Abstract
We propose a novel phase based analysis with the purpose of quantifying the periodic bursts of activity observed in various neuronal systems. The way bursts are intiated and propagate in a spatial network is still insufficiently characterized. In particular, we investigate here how these spatiotemporal dynamics depend on the mean connection length. We use a simplified description of a neuron’s state as a time varying phase between firings. This leads to a definition of network bursts, that does not depend on the practitioner’s individual judgment as the usage of subjective thresholds and time scales. This allows both an easy and objective characterization of the bursting dynamics, only depending on system’s proper scales. Our approach thus ensures more reliable and reproducible measurements. We here use it to describe the spatiotemporal processes in networks of intrinsically oscillating neurons. The analysis rigorously reveals the role of the mean connectivity length in spatially embedded networks in determining the existence of “leader” neurons during burst initiation, a feature incompletely understood observed in several neuronal cultures experiments. The precise definition of a burst with our method allowed us to rigorously characterize the initiation dynamics of bursts and show how it depends on the mean connectivity length. Although presented with simulations, the methodology can be applied to other forms of neuronal spatiotemporal data. As shown in a preliminary study with MEA recordings, it is not limited to in silico modeling.









Similar content being viewed by others
References
Barral, J., & Reyes, A. D. (2016). Synaptic scaling rule preserves excitatoryâinhibitory balance and salient neuronal network dynamics. Nature Neuroscience, 19(12), 1690–1696. https://doi.org/10.1038/nn.4415
Beggs, J. M., & Plenz, D. (2003). Neuronal Avalanches in Neocortical Circuits. The Journal of Neuroscience, 23(35), 11167–11177. https://doi.org/10.1523/JNEUROSCI.23-35-11167.2003
Bologna, L. L., Nieus, T., Tedesco, M., Chiappalone, M., Benfenati, F., Martinoia, S. (2010). Low-frequency stimulation enhances burst activity in cortical cultures during development. Neuroscience, 165(3), 692–704. https://doi.org/10.1016/j.neuroscience.2009.11.018
Brette, R., & Gerstner, W. (2005). Adaptive Exponential Integrate-and-Fire Model as an Effective Description of Neuronal Activity. Journal of Neurophysiology, 94(5), 3637–3642. https://doi.org/10.1016/j.neuroscience.2009.11.018
Butts, D. A., Feller, M. B., Shatz, C. J., & Rokhsar, D. S. (1999). Retinal Waves Are Governed by Collective Network Properties. The Journal of Neuroscience, 19(9), 3580–3593. https://doi.org/10.1523/JNEUROSCI.19-09-03580.1999
Chao, Z. C., Bakkum, D. J., Wagenaar, D. A., & Potter, S. M. (2005). Effects of Random External Background Stimulation on Network Synaptic Stability After Tetanization: A Modeling Study. Neuroinformatics, 3(3), 263. https://doi.org/10.1385/NI:3:3:263
Chiappalone, M., Bove, M., Vato, A., Tedesco, M., & Martinoia, S. (2006). Dissociated cortical networks show spontaneously correlated activity patterns during in vitro development. Brain Research, 1093(1), 41–53. https://doi.org/10.1016/j.brainres.2006.03.049
Cotterill, E., Charlesworth, P., Thomas, C. W., Paulsen, O., & Eglen, S. J. (2016). A comparison of computational methods for detecting bursts in neuronal spike trains and their application to human stem cell-derived neuronal networks. Journal of Neurophysiology, 116(2), 306–321. https://doi.org/10.1152/jn.00093.2016
Dayan, P., & Abbott L. F. (2001). Theoretical Neuroscience Computational and Mathematical Modeling of Neural Systems. MIT press,.
Derchansky, M., Jahromi, S. S., Mamani, M., Shin, D. S., Sik, A., & Carlen, P. L. (2008). Transition to seizures in the isolated immature mouse hippocampus: a switch from dominant phasic inhibition to dominant phasic excitation. The Journal of Physiology, 586(2), 477–494. https://doi.org/10.1113/jphysiol.2007.143065
Dhamala, M., Jirsa, V. K., & Ding, M. (2004). Transitions to Synchrony in Coupled Bursting Neurons. Physical Review Letters, 92(2), 028101. https://doi.org/10.1103/PhysRevLett.92.028101
Draguhn, A., Traub, R. D., Schmitz, D., & Jefferys, J. G. R. (1998). Electrical coupling underlies high-frequency oscillations in the hippocampus in vitro. Nature, 394(6689), 189–192. https://doi.org/10.1038/28184
Eckmann, J. P., Jacobi, S., Marom, S., Moses, E., & Zbinden, C. (2008). Leader neurons in population bursts of 2d living neural networks. New Journal of Physics, 10(1), 015011. https://doi.org/10.1088/1367-2630/10/1/01501
Ester, M., Kriegel, H. P., & Xu, X. (1996). A Density-Based Algorithm for Discovering Clusters in Large Spatial Databases with Noise. Kdd, 96(34), 6.
Eytan, D., & Marom, S. (2006). Dynamics and Effective Topology Underlying Synchronization in Networks of Cortical Neurons. Journal of Neuroscience, 26(33), 8465–8476. https://doi.org/10.1523/JNEUROSCI.1627-06.2006
Faci-Lázaro, S., Soriano, J., & Gómez-Gardeñes, J. (2019). Impact of targeted attack on the spontaneous activity in spatial and biologically-inspired neuronal networks. Chaos: An Interdisciplinary Journal of Nonlinear Science, 29(8), 083126.
Fardet, J. (2018). Growth and activity of neuronal cultures. PhD thesis, Paris Diderot, Paris.
Fardet, T. (2019). Silmathoron/NNGT: Version 1.2.0. https://doi.org/10.5281/zenodo.3402494
Gewaltig, M. O., & Diesmann, M. (2007). NEST (NEural Simulation Tool). Scholarpedia, 2(4), 1430. https://doi.org/10.4249/scholarpedia.1430
Grewe, B. F., Langer, D., Kasper, H., Kampa, B. M., & Helmchen, F. (2010). High-speed in vivo calcium imaging reveals neuronal network activity with near-millisecond precision. Nature Methods, 7(5), 399–405. https://doi.org/10.1038/nmeth.1453
Gritsun, T. A., le Feber, J., & Rutten, W. L. C. (2012). Growth Dynamics Explain the Development of Spatiotemporal Burst Activity of Young Cultured Neuronal Networks in Detail. PLoS ONE, 7(9), e43352. https://doi.org/10.1371/journal.pone.0043352
Hernandez-Navarro, L., Orlandi, J. G., Cerruti, B., Vives, E., Soriano, J. (2017). Dominance of Metric Correlations in Two-Dimensional Neuronal Cultures Described through a Random Field Ising Model. Physical Review Letters, 118(20). https://doi.org/10.1103/PhysRevLett.118.208101
Izhikevich, E. (2003). Simple model of spiking neurons. IEEE Transactions on Neural Networks, 14(6), 1569–1572. https://doi.org/10.1109/TNN.2003.820440
Izhikevich, E. (2007). Dynamical systems in neuroscience: the geometry of excitability and bursting. Cambridge, Mass: Computational neuroscience. MIT Press.
Kirst, C., Timme, M., & Battaglia, D. (2016). Dynamic information routing in complex networks. Nature Communications, 7, 11061. https://doi.org/10.1038/ncomms11061
Kitano, K., & Fukai, T. (2007). Variability v.s. synchronicity of neuronal activity in local cortical network models with different wiring topologies. Journal of Computational Neuroscience, 23(2), 237–250. https://doi.org/10.1007/s10827-007-0030-1
Levina, A., & Herrmann, J. M. (2006). Dynamical Synapses Give Rise to a Power-Law Distribution of Neuronal Avalanches. Advances in Neural Information Processing Systems, pages 771–778.
Levina, A., Herrmann, J. M., & Geisel, T. (2007). Dynamical Synapses Causin Self-Organized Criticality in Neural Networks. Nature Physics, 3(12), 857–860.
Lonardoni, D., Amin, H., Di Marco, S., Maccione, A., Berdondini, L., & Nieus, T. (2017). Recurrently connected and localized neuronal communities initiate coordinated spontaneous activity in neuronal networks. PLOS Computational Biology, 13(7), e1005672. https://doi.org/10.1371/journal.pcbi.1005672
Maccione, A., Hennig, M. H., Gandolfo, M., Muthmann, O., van Coppenhagen, J., Eglen, S. J., et al. (2014). Following the ontogeny of retinal waves: pan-retinal recordings of population dynamics in the neonatal mouse: Pan-retinal high-density retinal wave recordings. The Journal of Physiology, 592(7), 1545–1563. https://doi.org/10.1113/jphysiol.2013.262840
Massimini, M., Huber, R., Ferrarelli, F., Hill, S., & Tononi, G. (2004). The Sleep Slow Oscillation as a Traveling Wave. Journal of Neuroscience, 24(31), 6862–6870. https://doi.org/10.1523/JNEUROSCI.1318-04.2004
Mazzoni, A., Broccard, F. D., Garcia-Perez, E., Bonifazi, P., Ruaro, M. E., & Torre, V. (2007). On the Dynamics of the Spontaneous Activity in Neuronal Networks. PLOS ONE, 2(5), e439. https://doi.org/10.1371/journal.pone.0000439
McCormick, D. A., & Contreras, D. (2001). On The Cellular and Network Bases of Epileptic Seizures. Annual Review of Physiology, 63(1), 815–846. https://doi.org/10.1146/annurev.physiol.63.1.815
Muller, L., & Destexhe, A. (2012). Propagating waves in thalamus, cortex and the thalamocortical system: Experiments and models. Journal of Physiology-Paris, 106(5–6), 222–238. https://doi.org/10.1016/j.jphysparis.2012.06.00
Naud, R., Marcille, N., Clopath, C., & Gerstner, W. (2008). Firing patterns in the adaptive exponential integrate-and-fire model. Biological Cybernetics, 99(4–5), 335–347. https://doi.org/10.1007/s00422-008-0264-7
Olmi, S., Petkoski, S., Guye, M., Bartolomei, F., & Jirsa, V. (2019). Controlling seizure propagation in large-scale brain networks. PLOS Computational Biology, 15(2), e1006805. https://doi.org/10.1371/journal.pcbi.1006805
Orlandi, J. G., Soriano, J., Alvarez-Lacalle, E., Teller, S., & Casademunt, J. (2013). Noise focusing and the emergence of coherent activity in neuronal cultures. Nature Physics, 9(9), 582–590. https://doi.org/10.1038/nphys2686
Paraskevov, A., & Zendrikov, D. (2017). A spatially resolved network spike in model neuronal cultures reveals nucleation centers, circular traveling waves and drifting spiral waves. bioRxiv. https://doi.org/10.1101/073981
Pedregosa, F., Varoquaux, G., Gramfort, A., Michel, V., Thirion, B., Grisel, O., Blondel, M., Prettenhofer, P., Weiss, R., Dubourg, V., Vanderplas, J., Passos, A., Cournapeau, D. (2011). Scikit-learn: Machine Learning in Python. Journal of Machine Learning Research p. 6.
Penn, Y., Segal, M., & Moses, E. (2016). Network synchronization in hippocampal neurons. Proceedings of the National Academy of Sciences, 113(12), 3341–3346. https://doi.org/10.1073/pnas.1515105113
Pikovsky, A., Rosenblum, M., & Kurths, J. (2001). Synchronization. A universal concept in nonlinear sciences. Cambridge Nonlinear Science Series. Cambridge University Press, 1 edition.
Renault, R., Durand, J. B., Viovy, J. L., & Villard, C. (2016). Asymmetric axonal edge guidance: a new paradigm for building oriented neuronal networks. Lab on a Chip, 16(12), 2188–2191. https://doi.org/10.1371/journal.pone.0120680
Renault, R., Sukenik, N., Descroix, S., Malaquin, L., Viovy, J. L., Peyrin, J. M., et al. (2015). Combining Microfluidics, Optogenetics and Calcium Imaging to Study Neuronal Communication In Vitro. PLOS ONE, 10(4), e0120680. https://doi.org/10.1039/C6LC00479B
Rouach, N., Segal, M., Koulakoff, A., Giaume, C., & Avignone, E. (2003). Carbenoxolone Blockade of Neuronal Network Activity in Culture is not Mediated by an Action on Gap Junctions. The Journal of Physiology, 553(3), 729–745. https://doi.org/10.1113/jphysiol.2003.053439
Salinas, E., & Sejnowski, T. J. (2001). Correlated Neuronal Activity and the Flow of Neural Information. Nature reviews in Neuroscience, 2(8), 539–550. https://doi.org/10.1038/35086012
Sanchez-Vives, M. V. (2015). Slow wave activity as the default mode of the cerebral cortex. Archives Italiennes de Biologie, 497(23), 69–73. https://doi.org/10.12871/000298292014239
Sanchez-Vives, M. V., Massimini, M., Mattia, M. (2017). Shaping the Default Activity Pattern of the Cortical Network. Neuron, 94(5), 993–1001. https://doi.org/10.1016/j.neuron.2017.05.015
Sanchez-Vives, M. V., McCormick, D. A. (2000). Cellular and network mechanisms of rhythmic recurrent activity in neocortex. Nature Neuroscience, 3(10), 1027–1034. https://doi.org/10.1038/79848
Sipil, S. T., Huttu, K., Voipio, J., & Kaila, K. (2006). Intrinsic bursting of immature CA3 pyramidal neurons and consequent giant depolarizing potentials are driven by a persistent Na+ current and terminated by a slow Ca2+-activated K+ current. European Journal of Neuroscience, 23(9), 2330–2338. https://doi.org/10.1111/j.1460-9568.2006.04757.x
Stegenga, J., Le Feber, J., Marani, E., & Rutten, W. (2008). Analysis of Cultured Neuronal Networks Using Intraburst Firing Characteristics. IEEE Transactions on Biomedical Engineering, 55(4), 1382–1390. https://doi.org/10.1109/TBME.2007.913987
Tazerart, S., Vinay, L., & Brocard, F. (2008). The Persistent Sodium Current Generates Pacemaker Activities in the Central Pattern Generator for Locomotion and Regulates the Locomotor Rhythm. Journal of Neuroscience, 28(34), 8577–8589. https://doi.org/10.1523/JNEUROSCI.1437-08.2008
Tsai, D., Sawyer, D., Bradd, A., Yuste, R., Shepard, K. L. (2017). A very large-scale microelectrode array for cellular-resolution electrophysiology. Nature Communications, 8(1), 1–11. https://doi.org/10.1038/s41467-017-02009-x
Tibau, E., Ludl, A. A., Rdiger, S., Orlandi J. G., & Soriano, J. (2018). Neuronal spatial arrangement shapes effective connectivity traits of in vitro cortical networks. IEEE Transactions on Network Science and Engineering, pages 1–1. https://doi.org/10.1109/TNSE.2018.2862919
Touboul, J., & Destexhe, A. (2010). Can Power-Law Scaling and Neuronal Avalanches Arise from Stochastic Dynamics? PLoS ONE. https://doi.org/10.1371/journal.pone.0008982
Tsai, D., Sawyer, D., Bradd, A., Yuste, R., & Shepard, K. L. (2017). A very large-scale microelectrode array for cellular-resolution electrophysiology. Nature Communications, 8(1), 1–11. https://doi.org/10.1038/s41467-017-02009-x
Tsodyks, M., Uziel, A., & Markram, H. (2000). Synchrony Generation in Recurrent Networks with Frequency-Dependent Synapses. Journal of Neuroscience, 20, 5.
Wang, X. J., & Buzski, G. (1996). Gamma Oscillation by Synaptic Inhibition in a Hippocampal Interneuronal Network Model. The Journal of Neuroscience, 16(20), 6402–6413. https://doi.org/10.1523/JNEUROSCI.16-20-06402.1996
Yaghoubi, M., Graaf, T. D., Orlandi, J. G., Girotto, F., Colicos, M. A., & Davidsen, J. (2018). Neuronal avalanche dynamics indicates different universality classes in neuronal cultures. Scientific Reports, 8(1), 3417. https://doi.org/10.1038/s41598-018-21730-1
Yamamoto, H., Moriya, S., Ide, K., Hayakawa, T., Akima, H., Sato, S., Kubota, S., Tanii, T., Niwano, M., Teller, S., Soriano, J., & Hirano-Iwata, A. (2018). Impact of modular organization on dynamical richness in cortical networks. Science Advances, 4(11), eaau4914. https://doi.org/10.1126/sciadv.aau4914
Zierenberg, J., Wilting, J., & Priesemann, V. (2018). Homeostatic Plasticity and External Input Shape Neural Network Dynamics. Physical Review X, 8(3). https://doi.org/10.1103/PhysRevX.8.031018
Acknowledgements
The authors thanks the University of Paris and the doctoral school, Physique en Île-de-France, for the doctoral fellowship.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Additional information
Action Editor: David Golomb.
Appendices
Appendix
Simulation of neuronal network
1.1 Simulate neuronal activity
Simulations are carried out with the adaptive Exponential Integrate and Fire model (aEIF) (Brette & Gerstner, 2005) via the NNGT python library (Fardet, 2019) and NEST simulator (Gewaltig & Diesmann, 2007). This model is computationally reasonable and provide a large variety of activity patterns (Naud et al., 2008). Each neuron is described as a two dimensional system with the menbrane potential variable \(V_m\) (as in the Integrate-and-Fire model) and an adaptation current w which modulate neurons’ excitability (as in the Izhikevich (2003) model).
Where \(C_m\) is the membrane capacitance, \(E_L\) is the resting potential, \(g_L\) is the leak conductance, \(\Delta _T\) is a potential normalization constant that affect the spiking current, \(V_{th}\) is the soft threshold, \(\tau _w\) is the adaptation time scale, a relates to the sub-threshold adaptation, whereas b gives the spike-triggered adaptation strength and \(V_r\) is the reset potential after the potential \(V_m\) reaches \(V_{peak}\). \(I_e ~\text {and}~ I_s\) are currents that come from respectively external sources or neighboring spikes. The exponential non-linearity model the pre-spike membrane potential sharp increase and is needed to describe in-burst fast dynamics. In the end, we choose this model because it is more biologically relevant than the Izhikevich model (Izhikevich, 2003), and much less complex than the Hodgkin-Huxley model.
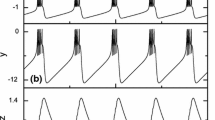
To follow indications of neurons in cultures being oscillators even when uncoupled, reported by Penn et al. (2016), we simulate the activity with three sets of parameters that display pacemaker neurons: Intrinsically Bursting (IB), Regular Spiking (RS) and Noise Driven neurons (ND) whose behavior is detailed in Fig. 9. A neuron said Regular Spiking has a very periodic activity even when submitted to noisy input. Its interspike interval varies by 3% when submitted to a 15 s\(^{-1}\) poisson spike train. A Noise Driven neuron, on the other hand, is much more dependant on the input it receives: its interspike interval varies by 50 % under the same conditions. Intrinsically Bursting neurons present a more complex frequency pattern: high frequencies are super-imposed over a natural small one. This can be seen in the resetting point after a spike: it is below the \(V_m\) nullcline (see Fig. 9). We use in the paper those 3 sets of parameters to show that the presented methods does not depend on specific values.
1.1.1 Model parameters
Table 1 lists all parameters with their values used in the paper.
1.2 Spatial network
To account for the spatial correlations that exist in cultures and shape its activity (Hernandez-Navarro et al., 2017), we use an Exponential Distance Rule (EDR) to connect all neurons. A population of 2000 excitatory neurons is randomly drawn in a circular culture of radius 800 \(\mu\)m. Then, with the same process as an Erdös-Renyi network generation, one connect node i to j with probability: \(p_{ij} = p_0 e^{d_{ij}/\lambda }\), where \(d_{ij}\) is the Euclidean distance between them. This results in a directional network, whose topological properties are predetermined by the magnitude of \(\lambda\), the EDR scale and a sharp border condition: neurons can connect only inside the circular culture.
1.2.1 Post synaptic current
Interactions are modeled as fast current injection into the post-synaptic neuron, following a pre-synaptic spike and a space dependent delay. The delay is set as a 3.0 ms constant plus a spike propagation of velocity 0.1 m.s\(^{-1}\), similar to what has been experimentaly observed in cultures (Barral and Reyes, 2016). Overall, it follows a log-normal distribution of mean 5. to 15. ms for every network. The network metric properties set up both specific connectivity patterns, and the delay in spike propagation with different connection spatial length. Miniature events are also set as a Poisson noise of rate 15 s\(^{-1}\) for each synapses and with a post synaptic current (PSC) of half the amplitude of a spike-triggered PSC.
Synaptic weights that determine the post synaptic current amplitude are set such that the rhythmic activity is observed and stable. Stability of this state is estimated with the mean average interspike interval and network phase.
Analysis with the Izhikevich model, synaptic depression and stochastic inputs
Analysis with the Izhikevich model. Top panel is a raster plot of 100 randomly selected neurons. Middle panel is a trace of the corresponding network phase. Bursts appear as in the paper, between 2 consecutive maxima and minima, however inter burst activity is always high thus the phase stays close to 0.5. Bottom left panel shows the first spike time probability distribution. This distribution shows the lack of first-to-fire dynamics neurons in this example. Middle bottom panel shows the clustering algorithm in space for 3 consecutive time step: visible neurons are represented as red dots, and all other as black dots. The corresponding performance is plotted in the bottom left panel. It shows the characteristic curve of localised growth with a region of initiation representing 14\(\%\) of the total culture
We want to show that our methods can be used to analyze simulations with different models. For examples, previous studies (Orlandi et al., 2013; Levina & Herrmann, 2006; Levina et al., 2007) described the neuronal activity with dynamical synapses and stochastic inputs. More specifically we want to bring together various point of view in the understanding of bursting networks. Orlandi et al proposed a mechanism called noise focusing, based on simulations and experimental recordings, in order to interpret activity during burst initiation. On the other hand, we based our simulations under the assumption that bursting states are an example of oscillator synchronization (Dhamala et al., 2004; Penn et al., 2016).
Inspired by in silico networks in Orlandi et al. (2013), the following simulations are done with an EDR network with mean in-degree 70 and scale 100 \(\mu\)m in a culture of radius 2.5 mm with 5000 neurons, so that the density is 250 mm\(^{-2}\). Following Izhikevich (2003) we look for parameters that display regular spiking neurons, who are not intrinsically spiking (synaptic connection and noise create the activity). This model is represented in its reduced form with the following equation
where v represents the membrane potential and u a membrane recovery variable, which accounts for ionic currents. The parameter a represents the recovery variable time scale, b represents the sub-threshold adaptation, c describes the after-spike polarisation, d the spike-triggered adaptation strenght, and \(I_s(t)\) is the post synaptic current. \(\eta\) is a Gaussian White Noise current of mean value 0 and standard deviation 10 pA. It stays constant for a duration of 5 times the simulation time step, then changes values etc... Miniature events are also set as a Poisson noise of rate 50 s\(^{-1}\). We set the following values: \(a = 0.02\), \(b = 0.25\), \(c = -65\) and \(d = 8\).
Following previous work, (Orlandi et al., 2013; Levina et al., 2007) we consider dynamical synapses with the Tsodyks et al. (2000) model described by the following equations:
where x, y and z are the fractions of synaptic ressources in a (respectively) recovered (ready), active, and inactive state ; \(\tau _{rec}\) is the recovery time scale for synaptic depression and is set to 1.2 s and U determines the decrease of available ressources used by each presynaptic spike and is set to 0.2; \(\tau _{PSC}\) is the post synaptic current time scale and is set to 10 ms. Facilitation has been taken away by setting \(\tau _{facil} = 0\)ms.
It results in a synaptic current for neuron i given by \(I_i = \sum _{j}^{k_i} g_{ij} y_{ij}(t)\), where \(g_{ij}\) is the absolute synaptic strength between i and j. The sum runs over all pre-synaptic neurons of i.
Figures 10 and 11 represent the overall analysis from the network phase maximum detection to the spatial representation of the activity with the neuron’s individual phase. The proposed methodology is here able to pinpoint that this model displays a different spatiotemporal dynamics, not seen with simulations of pacemaker neurons presented in the paper. The spike probability distribution does not display the hallmarks of first-to-fire specific dynamic. Since the global activity is high in between burst a co-activation structured in space as a propagation front can be created without specific initiation. Spatial initiation is still both localised, and structured into a propagation front.
Firing rate and first spike probability distribution
Spike time probability density for two different time references. Right panels show the spike count rate of 4 consecutive bursts aligned on their maximum (bottom) and on an arbitrary 20 Hz threshold crossing time (top). Left panels show the probability distributions with the corresponding time reference. The simulations correspond to Noise Driven neurons, used in the paper in Fig. 5. One can observe that changing the time reference does not strongly change the distribution shape, however here, we cannot observe first to fire behavior anymore
In order to show that the maximum of the phase represents a specific point in the bursting dynamics, we look at a time reference computed with the spike count rate. This firing rate was computed with a convolution with an exponential kernel first (with temporal scale 3 ms), then gaussian kernel (with temporal standard deviation 3 ms). The resulting function was searched for maximum above a certain threshold to detect bursting events. This maximum and a 20 Hz threshold value was then used as time references to compute the first spike probability distribution. Figure 12 shows an example of firing rate and spike time probability distribution for the same simulations as in the paper (Fig. 5) with two time references. The burst definition presented in the paper is specifically designed to look at the spiking pattern during initiation. It gives a time reference related to the network state with information about previous and future spikes and not only spikes in a couple of milliseconds time window. Hence, this time reference stays coherent over consecutive burst in the spike time probability distribution. The arbitrariness in the firing rate threshold method cannot achieve such coherence.
Data and code
Data and code are available in the a github repository:MalloryDazza/NN_Burst_Dynamics.
Activity snapshots
The following figures shows snapshots of the in-burst activity pattern, displayed with neurons individual phases. They correspond to the examples used in the paper (see Figs. 13, 14, 15, and 16).
Representation in space of the burst used for presenting the spatial cluster detection (Fig. 2) in the paper. Neurons’ phases are plot at the soma location. Each frame are separated by 22 milliseconds
Representation in space of the burst used for performance computation (Fig. 5) in the paper (top activity). Neurons’ phases are plot at the soma location. Each frame are separated by 5 milliseconds
Representation in space of the burst used for performance computation (Fig. 5) in the paper (bottom activity). Neurons’ phases are plot at the soma location. Each frame are separated by 4 milliseconds
Representation in space of the burst used for velocity computation (Fig. 6) in the paper (left panel). Neurons’ phases are plot at the soma location. Each frame are separated by 12 milliseconds
Rights and permissions
About this article
Cite this article
Dazza, M., Métens, S., Monceau, P. et al. A novel methodology to describe neuronal networks activity reveals spatiotemporal recruitment dynamics of synchronous bursting states. J Comput Neurosci 49, 375–394 (2021). https://doi.org/10.1007/s10827-021-00786-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10827-021-00786-5