Abstract
Purpose
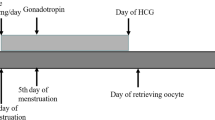
This study aimed to evaluate the impact of luteal phase ovarian stimulation (LPS) on the outcomes of assisted reproductive technology (ART) for infertile couples and patients desiring non-urgent egg cryopreservation.
Methods
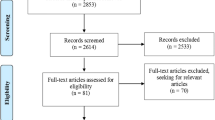
We included all studies reported patients who received LPS and that used follicular phase ovarian stimulation (FPS) as a comparison group until January 2021. Prior meta-analysis regarding the outcomes of LPS in double stimulation and fertility preservation have already been published, so these studies were excluded. Risk of Bias in Non-randomized Studies of Interventions was used to assess the study quality. The study was registered in the International Prospective Register of Systematic Reviews database (CRD42020183946).
Results
Twelve studies with a total of 4433 patients were included. The regimen employed can be categorized into two groups, but there is currently no evidence to support one over the other. After we excluded the largest study, the clinical pregnancy rate and live birth rate were similar after FPS and LPS. There were significantly more stimulation days and total gonadotropins used in the LPS group. After subgroup analysis, we found that poor responders received significantly more cumulus oocyte complexes (+0.64) in the LPS group.
Conclusion
Current evidence indicates that patients in the LPS group could achieve pregnancy outcomes non-inferior to those in the FPS group. Because of current debate over freeze-all policy and the limited data about live birth rate, the universal use of LPS is considered controversial. In the future, more well-designed studies are necessary to investigate the indications for LPS and its cost-effectiveness.




Similar content being viewed by others
References
Massin N. New stimulation regimens: endogenous and exogenous progesterone use to block the LH surge during ovarian stimulation for IVF. Hum Reprod Update. 2017;23(2):211–20. https://doi.org/10.1093/humupd/dmw047.
Heikinheimo O, Gordon K, Williams RF, Hodgen GD. Inhibition of ovulation by progestin analogs (agonists vs antagonists): preliminary evidence for different sites and mechanisms of actions. Contraception. 1996;53(1):55–64. https://doi.org/10.1016/0010-7824(95)00255-3.
von Wolff M, Thaler CJ, Frambach T, Zeeb C, Lawrenz B, Popovici RM, et al. Ovarian stimulation to cryopreserve fertilized oocytes in cancer patients can be started in the luteal phase. Fertil Steril. 2009;92(4):1360–5. https://doi.org/10.1016/j.fertnstert.2008.08.011.
Boots CE, Meister M, Cooper AR, Hardi A, Jungheim ES. Ovarian stimulation in the luteal phase: systematic review and meta-analysis. J Assist Reprod Genet. 2016;33(8):971–80. https://doi.org/10.1007/s10815-016-0721-5.
Sfakianoudis K, Pantos K, Grigoriadis S, Rapani A, Maziotis E, Tsioulou P, et al. What is the true place of a double stimulation and double oocyte retrieval in the same cycle for patients diagnosed with poor ovarian reserve? A systematic review including a meta-analytical approach. J Assist Reprod Genet. 2020;37(1):181–204. https://doi.org/10.1007/s10815-019-01638-z.
Nakasuji T, Kawai K, Ishikawa T, Teraoka K, Takeuchi S, Miyagawa T, et al. Random-start ovarian stimulation with aromatase inhibitor for fertility preservation in women with Japanese breast cancer. Reprod Med Biol. 2019;18(2):167–72. https://doi.org/10.1002/rmb2.12263.
Castillo J, Llacer J, Delgado R, Guerrero J, Bernabeu R. Ovarian hyperstimulation syndrome following GnRH agonist trigger for final follicular maturation in a patient undergoing random start ovarian stimulation for egg-donation cycle with an inadvertent concomitant early pregnancy. Gynecol Endocrinol. https://doi.org/10.1080/09513590.2019.1707178.
Alexander VM, Martin CE, Schelble AP, Laufer AB, Hardi A, McKenzie LJ, et al. Ovarian stimulation for fertility preservation in women with cancer: a systematic review and meta-analysis comparing random and conventional starts. J Gynecol Obstet Hum Reprod. 2021;50(8):102080. https://doi.org/10.1016/j.jogoh.2021.102080.
Moher D, Liberati A, Tetzlaff J, Altman DG. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. Ann Intern Med. 2009;151(4):264–9, w64. https://doi.org/10.7326/0003-4819-151-4-200908180-00135.
Cozzolino M, Cecchino GN, Troiano G, Romanelli C. Growth hormone cotreatment for poor responders undergoing in vitro fertilization cycles: a systematic review and meta-analysis. Fertil Steril. 2020;114(1):97–109. https://doi.org/10.1016/j.fertnstert.2020.03.007.
Sterne JA, Hernán MA, Reeves BC, Savović J, Berkman ND, Viswanathan M, et al. ROBINS-I: a tool for assessing risk of bias in non-randomised studies of interventions. Bmj. 2016;355:i4919. https://doi.org/10.1136/bmj.i4919.
Rstudio software v1.2. Available online: https://rstudio.com/products/rstudio/download/. Accessed 30 Sep 2019.
Pereira N, Voskuilen-Gonzalez A, Hancock K, Lekovich JP, Schattman GL, Rosenwaks Z. Random-start ovarian stimulation in women desiring elective cryopreservation of oocytes. Reprod BioMed Online. 2017;35(4):400–6. https://doi.org/10.1016/j.rbmo.2017.06.002.
Jin B, Niu Z, Xu B, Chen Q, Zhang A. Comparison of clinical outcomes among dual ovarian stimulation, mild stimulation and luteal phase stimulation protocols in women with poor ovarian response. Gynecol Endocrinol. 2018;34(8):694–7. https://doi.org/10.1080/09513590.2018.1435636.
Higgins JPT, Thomas J, Chandler J, Cumpston M, Li T, Page MJ, Welch VA. Cochrane Handbook for Systematic Reviews of Interventions version 6.1. Cochrane, 2020. http://www.training.cochrane.org/handbook. Accessed 12 Sep 2020.
Wang N, Wang Y, Chen Q, Dong J, Tian H, Fu Y, et al. Luteal-phase ovarian stimulation vs conventional ovarian stimulation in patients with normal ovarian reserve treated for IVF: a large retrospective cohort study. Clin Endocrinol. 2016;84(5):720–8. https://doi.org/10.1111/cen.12983.
Campos Olmedo LM, López Rioja MDJ, Sánchez González CM, Zavala González PN, Recio López Y, Chávez BA. Luteal phase stimulation and fertility: first outcomes. Gazzetta Medica Italiana Archivio per le Scienze Mediche. 2019;178(7-8):580–7. https://doi.org/10.23736/S0393-3660.18.03873-1.
Kuang Y, Hong Q, Chen Q, Lyu Q, Ai A, Fu Y, et al. Luteal-phase ovarian stimulation is feasible for producing competent oocytes in women undergoing in vitro fertilization/intracytoplasmic sperm injection treatment, with optimal pregnancy outcomes in frozen-thawed embryo transfer cycles. Fertil Steril. 2014;101(1):105–11. https://doi.org/10.1016/j.fertnstert.2013.09.007.
Wang T, Sun Z, Lim JP, Yu Y. Comparison of luteal phase ovulation induction and ultra-short gonadotropin-releasing hormone agonist protocols in older patients undergoing in vitro fertilization. Libyan J Med. 2019;14(1):1597327. https://doi.org/10.1080/19932820.2019.1597327.
Llacer J, Moliner B, Luque L, Bernabeu A, Lledo B, Castillo JC, et al. Luteal phase stimulation versus follicular phase stimulation in poor ovarian responders: results of a randomized controlled trial. Reprod Biol Endocrinol. 2020;18(1). https://doi.org/10.1186/s12958-020-00570-7.
Buendgen NK, Schultze-Mosgau A, Cordes T, Diedrich K, Griesinger G. Initiation of ovarian stimulation independent of the menstrual cycle: a case-control study. Arch Gynecol Obstet. 2013;288(4):901–4. https://doi.org/10.1007/s00404-013-2794-z.
Martínez F, Clua E, Devesa M, Rodríguez I, Arroyo G, González C, et al. Comparison of starting ovarian stimulation on day 2 versus day 15 of the menstrual cycle in the same oocyte donor and pregnancy rates among the corresponding recipients of vitrified oocytes. Fertil Steril. 2014;102(5):1307–11. https://doi.org/10.1016/j.fertnstert.2014.07.741.
Lin LT, Vitale SG, Chen SN, Wen ZH, Tsai HW, Chern CU, et al. Luteal phase ovarian stimulation may improve oocyte retrieval and oocyte quality in poor ovarian responders undergoing in vitro fertilization: preliminary results from a single-center prospective pilot study. Adv Ther. 2018;35(6):847–56. https://doi.org/10.1007/s12325-018-0713-1.
Wei LH, Ma WH, Tang N, Wei JH. Luteal-phase ovarian stimulation is a feasible method for poor ovarian responders undergoing in vitro fertilization/intracytoplasmic sperm injection-embryo transfer treatment compared to a GnRH antagonist protocol: a retrospective study. Taiwan J Obstet Gynecol. 2016;55(1):50–4. https://doi.org/10.1016/j.tjog.2015.07.001.
Li Y, Yang W, Chen X, Li L, Zhang Q, Yang D. Comparison between follicular stimulation and luteal stimulation protocols with clomiphene and HMG in women with poor ovarian response. Gynecol Endocrinol. 2016;32(1):74–7. https://doi.org/10.3109/09513590.2015.1081683.
Qin NX, Chen QJ, Hong QQ, Cai RF, Gao HY, Wang Y, et al. Flexibility in starting ovarian stimulation at different phases of the menstrual cycle for treatment of infertile women with the use of in vitro fertilization or intracytoplasmic sperm injection. Fertil Steril. 2016;106(2):334–U143. https://doi.org/10.1016/j.fertnstert.2016.04.006.
Wu Y, Zhao FC, Sun Y, Liu PS. Luteal-phase protocol in poor ovarian response: a comparative study with an antagonist protocol. J Int Med Res. 2017;45(6):1731–8. https://doi.org/10.1177/0300060516669898.
Zhang W, Wang M, Wang S, Bao H, Qu Q, Zhang N, et al. Luteal phase ovarian stimulation for poor ovarian responders. JBRA Assist Reprod. 2018;22(3):193–8. https://doi.org/10.5935/1518-0557.20180045.
Ferraretti AP, La Marca A, Fauser BC, Tarlatzis B, Nargund G, Gianaroli L. ESHRE consensus on the definition of ‘poor response’ to ovarian stimulation for in vitro fertilization: the Bologna criteria. Hum Reprod. 2011;26(7):1616–24. https://doi.org/10.1093/humrep/der092.
Blockeel C, Campbell A, Coticchio G, Esler J, Garcia-Velasco JA, Santulli P, et al. Should we still perform fresh embryo transfers in ART? Hum Reprod. 2019;34(12):2319–29. https://doi.org/10.1093/humrep/dez233.
Shi Y, Sun Y, Hao C, Zhang H, Wei D, Zhang Y, et al. Transfer of fresh versus frozen embryos in ovulatory women. N Engl J Med. 2018;378(2):126–36. https://doi.org/10.1056/NEJMoa1705334.
Roque M, Haahr T, Geber S, Esteves SC, Humaidan P. Fresh versus elective frozen embryo transfer in IVF/ICSI cycles: a systematic review and meta-analysis of reproductive outcomes. Hum Reprod Update. 2019;25(1):2–14. https://doi.org/10.1093/humupd/dmy033.
Sunkara SK, Rittenberg V, Raine-Fenning N, Bhattacharya S, Zamora J, Coomarasamy A. Association between the number of eggs and live birth in IVF treatment: an analysis of 400 135 treatment cycles. Hum Reprod. 2011;26(7):1768–74. https://doi.org/10.1093/humrep/der106.
Roque M, Valle M, Sampaio M, Geber S. Does freeze-all policy affect IVF outcome in poor ovarian responders? Ultrasound Obstet Gynecol. 2018;52(4):530–4. https://doi.org/10.1002/uog.19000.
Cobo A, Garrido N, Crespo J, José R, Pellicer A. Accumulation of oocytes: a new strategy for managing low-responder patients. Reprod BioMed Online. 2012;24(4):424–32. https://doi.org/10.1016/j.rbmo.2011.12.012.
Kuang Y, Chen Q, Hong Q, Lyu Q, Ai A, Fu Y, et al. Double stimulations during the follicular and luteal phases of poor responders in IVF/ICSI programmes (Shanghai protocol). Reprod BioMed Online. 2014;29(6):684–91. https://doi.org/10.1016/j.rbmo.2014.08.009.
Vaiarelli A, Cimadomo D, Conforti A, Schimberni M, Giuliani M, D’Alessandro P, et al. Luteal phase after conventional stimulation in the same ovarian cycle might improve the management of poor responder patients fulfilling the Bologna criteria: a case series. Fertil Steril. 2020;113(1):121–30. https://doi.org/10.1016/j.fertnstert.2019.09.012.
Suikkari AM, Tulppala M, Tuuri T, Hovatta O, Barnes F. Luteal phase start of low-dose FSH priming of follicles results in an efficient recovery, maturation and fertilization of immature human oocytes. Hum Reprod. 2000;15(4):747–51. https://doi.org/10.1093/humrep/15.4.747.
Chen H, Wang Y, Lyu Q, Ai A, Fu Y, Tian H, et al. Comparison of live-birth defects after luteal-phase ovarian stimulation vs. conventional ovarian stimulation for in vitro fertilization and vitrified embryo transfer cycles. Fertil Steril. 2015;103(5):1194–201.e2. https://doi.org/10.1016/j.fertnstert.2015.02.020.
Ubaldi FM, Capalbo A, Vaiarelli A, Cimadomo D, Colamaria S, Alviggi C, et al. Follicular versus luteal phase ovarian stimulation during the same menstrual cycle (DuoStim) in a reduced ovarian reserve population results in a similar euploid blastocyst formation rate: new insight in ovarian reserve exploitation. Fertil Steril. 2016;105(6):1488–95.e1. https://doi.org/10.1016/j.fertnstert.2016.03.002.
Zolfaroli I, Ferriol GA, Mora JH, Cano A. Impact of progestin ovarian stimulation on newborn outcomes: a meta-analysis. J Assist Reprod Genet. 2020;37(5):1203–12. https://doi.org/10.1007/s10815-020-01755-0.
Acknowledgements
We would like to express our gratitude for the help from Taipei Medical University, Division of Reproductive Medicine, Department of Obstetrics and Gynecology, Taipei Medical University Hospital, and Ministry of Science and Technology, Taiwan. Also, we would like to thank Online English for editing and proofreading this manuscript.
Funding
Ministry of Science and Technology, Taiwan (No. 106-2314-B-038-076, No. 107-2314-B-038-058, No. 108-2314-B-038-095) and Taipei Medical University (TMU109-F-002)
Author information
Authors and Affiliations
Contributions
BJ Lu: chart collection, data analysis, manuscript writing.
CJ Lin: data analysis, manuscript writing.
BZ Lin: manuscript writing.
Li Huang: data analysis.
LT Chien: database searching.
CH Chen: data collection, manuscript editing.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM 1
(DOCX 1477 kb)
Rights and permissions
About this article
Cite this article
Lu, BJ., Lin, CJ., Lin, BZ. et al. ART outcomes following ovarian stimulation in the luteal phase:a systematic review and meta-analysis. J Assist Reprod Genet 38, 1927–1938 (2021). https://doi.org/10.1007/s10815-021-02237-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10815-021-02237-7