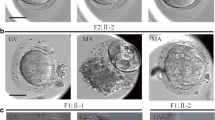
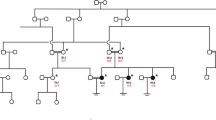
Abstract
Purpose
This study aims to identify the genetic causes of 12 women with primary infertility characterized by primarily oocyte maturation abnormality and consequent early embryonic arrest.
Methods
Genomic DNA was isolated from peripheral blood samples. Whole-exome sequencing was performed on the probands, and the identified variants were confirmed by Sanger sequencing. The pathogenicity of the identified variants on the protein was accessed in silico. And we used qRT-PCR to detect the possible effects of the novel mutation on the mRNA level of NLRP5.
Results
A novel homozygous frameshift variant (p.V429Efs*30) in NLRP5 and compound heterozygous variants with a novel frameshift variant (p.A297Efs*20) and a recurrent variant (c. 223-14_223-2delCCCTCCTGTTCCA) in PATL2 were identified in two unrelated affected individuals. qRT-PCR showed an obvious decrease of the mutant NLRP5 mRNA. In addition, the truncated proteins of NLRP5 and PATL2 were predicted to be non-functional due to the deletion of the most or the whole region of the critical functional domain(s) respectively.
Conclusions
This study identified novel mutations in NLRP5 and PATL2, further expanding the mutational and phenotypic spectrum of both genes. This is the first report of the NLRP5 mutations that associates with oocyte maturation abnormality in humans.


Similar content being viewed by others
References
Bar-Ami S, Zlotkin E, Brandes JM, Itskovitz-Eldor J. Failure of meiotic competence in human oocytes. Biol Reprod. 1994;50:1100–7.
Lu Y, Ferrer-Buitrago M, Popovic M, Neupane J, De Vos WH, Lierman S, et al. Patients with a high proportion of immature and meiotically resistant oocytes experience defective nuclear oocyte maturation patterns and impaired pregnancy outcomes. Reprod Biomed Online. 2018;36:396–407.
Beall S, Brenner C, Segars J. Oocyte maturation failure: a syndrome of bad eggs. Fertil Steril. 2010;94:2507–13.
Hourvitz A, Maman E, Brengauz M, Machtinger R, Dor J. In vitro maturation for patients with repeated in vitro fertilization failure due to “oocyte maturation abnormalities.” Fertil Steril. 2010;94:496–501.
Yanez LZ, Han J, Behr BB, Pera RAR, Camarillo DB. Human oocyte developmental potential is predicted by mechanical properties within hours after fertilization. Nat Commun. 2016;7:10809.
Li L, Zheng P, Dean J. Maternal control of early mouse development. Development. 2010;137:859–70.
Feng R, Sang Q, Kuang Y, Sun X, Yan Z, Zhang S, et al. Mutations in TUBB8 and human oocyte meiotic arrest. N Engl J Med. 2016;374:223–32.
Chen B, Zhang Z, Sun X, Kuang Y, Mao X, Wang X, et al. Biallelic mutations in PATL2 cause female infertility characterized by oocyte maturation arrest. Am J Hum Genet. 2017;101:609–15.
Maddirevula S, Coskun S, Alhassan S, Elnour A, Alsaif HS, Ibrahim N, et al. Female infertility caused by mutations in the oocyte-specific translational repressor PATL2. Am J Hum Genet. 2017;101:603–8.
Huang L, Tong X, Wang F, Luo L, Jin R, Fu Y, et al. Novel mutations in PATL2 cause female infertility with oocyte germinal vesicle arrest. Hum Reprod. 2018;33:1183–90.
Christou-Kent M, Kherraf ZE, Amiri-Yekta A, Le Blevec E, Karaouzene T, Conne B, et al. PATL2 is a key actor of oocyte maturation whose invalidation causes infertility in women and mice. EMBO Mol Med. 2018;10:e8515.
Zhang Z, Li B, Fu J, Li R, Diao F, Li C, et al. Bi-allelic missense pathogenic variants in TRIP13 cause female infertility characterized by oocyte maturation arrest. Am J Hum Genet. 2020;107:15–23.
Sang Q, Zhou Z, Mu J, Wang L. Genetic factors as potential molecular markers of human oocyte and embryo quality. J Assist Reprod Genet. 2021;38:993–1002.
Alazami AM, Awad SM, Coskun S, Al-Hassan S, Hijazi H, Abdulwahab FM, et al. TLE6 mutation causes the earliest known human embryonic lethality. Genome Biol. 2015;16:240.
Xu Y, Shi Y, Fu J, Yu M, Feng R, Sang Q, et al. Mutations in PADI6 cause female infertility characterized by early embryonic arrest. Am J Hum Genet. 2016;99:744–52.
Parry DA, Logan CV, Hayward BE, Shires M, Landolsi H, Diggle C, et al. Mutations causing familial biparental hydatidiform mole implicate c6orf221 as a possible regulator of genomic imprinting in the human oocyte. Am J Hum Genet. 2011;89:451–8.
Mu J, Wang W, Chen B, Wu L, Li B, Mao X, et al. Mutations in NLRP2 and NLRP5 cause female infertility characterised by early embryonic arrest. J Med Genet. 2019;56:471–80.
Docherty LE, Rezwan FI, Poole RL, Turner CL, Kivuva E, Maher ER, et al. Mutations in NLRP5 are associated with reproductive wastage and multilocus imprinting disorders in humans. Nat Commun. 2015;6:8086.
Sparago A, Verma A, Patricelli MG, Pignata L, Russo S, Calzari L, et al. The phenotypic variations of multi-locus imprinting disturbances associated with maternal-effect variants of NLRP5 range from overt imprinting disorder to apparently healthy phenotype. Clin Epigenetics. 2019;11:190.
Xu Y, Qian Y, Liu Y, Wang Q, Wang R, Zhou Y, et al. A novel homozygous variant in NLRP5 is associate with human early embryonic arrest in a consanguineous Chinese family. Clin Genet. 2020;98:69–73.
Zheng W, Hu H, Dai J, Zhang S, Gu Y, Dai C, et al. Expanding the genetic and phenotypic spectrum of the subcortical maternal complex genes in recurrent preimplantation embryonic arrest. Clin Genet. 2021;99:286–91.
Maddirevula S, Awartani K, Coskun S, AlNaim LF, Ibrahim N, Abdulwahab F, et al. A genomics approach to females with infertility and recurrent pregnancy loss. Hum Genet. 2020;139:605–13.
Li M, Jia M, Zhao X, Shi R, Xue X. A new NLRP5 mutation causes female infertility and total fertilization failure. Gynecol Endocrinol. 2021;37:283–4.
Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods. 2021;25:402–8.
Mendell JT, Sharifi NA, Meyers JL, Martinez-Murillo F, Dietz HC. Nonsense surveillance regulates expression of diverse classes of mammalian transcripts and mutes genomic noise. Nat Genet. 2004;36:1073–8.
Wu L, Chen H, Li D, Song D, Chen B, Yan Z, et al. Novel mutations in PATL2: expanding the mutational spectrum and corresponding phenotypic variability associated with female infertility. J Hum Genet. 2019;64:379–85.
Acknowledgements
We thank all families for participating in this study.
Funding
This study was supported by the National Natural Science Foundation of China (81801440 to L.H.), the Natural Science Foundation of Anhui Province (1808085MH241 to L.H.), the Fundamental Research Funds for the Central Universities (WK9110000028 to L.H.), and the National Key R&D Program of China (2018YFC1003900 to X.T.).
Author information
Authors and Affiliations
Contributions
L.H., R.J., and X.T. conceived and designed the experiments. L.H., F.L., Q.J, J.J., L.L., R.J., and X.T. collected the samples. L.H. and Y.W. performed the experiments. L.H., Y.W., and G.S. analyzed the data. L.H. wrote the paper. All authors approved the final manuscript.
Corresponding authors
Ethics declarations
Ethics approval
The study was approved by the biomedical research ethics committee of the University of Science and Technology of China.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Huang, L., Wang, Y., Lu, F. et al. Novel mutations in NLRP5 and PATL2 cause female infertility characterized by primarily oocyte maturation abnormality and consequent early embryonic arrest. J Assist Reprod Genet 39, 711–718 (2022). https://doi.org/10.1007/s10815-022-02412-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10815-022-02412-4