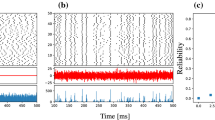
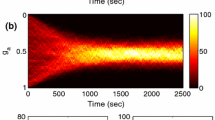
Abstract
Observations of finely-timed spike relationships in population recordings have been used to support partial reconstruction of neural microcircuit diagrams. In this approach, fine-timescale components of paired spike train interactions are isolated and subsequently attributed to synaptic parameters. Recent perturbation studies strengthen the case for such an inference, yet the complete set of measurements needed to calibrate statistical models is unavailable. To address this gap, we study features of pairwise spiking in a large-scale in vivo dataset where presynaptic neurons were explicitly decoupled from network activity by juxtacellular stimulation. We then construct biophysical models of paired spike trains to reproduce the observed phenomenology of in vivo monosynaptic interactions, including both fine-timescale spike-spike correlations and firing irregularity. A key characteristic of these models is that the paired neurons are coupled by rapidly-fluctuating background inputs. We quantify a monosynapse’s causal effect by comparing the postsynaptic train with its counterfactual, when the monosynapse is removed. Subsequently, we develop statistical techniques for estimating this causal effect from the pre- and post-synaptic spike trains. A particular focus is the justification and application of a nonparametric separation of timescale principle to implement synaptic inference. Using simulated data generated from the biophysical models, we characterize the regimes in which the estimators accurately identify the monosynaptic effect. A secondary goal is to initiate a critical exploration of neurostatistical assumptions in terms of biophysical mechanisms, particularly with regards to the challenging but arguably fundamental issue of fast, unobservable nonstationarities in background dynamics.







Similar content being viewed by others
Change history
20 March 2021
A Correction to this paper has been published: https://doi.org/10.1007/s10827-021-00783-8
Notes
To be explicit about the subtlety in the setting here: in fact, we will model E[μ(t)] as time-invariant (Eq. (4)). Thus, for an observer that does not know μ(t), the spike probabilities are time-invariant. On the other hand, for an observer that knows μ(t), the spike probabilities are not time-invariant.
No constraints are placed on the probability distribution of N(B) itself, and this is the source of robustness to nonstationarity, broadly defined. This conditional modeling framework for modeling temporal structure in spike trains is motivated and developed from several points of view in prior work (Amarasingham et al. 2012; Amarasingham et al. 2015; Harrison et al. 2015; Harrison et al. 2013). See Amarasingham et al. (2012) for a thorough introductory exposition. As an example, if B is conditionally a homogeneous Bernoulli process, conditioned on R, then Eq. (7) is satisfied. (The homogeneous Benoulli process approximates the homogeneous Poisson process in discrete time.) But the model is far broader than this. For example, N(B) can be deterministic and Eq. (7) can still be valid (cf., Section A.2 of Amarasingham et al. (2011)).
For intuition, a canonical example can be constructed by generating two distinct independent, background spike trains B1 and B2, which are conditionally uniform, conditioned on N(B1) and N(B2), and superposing a homogeneous Bernoulli process synchronously (with appropriate lag) onto both trains (see Amarasingham et al, 2012, for example, for examples based on Cox processes for the background.)
Note that i) these jitter perturbations are distorted slightly via the role of the interval positions, ii) those distortions are necessary (Platkiewicz et al. 2017), and also iii) such tests can in principle be performed analytically, without surrogate-generation.
Since R is in fact observable, this is a potential observation rather than an assumption.
References
Aertsen, A.M., & Gerstein, G.L. (1985). Evaluation of neuronal connectivity: sensitivity of cross-correlation. Brain research, 340(2), 341–354.
Amarasingham, A., Chen, T.-L., Geman, S., Harrison, M.T., & Sheinberg, D.L. (2006). Spike count reliability and the Poisson hypothesis. Journal of Neuroscience, 26(3), 801–809.
Amarasingham, A., Harrison, M.T., Hatsopoulos, N.G., & Geman, S. (2011). Conditional modeling and the jitter method of spike re-sampling: Supplement. arXiv preprint arXiv:1111.4296.
Amarasingham, A., Harrison, M.T., Hatsopoulos, N.G., & Geman, S. (2012). Conditional modeling and the jitter method of spike resampling. Journal of Neurophysiology, 107(2), 517–531.
Amarasingham, A., Geman, S., & Harrison, M.T. (2015). Ambiguity and nonidentifiability in the statistical analysis of neural codes. Proceedings of the National Academy of Sciences, 112(20), 6455–6460.
Aviel, Y., & Gerstner, W. (2006). From spiking neurons to rate models: A cascade model as an approximation to spiking neuron models with refractoriness. Physical Review E, 73(5), 051908.
Azouz, R., & Gray, C.M. (2003). Adaptive coincidence detection and dynamic gain control in visual cortical neurons in vivo. Neuron, 37(3), 513–523.
Barbosa, J., Stein, H., Martinez, R.L., Galan-Gadea, A., Li, S., Dalmau, J., Adam, K.C.S, Valls-Solé, J, Constantinidis, C., & Compte, A. (2020). Interplay between persistent activity and activity-silent dynamics in the prefrontal cortex underlies serial biases in working memory. Nature Neuroscience, 23, pp 1–9.
Brody, C.D. (1999). Correlations without synchrony. Neural computation, 11(7), 1537–1551.
Brunel, N., Chance, F.S., Fourcaud, N., & Abbott, L.F. (2001). Effects of synaptic noise and filtering on the frequency response of spiking neurons. Physical Review Letters, 86(10), 2186.
Buzsáki, G. (1984). Feed-forward inhibition in the hippocampal formation. Progress in Neurobiology, 22(2), 131–153.
Buzsáki, G. (2006). Rhythms of the brain. Oxford: Oxford University Press.
Casella, G., & Berger, R.L. (2002). Statistical inference. Duxbury Pacific Grove, CA, Vol. 2.
Churchland, M.M., & Abbott, L.F. (2012). Two layers of neural variability. Nature Neuroscience, 15(11), 1472.
Churchland, M.M., Byron, M.Y., Sahani, M., & Shenoy, K.V. (2007). Techniques for extracting single-trial activity patterns from large-scale neural recordings. Current opinion in neurobiology, 17(5), 609–618.
Cossell, L., Lacaruso, M.F., Muir, D.R., Houlton, R., Sader, E.N., Ko, H., Hofer, S.B., & Mrsic-Flogel, T.D. (2015). Functional organization of excitatory synaptic strength in primary visual cortex. Nature, 518(7539), 399.
Csicsvari, J., Hirase, H., Czurko, A., & Buzsáki, G. (1998). Reliability and state dependence of pyramidal cell–interneuron synapses in the hippocampus: an ensemble approach in the behaving rat. Neuron, 21 (1), 179–189.
Date, A., Bienenstock, E., & Geman, S. (1998). On the temporal resolution of neural activity. In Society for Neuroscience Abstracts (Brown University, Division of Applied Mathematics).
Dayan, P., & Abbott, L.F. (2001). Theoretical neuroscience: computational and mathematical modeling of neural systems. Computational Neuroscience Series.
Destexhe, A., Rudolph, M., & Paré, D. (2003). The high-conductance state of neocortical neurons in vivo. Nature Reviews Neuroscience, 4(9), 739.
DeWeese, M.R., & Zador, A.M. (2006). Non-gaussian membrane potential dynamics imply sparse, synchronous activity in auditory cortex. Journal of Neuroscience, 26(47), 12206–12218.
Droste, F., & Lindner, B. (2017). Exact results for power spectrum and susceptibility of a leaky integrate-and-fire neuron with two-state noise. Physical Review E, 95(1), 012411.
Dupret, D., O’Neill, J., & Csicsvari, J. (2013). Dynamic reconfiguration of hippocampal interneuron circuits during spatial learning. Neuron, 78(1), 166–180.
English, D.F., McKenzie, S., Evans, T., Kim, K., Yoon, E., & Buzsáki, G. (2017). Pyramidal cell-interneuron circuit architecture and dynamics in hippocampal networks. Neuron, 96(2), 505–520.
Eyal, G., Mansvelder, H.D., deKock, C.P., & Segev, I. (2014). Dendrites impact the encoding capabilities of the axon. Journal of Neuroscience, 34(24), 8063–8071.
Fontaine, B., Peña, JL, & Brette, R. (2014). Spike-threshold adaptation predicted by membrane potential dynamics in vivo. PLoS Computational Biology, 10(4), e1003560.
Fourcaud, N., & Brunel, N. (2002). Dynamics of the firing probability of noisy integrate-and-fire neurons. Neural Computation, 14(9), 2057–2110.
Fourcaud-Trocmé, N, Hansel, D., VanVreeswijk, C., & Brunel, N. (2003). How spike generation mechanisms determine the neuronal response to fluctuating inputs. The Journal of Neuroscience, 23 (37), 11628–11640.
Fujisawa, S., Amarasingham, A., Harrison, M.T., & Buzsáki, G. (2008). Behavior-dependent short-term assembly dynamics in the medial prefrontal cortex. Nature Neuroscience, 11(7), 823–833.
Gao, P., & Ganguli, S. (2015). On simplicity and complexity in the brave new world of large-scale neuroscience. Current Opinion in Neurobiology, 32, 148–155.
Gerhard, F., Kispersky, T., Gutierrez, G.J., Marder, E., Kramer, M., & Eden, U. (2013). Successful reconstruction of a physiological circuit with known connectivity from spiking activity alone. PLoS Computational Biology, 9(7), e1003138.
Gerstner, W. (2000). Population dynamics of spiking neurons: fast transients, asynchronous states, and locking. Neural Computation, 12(1), 43–89.
Gerstner, W., & Naud, R. (2009). How good are neuron models?. Science, 326(5951), 379–380.
Gerstner, W., Kistler, W.M., Naud, R., & Paninski, L. (2014). Neuronal dynamics: From single neurons to networks and models of cognition. Cambrdige: Cambridge University Press.
Goodman, D., & Brette, R. (2008). Brian: a simulator for spiking neural networks in python. Frontiers in Neuroinformatics, 2(5), 1–10.
Harrison, M.T., & Geman, S. (2009). A rate and history-preserving resampling algorithm for neural spike trains. Neural Computation, 21(5), 1244–1258.
Harrison, M.T., Amarasingham, A., & Kass, R.E. (2013). Statistical identification of synchronous spiking. In P. Di Lorenzo, & J. Victor (Eds.) Spike timing: Mechanisms and function. Taylor & Francis (pp. 77–120).
Harrison, M.T., Amarasingham, A., & Truccolo, W. (2015). Spatiotemporal conditional inference and hypothesis tests for neural ensemble spiking precision. Neural Computation, 27(1), 104–150.
Henze, D.A., & Buzsaki, G. (2001). Action potential threshold of hippocampal pyramidal cells in vivo is increased by recent spiking activity. Neuroscience, 105(1), 121–130.
Herrmann, A., & Gerstner, W. (2001). Noise and the psth response to current transients: I. general theory and application to the integrate-and-fire neuron. Journal of Computational Neuroscience, 11(2), 135–151.
Herrmann, A., & Gerstner, W. (2002). Noise and the psth response to current transients: Ii. integrate-and-fire model with slow recovery and application to motoneuron data. Journal of Computational Neuroscience, 12(2), 83–95.
Imbens, G.W., & Rubin, D.B. (2015). Causal inference in statistics, social, and biomedical sciences. Cambridge: Cambridge University Press.
Isaacson, J.S., & Scanziani, M. (2011). How inhibition shapes cortical activity. Neuron, 72(2), 231–243.
Jazayeri, M., & Afraz, A. (2017). Navigating the neural space in search of the neural code. Neuron, 93(5), 1003–1014.
Jiang, X., Shen, S., Cadwell, C.R., Berens, P., Sinz, F., Ecker, A.S., Patel, S., & Tolias, A.S. (2015). Principles of connectivity among morphologically defined cell types in adult neocortex. Science, 350(6264), aac9462.
Jolivet, R., Kobayashi, R., Rauch, A., Naud, R., Shinomoto, S., & Gerstner, W. (2008). A benchmark test for a quantitative assessment of simple neuron models. Journal of neuroscience methods, 169(2), 417–424.
Jun, J.J., Steinmetz, N.A., Siegle, J.H., Denman, D.J., Bauza, M., Barbarits, B., Lee, A.K., Anastassiou, C.A., Andrei, A., Aydın, C, & et al. (2017). Fully integrated silicon probes for high-density recording of neural activity. Nature, 551(7679), 232–236.
Kass, R.E., Amari, S.-I., Arai, K., Brown, E.N., Diekman, C.O., Diesmann, M., Doiron, B., Eden, U.T., Fairhall, A.L., Fiddyment, G.M., & et al. (2017). Computational neuroscience: Mathematical and statistical perspectives. Annual Review of Statistics and Its Application, 5, 183–214.
Kass, R.E., Eden, U.T., & Brown, E.N. (2014). Analysis of neural data Vol. 491. Berlin: Springer.
Kobayashi, R., Tsubo, Y., & Shinomoto, S. (2009). Made-to-order spiking neuron model equipped with a multi-timescale adaptive threshold. Frontiers in Computational Neuroscience, 3, 9.
Kobayashi, R., Kurita, S., Kurth, A., Kitano, K., Mizuseki, K., Diesmann, M., Richmond, B.J., & Shinomoto, S. (2019). Reconstructing neuronal circuitry from parallel spike trains. Nature Communications, 10(1), 1–13.
Lewis, D. (1974). Causation. The Journal of Philosophy, 70(17), 556–567.
Marshall, L., Henze, D.A., Hirase, H., Leinekugel, X., Dragoi, G., & Buzsáki, G. (2002). Hippocampal pyramidal cell-interneuron spike transmission is frequency dependent and responsible for place modulation of interneuron discharge. Journal of Neuroscience, 22(2), RC197.
Martin, A.B., & von der Heydt, R. (2015). Spike synchrony reveals emergence of proto-objects in visual cortex. Journal of Neuroscience, 35(17), 6860–6870.
Mensi, S., Hagens, O., Gerstner, W., & Pozzorini, C. (2016). Enhanced sensitivity to rapid input fluctuations by nonlinear threshold dynamics in neocortical pyramidal neurons. PLoS Computational Biology, 12(2), e1004761.
Miyawaki, H., & Diba, K. (2016). Regulation of hippocampal firing by network oscillations during sleep. Current Biology, 26(7), 893–902.
Neyman, J.S. (1923). On the application of probability theory to agricultural experiments. essay on principles. section 9.(tlanslated and edited by dm dabrowska and tp speed, statistical science (1990), 5, 465-480). Annals of Agricultural Sciences, 10, 1–51.
Ostojic, S. (2011). Interspike interval distributions of spiking neurons driven by fluctuating inputs. Journal of Neurophysiology, 106(1), 361–373.
Ostojic, S., Brunel, N., & Hakim, V. (2009). How connectivity, background activity, and synaptic properties shape the cross-correlation between spike trains. The Journal of Neuroscience, 29(33), 10234–10253.
Ostojic, S., Szapiro, G., Schwartz, E., Barbour, B., Brunel, N., & Hakim, V. (2015). Neuronal morphology generates high-frequency firing resonance. The Journal of Neuroscience, 35(18), 7056–7068.
Pearl, J. (2009). Causality. Cambridge: Cambridge university press.
Pillow, J.W., Shlens, J., Paninski, L., Sher, A., Litke, A.M., Chichilnisky, E.J., & Simoncelli, E.P. (2008). Spatio-temporal correlations and visual signalling in a complete neuronal population. Nature, 454(7207), 995–999.
Platkiewicz, J., & Brette, R. (2011). Impact of fast sodium channel inactivation on spike threshold dynamics and synaptic integration. PLoS Computational Biology, 7(5), e1001129.
Platkiewicz, J., Stark, E., & Amarasingham, A. (2017). Spike-centered jitter can mistake temporal structure. Neural computation, 29(3), 783–803.
Pouille, F., & Scanziani, M. (2001). Enforcement of temporal fidelity in pyramidal cells by somatic feed-forward inhibition. Science, 293(5532), 1159–1163.
Quilichini, P., Sirota, A., & Buzsáki, G. (2010). Intrinsic circuit organization and theta–gamma oscillation dynamics in the entorhinal cortex of the rat. The Journal of Neuroscience, 30(33), 11128–11142.
Rall, W. (1960). Membrane potential transients and membrane time constant of motoneurons. Experimental Neurology, 2(5), 503–532.
Richardson, M.J.E., & Swarbrick, R. (2010). Firing-rate response of a neuron receiving excitatory and inhibitory synaptic shot noise. Physical Review Letters, 105(17), 178102.
Rossant, C., Kadir, S.N., Goodman, DanFM, Schulman, J., Hunter, MaximilianLD, Saleem, A.B., Grosmark, A., Belluscio, M., Denfield, G.H., Ecker, A.S., & et al. (2016). Spike sorting for large, dense electrode arrays. Nature Neuroscience, 19(4), 634.
Rubin, D.B. (1974). Estimating causal effects of treatments in randomized and nonrandomized studies. Journal of Educational Psychology, 66(5), 688.
Schneidman, E., Berry, M.J., Segev, R., & Bialek, W. (2006). Weak pairwise correlations imply strongly correlated network states in a neural population. Nature, 440(7087), 1007–1012.
Shahidi, N., Andrei, A.R., Hu, M., & Dragoi, V. (2015). High-order coordination of cortical spiking activity modulates perceptual accuracy. Nature Neuroscience, 22(7), 1148.
Sirota, A., Montgomery, S., Fujisawa, S., Isomura, Y., Zugaro, M., & Buzsáki, G. (2008). Entrainment of neocortical neurons and gamma oscillations by the hippocampal theta rhythm. Neuron, 60(4), 683–697.
Smith, M.A., & Kohn, A. (2008). Spatial and temporal scales of neuronal correlation in primary visual cortex. Journal of Neuroscience, 28(48), 12591–12603.
Song, S., Sjöström, P.J., Reigl, M., Nelson, S., & Chklovskii, D.B. (2005). Highly nonrandom features of synaptic connectivity in local cortical circuits. PLoS Biology, 3(3), 507–519.
Stevens, C.F., & Zador, A.M. (1996). When is an integrate-and-fire neuron like a poisson neuron?. In Advances in Neural Information Processing Systems, pp 103–109.
Tan, A.Y., Chen, Y., Scholl, B., Seidemann, E., & Priebe, N.J. (2014). Sensory stimulation shifts visual cortex from synchronous to asynchronous states. Nature, 509(7499), 226–229.
Tchumatchenko, T., Malyshev, A., Wolf, F., & Volgushev, M. (2011). Ultrafast population encoding by cortical neurons. The Journal of Neuroscience, 31(34), 12171–12179.
Ventura, V., Cai, C., & Kass, R.E. (2005). Trial-to-trial variability and its effect on time-varying dependency between two neurons. Journal of Neurophysiology, 94(4), 2928–2939.
Wasserman, L. (2013). All of statistics: a concise course in statistical inference. Berlin: Springer Science & Business Media.
Watson, B.O., Levenstein, D., Greene, J.P., Gelinas, J.N., & Buzsáki, G. (2016). Network homeostasis and state dynamics of neocortical sleep. Neuron, 90(4), 839–852.
Wilent, W.B., & Contreras, D. (2005). Stimulus-dependent changes in spike threshold enhance feature selectivity in rat barrel cortex neurons. Journal of Neuroscience, 25(11), 2983–2991.
Yu, J., & Ferster, D. (2013). Functional coupling from simple to complex cells in the visually driven cortical circuit. The Journal of Neuroscience, 33(48), 18855–18866.
Acknowledgments
We thank G. Buzsáki for providing advice and inspiring our work on this problem, and T. Evans, M. Regnaud, and H. Rotstein for advice and comments. This work was supported by NIMH R01-MH102840 (A.A.), DOD ARO W911NF-15-1-0426 (A.A. and J.P.), PSC-CUNY 68521-00 46 (A.A.), and NIMH K99 MH118423 (S.M.). We warmly acknowledge the hospitality of the Initiative for Theoretical Sciences (ITS) at the CUNY Graduate Center.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interests
The authors declare that they have no conflict of interest.
Additional information
Action Editor: Albert Compte
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Jonathan Platkiewicz and Zachary Saccomano contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Platkiewicz, J., Saccomano, Z., McKenzie, S. et al. Monosynaptic inference via finely-timed spikes. J Comput Neurosci 49, 131–157 (2021). https://doi.org/10.1007/s10827-020-00770-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10827-020-00770-5