which included a papillary muscle, and a transmural cross section. Action potential duration (APD80) was determined, and VF was initiated. A fast Fourier transform was calculated, and the dominant frequency (DF) was determined.
Results
The CHF group showed increased VF vulnerability (69 vs 26 %, p < 0.03), and every mapped surface showed an APD80 gradient which included islands of higher APDs on the transmural surface (M cells) which was not observed in controls. VF in the CHF group was characterized by stable, discrete, high-DF areas that correlated to either foci or spiral waves located on the transmural surface at the site of the papillary muscle. Overall, the top 10 % of DFs correlated to an APD of 101 ms while the bottom 10 % of DFs correlated to an APD of 126 ms (p < 0.01).
Conclusions
In the CHF model, APD gradients correlated with an increased vulnerability to VF, and the highest stable DFs were located on the transmural surface which was not seen in controls. This indicates that the CHF substrate creates unique APD and DF characteristics.








Similar content being viewed by others
Abbreviations
- APD:
-
Action potential duration
- BCL:
-
Basic cycle length
- CHF:
-
Congestive heart failure
- DF:
-
Dominant frequency
- VF:
-
Ventricular fibrillation
References
Everett, T. H., & Olgin, J. E. (2013). Ventricular arrhythmia in structurally remodeled hearts and potential role of myocardial fibrosis. In S. C. Dudley, A. A. Sovari, & A. G. Kocherile (Eds.), Ventricular arrhythmia from principles to patients (pp. 37–50). New York: Nova Science Publishers, Inc.
Anderson, M. E., & Hodgson-Zingman, D. M. (2009). Ventricular tachycardia in patients with heart failure. In D. P. Zipes & J. Jalife (Eds.), Cardiac electrophysiology from cell to bedside (5th ed., pp. 707–716). Philadelphia: Saunders Elsevier.
Holzem, K. M., & Efimov, I. R. (2012). Arrhythmogenic remodelling of activation and repolarization in the failing human heart. Europace, 14(Suppl 5), v50–v57.
Choi, B. R., Liu, T., & Salama, G. (2001). The distribution of refractory periods influences the dynamics of ventricular fibrillation. Circulation Research, 88(5), E49–E58.
Laurita, K. R., Girouard, S. D., & Rosenbaum, D. S. (1996). Modulation of ventricular repolarization by a premature stimulus. Role of epicardial dispersion of repolarization kinetics demonstrated by optical mapping of the intact guinea pig heart. Circulation Research, 79(3), 493–503.
Akar, F. G., & Rosenbaum, D. S. (2003). Transmural electrophysiological heterogeneities underlying arrhythmogenesis in heart failure. Circulation Research, 93(7), 638–645.
Poelzing, S., Akar, F. G., Baron, E., & Rosenbaum, D. S. (2004). Heterogeneous connexin43 expression produces electrophysiological heterogeneities across ventricular wall. American Journal of Physiology. Heart and Circulatory Physiology, 286(5), H2001–H2009.
Yan, G. X., Shimizu, W., & Antzelevitch, C. (1998). Characteristics and distribution of M cells in arterially perfused canine left ventricular wedge preparations. Circulation, 98(18), 1921–1927.
Nattel, S., Antzelevitch, C., & Noble, D. (2011). Resolving the M-cell debate: why and how. Heart Rhythm, 8(8), 1293–1295.
Kim, Y. H., Xie, F., Yashima, M., et al. (1999). Role of papillary muscle in the generation and maintenance of reentry during ventricular tachycardia and fibrillation in isolated swine right ventricle. Circulation, 100(13), 1450–1459.
Pak, H. N., Kim, Y. H., Lim, H. E., Chou, C. C., Miyauchi, Y., Fang, Y. H., Sun, K., Hwang, C., & Chen, P. S. (2006). Role of the posterior papillary muscle and purkinje potentials in the mechanism of ventricular fibrillation in open chest dogs and Swine: effects of catheter ablation. Journal of Cardiovascular Electrophysiology, 17(7), 777–783.
Everett, T. H., Wilson, E. E., Foreman, S., & Olgin, J. E. (2005). Mechanisms of ventricular fibrillation in canine models of congestive heart failure and ischemia assessed by in vivo noncontact mapping. Circulation, 112(11), 1532–1541.
Wu, J., Biermann, M., Rubart, M., & Zipes, D. P. (1998). Cytochalasin D as excitation-contraction uncoupler for optically mapping action potentials in wedges of ventricular myocardium. Journal of Cardiovascular Electrophysiology, 9(12), 1336–1347.
Salama, G., Choi, B. R., Azour, G., Lavasani, M., Tumbev, V., Salzberg, B. M., Patrick, M. J., Ernst, L. A., & Waggoner, A. S. (2005). Properties of new, long-wavelength, voltage-sensitive dyes in the heart. Journal of Membrane Biology, 208(2), 125–140.
Everett, T. H., Kok, L. C., Vaughn, R. H., Moorman, J. R., & Haines, D. E. (2001). Frequency domain algorithm for quantifying atrial fibrillation organization to increase defibrillation efficacy. IEEE Transactions on Biomedical Engineering, 48(9), 969–978.
Nguyen, D. T., Ding, C., Wilson, E., Marcus, G. M., & Olgin, J. E. (2010). Pirfenidone mitigates left ventricular fibrosis and dysfunction after myocardial infarction and reduces arrhythmias. Heart Rhythm, 7(10), 1438–1445.
Liu, D. W., & Antzelevitch, C. (1995). Characteristics of the delayed rectifier current (IKr and IKs) in canine ventricular epicardial, midmyocardial, and endocardial myocytes. A weaker IKs contributes to the longer action potential of the M cell. Circulation Research, 76(3), 351–365.
Sicouri, S., & Antzelevitch, C. (1991). A subpopulation of cells with unique electrophysiological properties in the deep subepicardium of the canine ventricle. The M cell. Circulation Research, 68(6), 1729–1741.
Poelzing, S. (2009). Are electrophysiologically distinct M-cells a characteristic of the wedge preparation? Heart Rhythm, 6(7), 1035–1037.
Dean, J. W., & Lab, M. J. (1990). Regional changes in ventricular excitability during load manipulation of the in situ pig heart. The Journal of Physiology, 429, 387–400.
Burton, F. L., & Cobbe, S. M. (1998). Effect of sustained stretch on dispersion of ventricular fibrillation intervals in normal rabbit hearts. Cardiovascular Research, 39(2), 351–359.
Efimov, I. R., Sidorov, V., Cheng, Y., & Wollenzier, B. (1999). Evidence of three-dimensional scroll waves with ribbon-shaped filament as a mechanism of ventricular tachycardia in the isolated rabbit heart. Journal of Cardiovascular Electrophysiology, 10(11), 1452–1462.
Myles, R. C., Burton, F. L., Cobbe, S. M., & Smith, G. L. (2008). The link between repolarisation alternans and ventricular arrhythmia: does the cellular phenomenon extend to the clinical problem? Journal of Molecular and Cellular Cardiology, 45(1), 1–10.
Laurita, K. R., Wilson, L. D., & Rosenbaum, D. S. (2009). Cardiac alternans as a pathophysiologic mechanism of arrhythmias (5th ed.). Philadelphia, PA: Saunders Elsevier.
Wilson, L. D., Jeyaraj, D., Wan, X., Hoeker, G. S., Said, T. H., Gittinger, M., Laurita, K. R., & Rosenbaum, D. S. (2009). Heart failure enhances susceptibility to arrhythmogenic cardiac alternans. Heart Rhythm, 6(2), 251–259.
Huang, J., Rogers, J. M., Killingsworth, C. R., Walcott, G. P., KenKnight, B. H., Smith, W. M., & Ideker, R. E. (2001). Improvement of defibrillation efficacy and quantification of activation patterns during ventricular fibrillation in a canine heart failure model. Circulation, 103(10), 1473–1478.
Moreno, J., Zaitsev, A. V., Warren, M., Berenfeld, O., Kalifa, J., Lucca, E., Mironov, S., Guha, P., & Jalife, J. (2005). Effect of remodelling, stretch and ischaemia on ventricular fibrillation frequency and dynamics in a heart failure model. Cardiovascular Research, 65(1), 158–166.
Berenfeld, O., & Pertsov, A. M. (1999). Dynamics of intramural scroll waves in three-dimensional continuous myocardium with rotational anisotropy. Journal of Theoretical Biology, 199(4), 383–394.
Li, L., Jin, Q., Huang, J., Cheng, K. A., & Ideker, R. E. (2008). Intramural foci during long duration fibrillation in the pig ventricle. Circulation Research, 102(10), 1256–1264.
Valderrabano, M., Lee, M. H., Ohara, T., Lai, A. C., Fishbein, M. C., Lin, S. F., Karagueuzian, H. S., & Chen, P. S. (2001). Dynamics of intramural and transmural reentry during ventricular fibrillation in isolated swine ventricles. Circulation Research, 88(8), 839–848.
Glukhov, A. V., Fedorov, V. V., Lou, Q., Ravikumar, V. K., Kalish, P. W., Schuessler, R. B., Moazami, N., & Efimov, I. R. (2010). Transmural dispersion of repolarization in failing and nonfailing human ventricle. Circulation Research, 106(5), 981–991.
Voss, F., Opthof, T., Marker, J., Bauer, A., Katus, H. A., & Becker, R. (2009). There is no transmural heterogeneity in an index of action potential duration in the canine left ventricle. Heart Rhythm, 6(7), 1028–1034.
Biermann, M., Rubart, M., Moreno, A., Wu, J., Josiah-Durant, A., & Zipes, D. P. (1998). Differential effects of cytochalasin D and 2,3 butanedione monoxime on isometric twitch force and transmembrane action potential in isolated ventricular muscle: implications for optical measurements of cardiac repolarization. Journal of Cardiovascular Electrophysiology, 9(12), 1348–1357.
Lou, Q., Li, W., & Efimov, I. R. (2012). The role of dynamic instability and wavelength in arrhythmia maintenance as revealed by panoramic imaging with blebbistatin vs. 2,3-butanedione monoxime. American Journal of Physiology. Heart and Circulatory Physiology, 302(1), H262–H269.
Acknowledgments
The authors would like to thank Changyu Shen, PhD, and Hongbo Lin for their help with the statistical analysis.
Author contributions
Thomas Everett was responsible for the concept and design of the experiments and collection of data, performed the data analysis and interpretation, and drafted the manuscript. George Hulley and Roger Chang were trainees who performed the data analysis and interpretation. Ken Lee assisted in the concept and design of the experiments and collection of data. Emily Wilson assisted in the concept and design of the experiments and collection of data. Jeffrey Olgin was responsible for the concept and design of the experiments, interpretation of the data, and generation of the manuscript.
Funding sources
This study received funding from the NIH grant RO1-HL072854 (JEO), RC1 HL099789 (JEO & THE), and AHA Western States Affiliate Beginning Grant-in-Aid 0765177Y (THE).
Conflict of interest
The authors declare that they have no competing interests.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Everett, T.H., Hulley, G.S., Lee, K.W. et al. The effects of remodeling with heart failure on mode of initiation of ventricular fibrillation and its spatiotemporal organization. J Interv Card Electrophysiol 43, 205–215 (2015). https://doi.org/10.1007/s10840-015-0016-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10840-015-0016-2