Abstract
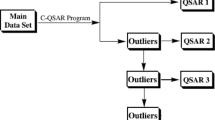
A lead optimization is usually carried out by structure-activity relationship (SAR) and/or quantitative structure-activity relationship (QSAR) studies. One of the assumptions in SAR and QSAR studies is that similar analogs bind to the same binding site in a similar binding mode. One often observes that there are outliers, especially in QSAR. However, most QSAR studies are carried out focusing their attention to the development of QSAR and leave the outliers without much attention. We searched a number of ligand-bound X-ray crystal structures from the protein structure database to find evidences that could indicate a possible source of outliers in SAR or QSAR. Our results show that unusual binding mode could be a source of outliers.











Similar content being viewed by others
References
Kurup A (2003) J Comput Aided Mol Des 17:187–196
BioByte 201 W. 4th St., #204, Claremont, CA 91711-4707. clogp@biobyte.com. 909-624-5992
Berman HM, Westbrook J, Feng Z, Weissig H, Shindyalov IN, Bourne PE (2000) Nucleic Acids Res 28:235–242
CCDC Relibase (version 1.3.2, August 2005), Cambridge Crystallographic Data Center, 12 Union Road, Cambridge CB2 1EZ, United Kingdom
Thompson J, Jeanmougin F (2003) ClustalW multiple sequence alignment program (Version 1.83, June 2003)
Pettersen EF, Goddard TD, Huang CC, Couch GS, Greenblatt DM, Meng EC, Ferrin TE (2004) J Comput Chem 25:1605–1612
Kim KH (1995) In: Hansch C, Fujita T (eds) Classical and three-dimensional QSAR in agrochemistry. American Chemical Society, Washington, DC, vol ACS Symposium Series 606, pp 302–317
Hansch C (1995) In: Hansch C, Fujita T (eds) Classical and three-dimensional QSAR in agrochemistry. American Chemical Society: Washington, DC, vol ACS Symposium Series 606, pp 254–262
Lindskog S (1997) Pharmacol Ther 74:1–20
Kim C-Y, Chang JS, Doyon JB, Baird TT Jr, Fierke CA, Jain A, Christianson DW (2000) J Am Chem Soc 122:12125–12134
Kim CY, Chandra PP, Jain A, Christianson DW (2001) J Am Chem Soc 123:9620–9627
Orville AM, Lipscomb JD, Ohlendorf DH (1997) Biochemistry 36:10052–10066
Orville AM, Elango N, Lipscomb JD, Ohlendorf DH (1997) Biochemistry 36:10039–10051
Elgren TE, Orville AM, Kelly KA, Lipscomb JD, Ohlendorf DH, Que Jr L (1997) Biochemistry 36:11504–11513
Christianson DW, Lipscomb WD (1989) Acc Chem Res 22:62–69
Chung SJ, Kim DH (2001) Bioorg Med Chem 9:185–189
Cho JH, Kim DH, Chung SJ, Ha N-C, Oh B-H, Choi KY (2002) Bioorg Med Chem 10:2015–2022
Massova I, Martin P, de Mel S, Tanaka Y, Edwards B, Mobashery S (1996) J Am Chem Soc 118:12479–12480
Park JD, Kim DH, Woo J-R, Ryu SE (2002) J Med Chem 45:5295–5302
Teplyakov A (1993) Acta Cryst D 49:534–540
Goldstein IM, Ostwald P, Roth S (1996) Vision Res 36:2979–2994
Bredt DS, Snyder SH (1992) Neuron 8:3–11
Li H, Shimizu H, Flinspach ML, Jamal J, Yang W, Xian M, Cai T, Wen EZ, Jia Q, Wang PG, Poulos TL (2002) Biochemistry 41:13868–13875
Reddy AV (1991) Indian J Biochem Biophys 28:10–15
Kester WR (1977) Biochemistry 16:2506–2516
Senda M, Senda T, Ogi T, Kidokoro S (2002) Acta Cryst A58:C278
Skulachev VP (1998) FEBS Lett 423:275–280
Fitzgerald MM (1994) Biochemistry 33:3807–3818
Fitzgerald MM (1996) Nat Struct Biol 3:626–631
Musah RA (1997) Biochemistry 36:11665–11674
Musah RA (1997) J Am Chem Soc. 119:9083–9084
Musah RA (2002) J Mol Biol 315:845–857
Brenk R (2006) J Mol Biol 357:1449–1470
Gomez A (2006) Protein Sci 15:58–64
Gomez GA (2004) Biochemistry 43:4716–4723
Argiriadi MA (1999) Proc Natl Acad Sci USA 96:10637–10642
Argiriadi MA (2000) J Biol Chem 275:15265–15270
Schweitzer BI, Dicker AP, Bertino JR (1990) FABES J 4:2441–2452
Whitlow M, Howard AJ, Stewart D, Hardman KD, Chan JH, Baccanari DP, Tansik RL, Hong JS, Kuyper LF (2001) J Med Chem 44:2928–2932
Whitlow M, Howard AJ, Stewart D, Hardman KD, Kuyper LF, Baccanari DP, Fling ME, Tansik RL (1997) J Biol Chem 272:30289–30298
Patick AK, Potts KE (1998) Clin Microbiol Rev 11:614–627
Hiremath CN, Filman DJ, Grant RA, Hogle JM (1997) Acta Crystallogr D Biol Crystallogr 53:558–570
Grant RA, Hiremath CN, Filman DJ, Syed R, Andries K, Hogle JM (1994) Curr Biol 4:784–797
Turner RB (2001) Antiviral Res 49:1–14
Badger J, Minor I, Oliveira MA, Smith TJ, Rossmann MG (1989) Proteins: Struct Funct Genet 6:1–19
Hadfield AT, Diana GD, Rossmann MG (1999) Proc Natl Acad Sci USA 96:14730–14735
Kim KH, Willingmann P, Gong ZX, Kremer MJ, Chapman MS, Minor I, Oliveira MA, Rossmann MG, Andries K, Diana GD, Dutko FJ, McKinlay MA, Pevear DC (1993) J Mol Biol 230:206–227
Zhang Y, Simpson AA, Ledford RM, Bator CM, Chakravarty S, Skochko GA, Demenczuk TM, Watanyar A, Pevear DC, Rossmann MG (2004) J Virol 78:11061–11069
Hadfield AT, Oliveira MA, Kim KH, Minor I, Kremer MJ, Heinz BA, Shepard D, Pevear DC, Rueckert RR, Rossmann MG (1995) J Mol Biol 253:61–73
Chakravarty S, Bator CM, Pevear DC, Diana GD, Rossmann MG The refined structure of a piconavirus inhibitor currently in clinical trials, when complexed with human rhinovirus 16. to be published
Smith TJ, Kremer MJ, Luo M, Vriend G, Arnold E, Kamer G, Rossmann MG, McKinlay MA, Diana GD, Otto MJ (1986) Science 233:1286–1293
Baldwin ET, Weber IT, Charles RS, Xuan J.-C, Appella E, Yamada M, Matsushima K, Edwards BFP, Clore GM, Gronenborn AM, Wlodawer A (1991) Proc Natl Acad Sci USA 88:502–506
Braun W, Vasak M, Robbins AH, Stout CD, Wagner G, Kagi JHR, Wuthrich K (1992) Proc Natl Acad Sci USA 89:10124–10128
Nilges M, Macias MJ, O’Donoghue SI, Oschkinat H (1997) J Mol Biol 269:408–422
Holak TA, Bode W, Huber J, Otlewski J, Wilusz T (1989) J Mol Biol 210:649–654
Montelione GT, Zheng D, Huang YJ, Gunsalus KC, Szyperski T (2000) Nat Struct Biol 7:982–985
Billeter M (1992) Q Rev Biophys 25:325–377
Hiller N (2005) Protein Sci 15:281–289
Segelke BW, Forstner M, Knapp M, Trakhanov SD, Parkin S, Newhouse YM, Bellamy HD, Weisgraber KH, Rupp B (2000) Protein Sci 9:886–897
Longhi S, Nicolas A, Creveld L, Egmond M, Verrips CT, de Vlieg J, Martinez C, Cambillau C (1996) Proteins: Struct Funct Genet 26:442–458
Acknowledgments
The author express sincere gratitude to both Professor Corwin Hansch and Dr. Albert Leo for their generous permission to use their C-QSAR database and the BioByte program Bio-Loom. The author thanks to Drs. Albert Leo and Yvonne Martin for their critical evaluation of the manuscript and valuable suggestions. The author dedicates this paper to the lifetime advisors and friends, Drs. Corwin Hansch, Albert Leo, Yvonne Martin, and Gary Grunewald.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kim, K.H. Outliers in SAR and QSAR: Is unusual binding mode a possible source of outliers?. J Comput Aided Mol Des 21, 63–86 (2007). https://doi.org/10.1007/s10822-007-9106-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10822-007-9106-2