Abstract
Purpose
Male infertility is a complex multifactorial pathological condition, and asthenozoospermia (AZS) is one of the most common causes. Current evidence suggests the underlying role of the circadian clock on male fertility. This study aims to evaluate the expression levels of five principal clock genes in the sperm and their correlations with the sperm parameters in male infertility.
Methods
We determined the expression profiles of BMAL1, CLOCK, CRY1, PER1, and PER2 in the sperm of infertile men with AZS (n=38) and healthy fertile men (n=40) using quantitative real-time PCR. Then we performed comprehensive association analyses on the clock gene levels and the sperm parameters, including progressive and total motility, concentration, and normal morphology of the sperm.
Results
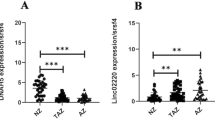
Our results showed that the expression levels of five clock genes (BMAL1, CLOCK, CRY1, PER1, and PER2) are significantly decreased in the sperm of the infertile men with AZS as compared with that of healthy fertile men (P< 0.01). All five clock gene levels are associated with the percentage of progressive/total sperm motility (r= 0.546/0.589~0.677/0.695, P< 0.01). We also discovered that a combination of BMAL1, CLOCK, CRY1, PER1, and PER2 could reach a high diagnostic performance (areas under the curves, 92%) for infertility with AZS.
Conclusions
This study first reports that sperm BMAL1, CLOCK, CRY1, PER1, and PER2 levels are altered in AZS and may be molecular markers for male infertility with AZS. These findings indicate the possibility of stabilizing circadian rhythmicity through therapeutic intervention on clock genes to prevent and treat infertility.



Similar content being viewed by others
References
Petok WD. Infertility counseling (or the lack thereof) of the forgotten male partner. Fertil Steril. 2015;104(2):260–6.
Coutton C, Fissore RA, Palermo GD, Stouffs K, Touré A. Male infertility: genetics, mechanism, and therapies. Biomed Res Int. 2016;2016:7372362.
Shahrokhi SZ, Salehi P, Alyasin A, Taghiyar S, Deemeh MR. Asthenozoospermia: cellular and molecular contributing factors and treatment strategies. Andrologia. 2020;52(2):e13463. https://doi.org/10.1111/and.13463.
Rybar R, Kopecka V, Prinosilova P, Markova P, Rubes J. Male obesity and age in relationship to semen parameters and sperm chromatin integrity. Andrologia. 2011;43(4):286–91.
Salas-Huetos A, Bullo M, Salas-Salvado J. Dietary patterns, foods and nutrients in male fertility parameters and fecundability: a systematic review of observational studies. Hum Reprod Update. 2017;23(4):371–89.
Hosseinpour E, Shahverdi A, Parivar K, et al. Sperm ubiquitination and DNA fragmentation in men with occupational exposure and varicocele. Andrologia. 2014;46(4):423–9.
Zhang B, Ma H, Khan T, et al. A DNAH17 missense variant causes flagella destabilization and asthenozoospermia. J Exp Med. 2020;217(2):e20182365. https://doi.org/10.1084/jem.20182365.
Ghandehari-Alavijeh R, Zohrabi D, Tavalaee M, Nasr-Esfahani MH. Association between expression of TNF-alpha, P53 and HIF1alpha with asthenozoospermia. Hum Fertil (Camb). 2019;22(2):145–51.
Peterlin A, Kunej T, Peterlin B. The role of circadian rhythm in male reproduction. Curr Opin Endocrinol Diabetes Obes. 2019;26(6):313–6.
Roenneberg T, Merrow M. The circadian clock and human health. Curr Biol. 2016;26(10):R432–43.
Pan X, Taylor MJ, Cohen E, Hanna N, Mota S. Circadian clock, time-restricted feeding and reproduction. Int J Mol Sci. 2020;21(3):831. https://doi.org/10.3390/ijms21030831.
Cypryjański J. Changes in seasonality of births in Poland in the years 1900–2009. Demog Res. 2019;40:1441–54.
Saint Pol P, Beuscart R, Leroy-Martin B, Hermand E, Jablonski W. Circannual rhythms of sperm parameters of fertile men. Fertil Steril. 1989;51(6):1030–3.
Xie M, Utzinger KS, Blickenstorfer K, Leeners B. Diurnal and seasonal changes in semen quality of men in subfertile partnerships. Chronobiol Int. 2018;35(10):1375–84.
Ni W, Liu K, Hou G, et al. Diurnal variation in sperm DNA fragmentation: analysis of 11,382 semen samples from two populations and in vivo animal experiments. Chronobiol Int. 2019;36(11):1455–63.
Rijo-Ferreira F, Takahashi JS. Genomics of circadian rhythms in health and disease. Genome Med. 2019;11(1):1–16. https://doi.org/10.1186/s13073-019-0704-0.
Takahashi JS. Transcriptional architecture of the mammalian circadian clock. Nat Rev Genet. 2017;18(3):164–79. https://doi.org/10.1038/nrg.2016.150.
Hergenhan S, Holtkamp S, Scheiermann C. Molecular interactions between components of the circadian clock and the immune system. J Mol Biol. 2020;432(12):3700–13. https://doi.org/10.1016/j.jmb.2019.12.044.
Sen A, Hoffmann HM. Role of core circadian clock genes in hormone release and target tissue sensitivity in the reproductive axis. Mol Cell Endocrinol. 2020;501:110655. https://doi.org/10.1016/j.mce.2019.110655.
Schoeller EL, Clark DD, Dey S, et al. Bmal1 is required for normal reproductive behaviors in male mice. Endocrinol. 2016;157(12):4914–29.
Li C, Xiao S, Hao J, Liao X, Li G. Cry1 deficiency leads to testicular dysfunction and altered expression of genes involved in cell communication, chromatin reorganization, spermatogenesis, and immune response in mouse testis. Mol Reprod Dev. 2018;85(4):325–35. https://doi.org/10.1002/mrd.22968.
Organization WHO. WHO laboratory manual for the examination and processing of human semen. Geneva, Switzerland: World Health Organization Press; 2010. p. 226.
Lunenfeld B, Mskhalaya G, Zitzmann M, et al. Recommendations on the diagnosis, treatment and monitoring of testosterone deficiency in men. Aging Male. 2021;24(1):119–38.
Cox KH, Takahashi JS. Circadian clock genes and the transcriptional architecture of the clock mechanism. J Mol Endocrinol. 2019;63(4):R93–102.
Liu B, Wang P, Wang Z, et al. Analysis and difference of voltage-dependent anion channel mRNA in ejaculated spermatozoa from normozoospermic fertile donors and infertile patients with idiopathic asthenozoospermia. J Assist Reprod Genet. 2010;27(12):719–24.
Lambard S, Galeraud-Denis I, Martin G, Levy R, Chocat A, Carreau S. Analysis and significance of mRNA in human ejaculated sperm from normozoospermic donors: relationship to sperm motility and capacitation. Mol Hum Reprod. 2004;10(7):535–41.
Sciarra F, Franceschini E, Campolo F, et al. Disruption of circadian rhythms: a crucial factor in the etiology of infertility. Int J Mol Sci. 2020;21(11):3943.
Morse D, Cermakian N, Brancorsini S, Parvinen M, Sassone-Corsi P. No circadian rhythms in testis: Period1 expression is clock independent and developmentally regulated in the mouse. Mol Endocrinol. 2003;17(1):141–51.
Shen O, Ding X, Nie J, et al. Variants of the CLOCK gene affect the risk of idiopathic male infertility in the Han-Chinese population. Chronobiol Int. 2015;32(7):959–65. https://doi.org/10.3109/07420528.2015.1056305.
Zhang J, Ding X, Li Y, et al. Association of CLOCK gene variants with semen quality in idiopathic infertile Han-Chinese males. Reprod Biomed Online. 2012;25(5):536–42.
Li R, Cheng S, Wang Z. Circadian clock gene plays a key role on ovarian cycle and spontaneous abortion. Cell Physiol Biochem. 2015;37(3):911–20.
Kumar S, Deb R, Singh U, et al. Bovine circadian locomotor output cycles kaput (CLOCK) and clusterin (CLU) mRNA quantitation in ejaculated crossbred bull spermatozoa. Reprod Domest Anim. 2015;50(3):505–9.
Liu XX, Cai L, Liu FJ. An in silico analysis of human sperm genes associated with asthenozoospermia and its implication in male infertility. Medicine (Baltimore). 2018;97(49):e13338.
Caballero-Campo P, Lira-Albarrán S, Barrera D, et al. Gene transcription profiling of astheno- and normo-zoospermic sperm subpopulations. Asian J Androl. 2020;22(6):608–15.
Bansal SK, Gupta N, Sankhwar SN, Rajender S. Differential genes expression between fertile and infertile spermatozoa revealed by transcriptome analysis. PLoS One. 2015;10(5):e0127007.
Author information
Authors and Affiliations
Contributions
PZ and YL designed the project of this manuscript, and PZ and CL carried out all the experiments. PZ and YG contributed to statistical analyses and results interpretation. PZ and YL contributed to the drafting of the manuscript. YL revised the paper. All authors edited and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Ethics approval
This study has been granted ethical approval by the Ethics Committee of The Affiliated Zhongshan Hospital of Dalian University (approval number: 2019326).
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zhang, P., Li, C., Gao, Y. et al. Altered circadian clock gene expression in the sperm of infertile men with asthenozoospermia. J Assist Reprod Genet 39, 165–172 (2022). https://doi.org/10.1007/s10815-021-02375-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10815-021-02375-y