Abstract
Green seaweed species of the genus Ulva are promising protein sources for food production. Various factors affect the biochemical composition of Ulva spp. (hereafter Ulva), which can complicate the utilization in a commercial scale food production. This study investigated the composition of Ulva harvested in 2021 in May (dark green/black biomass), June (light green biomass) and August (mixed colored biomass). The August harvest was divided into three different color codes: light green, green, and dark green/black, to explore a relation between color and composition as an indicator for biomass quality. The crude protein (CP) and ash contents were highest in the dark August biomass (16.4±0.4% and 35.9±3.0% of dry matter (DM), respectively) and in the dark biomass harvested in May (20.6±0.5% and 41.2±3.9% of DM, respectively). The light green Ulva harvested in June had a lower CP content (6.5±1.5% of DM) similarly to what was observed for light green biomass in August (7.7±0.1% of DM). The light green biomasses were found to have a higher nitrogen to amino acid (N-to-AA) conversion factor and proportion of essential amino acids (EAA). The lipid content ranged between 1.0-2.2±0.1-0.2% of DM for the different biomasses and a higher lipid content was related with a lighter biomass color. Phosphorus and potassium concentrations were highest in the green fraction, whereas the darker green/black biomass had higher contents of all other elements, including the toxic elements, arsenic and cadmium.
Graphical abstract

Similar content being viewed by others
Explore related subjects
Find the latest articles, discoveries, and news in related topics.Avoid common mistakes on your manuscript.
Introduction
Seaweeds are gaining interest as an alternative biomass for food production due to their high productivity, bioremediation capacity, biochemical composition and lack of need for land and freshwater resources (van Den Burg et al. 2016; Bleakley and Hayes 2017; Neveux et al. 2018). The green seaweeds belonging to the Ulva genus are promising sources for food production, due to their high content of protein (6-29% of DM) and high proportion of EAA (Fleurence 1999; Magnusson et al. 2019; Juul et al. 2022b). By now, the genus Ulva is comprised of 130 taxonomically accepted species distributed worldwide (Guiry and Guiry 2020). Various factors can affect the biochemical composition of Ulva, such as species, geographic origin, season, growth stage and environmental factors (Bikker et al. 2020; Toth et al. 2020; Queirós et al. 2021; Jansen et al. 2022). Besides protein, Ulva consists of carbohydrates (9-62% of DM), ash (17-60% of DM) and lipids (0.5-8% of DM) (Yaich et al. 2011; Cherry et al. 2019; Jansen et al. 2022; Juul et al. 2022b). These large variations in composition can complicate the application of seaweed on a commercial scale, where control of quality, efficacy and traceability is of utmost importance (Hafting et al. 2011). Juul (2022) investigated Ulva from Skive Fjord, Denmark in 2019 and 2020 during June to September, where the variations in composition were most likely attributed to the availability of nutrients, temperature and irradiance, which is in alignment with previous observations (Bruhn et al. 2011; Toth et al. 2020). Higher temperature and irradiance resulted in a decrease in protein content, whereas addition of dissolved inorganic nitrogen increased the protein and fatty acid content while decreasing the ash content (Toth et al. 2020). For wild growth, nitrogen has been found to be the limiting factor for growth of Ulva fenestrata (formerly believed to be Ulva lactuca in North Atlantic regions (Hughey et al. 2019)) in summer, when light is not the limiting factor. Moreover, lower availability of inorganic nitrogen in Roskilde Fjord, Denmark during the summer months with high irradiance, resulted in a lower protein content in U. fenestrata (Pedersen and Borum 1996). The same tendency has also been observed by Rouxel et al. (2001), showing lower protein content in summer and higher levels in autumn and winter. This means that the protein content is reduced or diluted in the tissue during growth when nitrogen is limiting, while the carbohydrate content is high as protein and carbohydrate concentrations are always reciprocal (Martinez and Rico 2002). For U. lactuca, the biochemical composition was found to be more affected by abiotic factors during time of season than by their maturity stage (Unis et al. 2023). Especially, the protein, AA, and monosaccharide content was affected by seasonal factors. A higher protein and AA content was observed in winter, whereas the monosaccharide content was increased upon summer, reflecting the protein and carbohydrate content. While glutamic acid, aspartic acid and alanine had the highest concentrations in all seasons and harvesting ages, no relation in terms of EAA content upon season could be established (Unis et al. 2023). The nitrogen and hence the protein content has been found to relate with the color of U. lactuca (Robertson-Anderson et al. 2010) and U. fenestrata (Stedt et al. 2022a), where a darker green color was related with a higher nitrogen content and a higher chlorophyll content. To use the color of Ulva at the time of harvest as an indicator for biomass quality would be an advantageous tool for the industry in food and feed. So far, the color is only described in relation to the nitrogen content, and it would be valuable if it could be related to other factors in the biochemical composition.
The aim of this study was to investigate Ulva harvested in Skive Fjord, Denmark during late spring and summer (May, June, and August 2021) to explore a possible connection between color and the biochemical composition besides nitrogen, such as ash, lipid and the amino acid profile.
Materials and methods
Chemicals and reagents
Milli-Q water was deionized (18.2 MΩ) filtered water (0.22 μM) from a Milli-Q system, Millipore SAS (France). HCl 37% was obtained from Merck (Germany) and petroleum ether from Fisher Scientific (Denmark).
Harvest and color separation
Wild Ulva was harvested May 18, and June 21, 2021, by hand (approx. 200 kg per harvest for multiple studies) and the 10th of August 2021 by a specialized harvesting boat by Hede Denmark (approx. 3 t for multiple studies). All harvests were performed in Skive Fjord, Denmark. The harvest by hand was performed in the inner areas of the fjord (0-1 m depth), whereas the harvest by boat was further out but still in the inner areas of the fjord (1-2 m depth). The boat was equipped with a transport belt that was able to collect the seaweed from the entire water column at depths of 1-2 m with a maximum capacity of 1 t per batch. The harvest dates were chosen due to availability of biomass, weather conditions and logistics. The harvest consisted of different Ulva species, where the dominating foliose species likely was Ulva compressa, as this was the dominating species observed in inner Skive Fjord in September 2021 (Unpublished results). Furthermore, the harvest could consist of other minor bycatches in the form of mussels, shrimps, fish and debris of terrestrial origin, and sediments from the fjord, why all biomasses were washed. Biomass from May and June was washed (tap water) by hand with a ratio of biomass:water of 1:2 in smaller buckets, whereas the biomass from August was washed in pilot scale using a large basin with stirring caused by the water hose. Hence, the exact ratio of biomass to water from the latter is unknown. Approximately 10 kg of biomass from August was separated into three color codes by visual inspection: Light green, green and dark green/black. These color codes were afterwards determined by a colorimeter. From all biomasses, triplicate portions of 0.5 kg were selected randomly and frozen at –20 °C on the day of harvest and subsequently freeze dried and grinded to powder using a mini chopper.
Color measurement
The color of the different biomasses was measured using a handheld colorimeter (Konica-Minolta, Japan) to investigate the color components L* (black-white), a* (green-red), and b* (blue-yellow). The biomass powders were analyzed using a petri-dish (D = 5.3 cm), filling to a height of 0.5 cm. All biomasses were analysed in triplicates.
DM, CP, and ash content
The biomasses were analyzed in terms of DM, CP and ash in triplicates. The DM content was measured after freeze-drying using a moisture analyzer (HR73 Halogen Moisture Analyzer, Mettler Toledo, USA). The analyzed moisture content was added to the moisture loss happening during freeze drying, thereby being able to calculate the DM content of the fresh biomass. Total nitrogen was analyzed by Dumas combustion (DUMATHERM, Gerhart Analytical Systems, Germany) using 1.4 mg O2 mg-1 sample and an O2 flowrate of 200 mL min-1. The CP content was calculated using a N-to-protein conversion factor of 5 according to Angell et al. (2016). Ash content was measured by heating 0.5 g of sample at 550 °C for 5 h using a muffle furnace.
Lipid content
The lipid content of the biomasses was quantified in triplicates by digesting one gram of sample with boiling hydrochloric acid to break the lipo-protein bonds using a Hydrotherm (Gerhardt Analytical systems, Germany). The resulting solution was collected in a filter, dried, and the lipid was subsequently extracted with petroleum ether using a Soxtherm (Gerhardt Analytical systems, Germany). The lipid content was calculated based on the initial sample weight and the weight at the end of the analysis after drying.
Amino acids
The different biomasses were analyzed for amino acids (AA) by Eurofins, Denmark. The AA analysis without tryptophan (DI004) was chosen together with an oxidative hydrolysis (DJ011) for cystine and methionine. The following AA were analyzed: Alanine, arginine, aspartic acid, cysteine + cystine, glutamic acid, glycine, histidine, isoleucine, leucine, lysine, methionine, phenylalanine, proline, serine, threonine, tyrosine, and valine. The sum of the AAs is referred to as total AAs (TAA). It should be noted that tryptophan is not included in this analysis and hence the true TAA would be slightly higher. One analysis per biomass was performed due to project limitations, where each replicate was pooled to include the variations.
Elemental composition
The element analyzes were performed using Inductively Coupled Plasma Mass Spectrometry (ICP-MS). A series of procedural blanks and triplicates of Certified Reference Material (CRM) (NIST 3232 Kelp Powder (Thallus laminariae)) were prepared along with the samples for analytical quality assessment and control. Aliquots of 300 mg seaweed powder were digested in 4 mL Milli-Q water and 4 mL Merck Suprapure HNO3 mixtures in an Anton Paar Multiwave 7000 microwave oven (220 °C: 30 min). Digestion solutions were subsequently diluted with Milli-Q water to approximately 60 g, weighted and analyzed using an Agilent 7900 ICP-MS for the elements listed in Table 1. The detection limits reported in Table 1 were determined as 3 standard deviations (SD) on the series of blank samples. The relative SD (in %) of duplicate samples (n=4x2) for the following key elements: calcium (Ca) (8-12), magnesium (Mg) (1-2), phosphorus (P) (1-6), potassium (K) (0-4), sodium (Na) (0-4), iron (Fe) (1-11), zinc (Zn) (1-4), cobalt (Co) (2-8), manganese (Mn) (0-5), copper (Cu) (5-7), nickel (Ni) (1-4), selenium (Se) (0-7), arsenic (As) (2-5), cadmium (Cd) (2-4), mercury (Hg) (0-7) and lead (Pb) (3-9). The CRM recovery % (±SD) (n=11) for the key elements were: Ca (126±18), Mg (108±13), P* (152±26), K (105±13), Na (102±11), Fe (101±7), Zn (94±3), Co* (95±4), Mn (93±9), Cu (91±6), As (104±6), Cd (106±3), Hg (99±11) and Pb (97±6). The elements listed with an * above do not have a certified value, only a reference value, and CRM recovery % values are therefore more uncertain. There are no values for Ni and Se in NIST 3232. All elements are measured as the total amount, including both inorganic and organic. The maximum intake of the different biomasses was calculated based on the content of toxic elements iAs, Cd, Hg and Pb (Table 3). Maximum tolerable intake of these elements has been determined by The European Food Safety Authority (EFSA). An averaged weighed adult (in Europe, 70.8 kg) was used as an example (Walpole et al. 2012). The iAs content was assumed to account for 1-70% of tAs in the investigated biomasses as it is found to vary highly (Holdt and Kraan 2011).
Statistical analysis
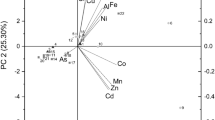
Results are presented as means ± SD. One-way analysis of variance (ANOVA) was carried out to test the significance of difference between the different biomasses. Differences were regarded as significant at a minimum level at 95% (P < 0.05). A Duncan’s Multiple Range test was used as a post-hoc test to designate significantly different averages. Normal distribution and homogeneity of variance of data was confirmed using QQ-plot and Shapiro test (P > 0.05), and Bartlett test (P > 0.05), respectively. Statistical analysis was carried out using RStudio (R Core Team 2021). Furthermore, multivariate data analysis was conducted for all samples and variables using SIMCA 16 (MKS Data Analytics Solutions, Sweden). Data was unit-variance (UV) scaled prior to analysis. A biplot was used to evaluate connections between samples and variables.
Results
Color and DM content
Wild Ulva biomass appeared dark green/black in May, light green in June and a mix of color in August 2021 (Fig. 1 a-c). The division of the biomass upon color in August is shown in Fig. 1 d-f. The light green biomass from June and the light green fraction from August were both found to have higher values of a*, b* and L* (Fig. 2), indicating a color of more green, more light and more yellow, respectively. On the other hand, the dark green/black biomass from May and the dark green/black biomass from August had lowest values of a*, b* and L*, indicating a darker biomass overall. Fig. 3 illustrates the DM content of the biomass according to month of harvest (a) and color in August (b). Ulva harvested in May and August had a similar DM content of 15.5±3.0% and 15.6±1.1%, respectively, whereas the DM content of the June harvest was significantly (P < 0.05) lower (8.4±0.7%). In August, the lighter green biomass had a DM content of 12.5±2.0%, which increased with a darker biomass, reaching 19.2±2.2%.
Values of color components L* (black-white), a* (green-red), and b* (blue-yellow) for Ulva according to month of harvest (a, c and e) and color in August (b, d and f). The color of the biomasses in May, June and August are listed below the line. Data are represented as means ± SD. Different letters indicate significance of difference (P < 0.05) using one-way ANOVA followed with a Duncan’s Multiple Range test, n = 3
Dry matter (DM) content of Ulva according to month of harvest (a) and color in August (b). The color of the biomasses in May, June and August are listed below the line. Data are represented as means ± SD. Different letters indicate significance of difference (P < 0.05) using one-way ANOVA followed with a Duncan’s Multiple Range test, n = 3
Biochemical composition
The overall biochemical composition of the different Ulva biomasses is illustrated in Fig. 4 according to month (a) and color in August (b). The CP content was significantly (P < 0.05) higher in May (dark green/black color) compared to June and August, reaching 20.6±0.5% of DM. The June harvest (light green) had a CP content of 6.5±1.5% of DM, which increased to 10.7±0.3% of DM in August (mixed color). A higher CP content was related with a darker biomass upon color separation in August (Fig. 4b), ranging from 7.7±0.1% for the light green biomass to 16.4±0.4% for the dark green/black biomass. The lipid content ranged from 1.0-1.7±0.1-0.2% of DM for the different harvest times, reaching the highest content in June and August and lowest value in May. In August, a significantly (P < 0.05) higher lipid content (2.2±0.2% of DM) was observed to be related with a lighter color.
Biochemical composition of Ulva according to month of harvest (a, c, and e) and color in August (b, d, and f). The color of the biomasses in May, June and August are listed below the line. The content of ash (a and b), crude protein (CP) (c and d), and lipid (e and f) are presented as percentage of dry matter (DM). Data are represented as means ± SD. Different letters within the same legend indicate significance of difference (P < 0.05) using one-way ANOVA followed with a Duncan’s Multiple Range test, n = 3
The ash content was highest in the May harvest, reaching 41.2±3.9% of DM, followed by 36.9±0.2% of DM in June and 35.5±2.3% of DM in August. However, with no significant (P > 0.05) differences. In August, the darker biomass reached a significantly (P < 0.05) higher ash content (35.9±3.0% of DM) compared to the lighter biomasses.
Amino acid content and composition
To further investigate if the color of the biomass is related to the nutritional value, the composition of AA was investigated (Table 1). For the total AA, the same tendency as for the content of CP was observed. The dark green/black August biomass and the May biomass had the highest content of AA, reaching 16.2 and 19.8% of DM, respectively. The light green August biomass and the June biomass had the lowest AA contents of 7.6 and 8.2% of DM, respectively. These values are close to the CP content, which is also shown by the N-to-AA conversion factor, being close to 5 as used in this study. However, the light green August biomass and the June biomass were furthest from 5 (5.4 and 5.8, respectively), resulting in an underestimation for the CP content.
The light green August biomass and the June biomass resulted in the highest amount of EAA, reaching 38.2 and 38.7%, respectively. This fraction decreased with a darker green biomass, resulting in an amount of 35.1 and 36.8% for the May and the dark green/black August biomass, respectively. A higher amount of non-essential amino acids (NEAA) was observed for the darker green biomasses, which especially was due to a higher content of arginine. In general, glutamic and aspartic acid were found in highest concentrations for all biomasses.
Elemental composition
To specify the ash fraction, the Ulva biomasses according to harvest (May, June and August) and color in August (Light green, green and dark green/black) were analyzed for selected elements (Table 2). For the three harvest times, the May harvest had the highest content of several elements (Ca, Fe, Zn, Co, Mn, Cu, Ni, Se, As, Cd, Hg, and Pb) corresponding to the higher ash content found in this biomass. However, for Na, K, and Mg, the highest contents were observed in June (40.5±0.6, 36.8±2.5, and 23.5±1.0 mg g-1 DM, respectively). In August, higher contents of all elements except P and K were found in the dark green/black biomass, corresponding with the higher ash content observed. In contrast, the content of P and K was highest in the green fraction.
The highest concentrations of toxic elements were observed in May and in the darker green/black August biomass. Of the toxic elements, total arsenic (tAs) was found in highest concentrations, ranging from 4.7-16.4±0.2-1.0 µg g-1 DM. The European Food Safety Authority (EFSA) has determined tolerable intakes of different elements that can pose a risk of health effects to ensure food safety. Considering the content of As, a maximum intake of 2-146 g (depending on the fraction of iAs, 70-1%) DM per day for the dark biomasses from May and August would be allowed (Table 3). If iAs accounts for more than 2% of tAs, this would be the limiting factor for intake. Besides As, the content of Cd could be of concern. The highest contents are likewise found in the darker biomasses, allowing a maximum intake of 64-87 g DM per day.
Color vs. composition of biomass
A biplot was conducted to evaluate the different biomasses based on the different variables investigated (Fig. 5). The biomass from May and biomass with a dark green/black color from August were found to be related with a high content of DM, CP and ash. Most of the elements were also positively related with these biomasses, corresponding to the higher content of ash. However, high concentrations of K and Na were found to be more associated with the biomasses from June. A higher content of lipids as well as EAA was found to be related with the biomass from June and with the light green August biomass. For the NEAA, some were more related with the June harvest (tyrosine and proline), whereas the May harvest was associated with arginine and glutamic acid.
Biplot of the different biomasses (orange) divided in season (May, June and August) and color in August (light green, green and dark green/black) and 37 different variables investigated. Variables are colored according to the biochemical composition (purple) as dry matter (DM), crude protein (CP), lipid and ash content, essential amino acids (EAA, blue), nonessential amino acids (NEAA, red) and elements (green)
Discussion
Harvest
A wild population of Ulva was used, which probably consisted mostly of the species U. compressa, as this was the main species observed in Skive Fjord in September 2021, and also in previous years. Still, the species composition of Ulva in Skive Fjord fluctuates on a temporal and spatial scale (Unpublished results). Although unlikely, it would be possible that the composition of Ulva varied during the harvest times, which could affect the biochemical composition (Jansen et al. 2022). The different harvest times (May 18, June 21 and August 10, 2021) are referred to as their respective months. However, this is a generalization as abiotic factors in Skive Fjord fluctuate considerably on a temporal and spatial scale in the area. This is also one of the reasons for the area being a ‘green tide hotspot’. Thus, the variations in composition can be attributed to abiotic factors changing upon season as temperature, irradiance, rain and consequently nutrient run-off from land and salinity, wind inducing mixing of the shallow water, and consequently nutrient input from the sediment etc. Abiotic factors, in particular availability of nutrients and irradiance have been seen to affect the composition of Ulva (Toth et al. 2020; De Melo et al. 2021).
DM and lipids
The DM content of Ulva in this study is in accordance with literature, ranging from 8-18% (Paiva et al. 2017; Postma et al. 2018; Juul et al. 2022a). The June harvest (light green) showed the lowest amount of DM (Fig. 1) and the light green biomass in August also had a tendency to contain the lowest DM content. Bruhn et al. (2020) related a low DM content with a low biomass density at the on-set of a fast growth response, eventually leading to a high biomass density. This suggests that the light green biomasses could be correlated with the onset of a green tide. The higher content of lipids in the light green biomasses from June and August could be due to limited availability of nitrogen in the environment, resulting in a turnover of photosynthetic products to lipids instead of proteins as observed in microalgae (Rodolfi et al. 2009; Brennan and Owende 2010). This would also correspond with the low amount of CP in these biomasses as explained in “Crude protein and amino acids”. However, nitrogen limitation is also found to enhance the lipid content in U. rigida (Gordillo et al. 2001). Moreover, the lipid content in U. rigida and U. fenestrata is observed to vary due to different factors such as temperature, nutrient supply, and CO2 assimilation (Gao et al. 2017, 2018; Toth et al. 2020).
Crude protein and amino acids
The higher nitrogen content and thereof higher CP and TAA in the biomass in May might be due to a higher amount of available dissolved inorganic nitrogen in the surroundings as other studies have observed this relation for Ulva (Nielsen et al. 2012; Sode et al. 2013; Stedt et al. 2022b). Hence, the June biomass with lower CP and TAA content could possibly have had limited amount of nitrogen available, which is in agreement with a dry and calm June in 2021 (Hansen and Høgslund 2023). Lower availability of nutrients is found to be related with warmer and sunnier weather as a consequence of nutrients being taken up by phytoplankton blooms. Moreover, reduced precipitation and wash out of nutrients from land, and less wind can contribute to lower availability of nutrients (Hansen and Høgslund 2023). The increased CP content in August is most likely due to nutrient input to the system, either as a consequence of run-off from land or wind-induced mixing, introducing remineralised nutrients from the sediment to the water column. As 2021 was a relatively dry summer and August was relatively windy in comparison to June and July, the latter would probably be the main contributor (Hansen and Høgslund 2023). Bruhn et al. (2020) also observed a higher amount of nitrogen in Ulva harvested at the end of the season (October and November 2019). The total amount of biomass and area density was decreasing at this time point but also had a higher availability of inorganic nitrogen due to increased precipitation in autumn followed by increased washing out of nutrients from land. The first was also observed in the study of Pedersen and Borum (1996), where the main reason could be attributed to a higher availability of inorganic nitrogen. The nitrogen content in U. fenestrata has additionally been found to be affected by temperature and irradiance (Toth et al. 2020).
Robertson-Andersson et al. (2010) and Stedt et al. (2022a) observed that a darker green color of U. lactuca and U. fenestrata, respectively was correlated with a higher nitrogen content, which was also observed in this study. In terms of the color components, this was observed as an a* value around 0, resembling a darker green/black color. A higher content of chlorophyll might be due to a higher availability of nitrogen, facilitating the creation of chlorophyll as nitrogen is a major component of this molecule (Tumbo et al. 2002; Robertson-Andersson et al. 2010). Moreover, a higher availability of nitrogen leads to a higher content of protein and hence the protein Ribulose-1,5-bisphosphate carboxylase/oxygenase (RuBisCO). RuBisCO is a key enzyme in plants, which initiate the Carbon-Benson cycle, generating ATP and NADPH that can facilitate the synthesis of chlorophyll (von Wettstein et al. 1995; Buapet et al. 2008; Vicente et al. 2011). This could explain the relation between a higher CP and a darker green color observed both upon harvest time and color separation in August.
The N-to-AA conversion factor was higher for June (light green) and the light green fraction from August. This indicate that more nitrogen in the light green biomasses were related to protein compared to the darker biomass, which could be due to structural differences. In the darker biomass, excess nitrogen could have been present in the surroundings based on the higher protein content (Nielsen et al. 2012; Sode et al. 2013; Stedt et al. 2022b). Hence, inorganic nitrogen could be stored in vacuoles in the cells, which yet not have been converted to protein, resulting in an increased nitrogen content.
A higher relative amount of EAA was observed in the lighter green color of the biomass, indicating a higher nutritional value of the protein in these biomasses. This corresponds with the higher content of NEAA in the darker biomasses, which especially was due to a higher content of arginine. Arginine is the AA with the highest nitrogen to carbon ratio, which makes it great for storing nitrogen (Winter et al. 2015), which could be related with a possibly excess of nitrogen in the surroundings. The overall higher contents of glutamic and aspartic acid have been observed in previous studies (Fleurence et al. 1995; Unis et al. 2023), whereas other studies observed highest content of alanine or arginine (Pallaoro et al. 2016; Shpigel et al. 2018). Unis et al. (2023) investigated the EAA content upon season and maturity stage but did not see any correlations.
Ash and elemental composition
The ash content revealed significant (P < 0.05) differences upon color in August, where a higher ash content was related with a darker color. However, no significant (P > 0.05) differences were observed upon harvest time, suggesting that the ash content might be more difficult to relate with the color. Toth et al. (2020) observed that the ash content in U. fenestrata decreased with higher environmental concentrations of nitrate. However, higher environmental concentrations of nitrate were also found to result in a higher content of CP (Toth et al. 2020). The dark green/black biomass from August in this study was both higher in CP and ash, counteracting this statement. In another study, Villaresa et al. (2002) observed that the ash content in U. rigida increased upon slow growth periods, whereas maximum growth periods were correlated with a decreased content. This would suggest that the dark green/black biomass from August with higher ash content would be related with inactive growth, and the biomasses with lower ash content (light green and green colored biomass from August) would be related with a higher growth. The lower ash content upon maximum growth is caused by dilution due to synthesis of carbohydrates and proteins during high growth rate periods (Villaresa et al. 2002). Bruhn et al. (2011) studied the growth of U. fenestrata, where increased irradiance was found to increase the biomass production as well as the C:N ratios, showing that a high growth rate is related with a lower content of protein. Hence, the light green biomass from August with both low CP and ash content might indicate a high growth period. For the light green biomass from June with low CP and higher ash content, this could still be due to a growth period but with higher rates of photosynthesis and respiration, facilitating the assimilation of metals (Catsiki and Papathanassiou 1993). This would also correlate with the lower DM content, suggesting an onset of a green tide as explained in “DM and lipids”. However, the ash content of Ulva is highly dependent on the environment, as it takes up nutrients present and hence the variations in content and composition could be explained by variations in the fjord during the different harvest times (Mantri et al. 2020; Hansen and Høgslund 2023). Other factors that could contribute to the ash content could be presence of sediments depending on the efficiency of the washing, rainy periods diluting the levels of elements, location of the harvest (inner areas for the harvest by hand vs further out for the harvest by boat), human activity influencing the levels of elements available, age of tissue and abiotic factors such as temperature and salinity (Malea et al. 1995; Haritonidis and Malea 1999; Villaresa et al. 2002).
The higher concentrations of K, Mg and Na observed in the biomass from June could be as a result of higher concentrations of these elements in the surroundings. Resulting either in a higher uptake or simply by being present on the thallus as these elements have been found to be greatly affected upon different washing times and temperatures (Magnusson et al. 2016).
Of the toxic elements, As was present in highest concentrations in the darker biomasses from May and August, making it the limiting factor for intake, if the iAs fraction was above 2% of tAs. The concentrations of tAs found in these biomasses are, however, in accordance with literature ranging from 1.1-15.4 µg g-1 DM in U. fenestrata and U. lactuca (Nielsen et al. 2012; Roleda et al. 2021). To determine the toxicity, the inorganic fraction (iAs) should be analyzed. This fraction is found to vary highly from 1-70% in seaweeds (Holdt and Kraan 2011). Almela et al. (2002) observed that iAs accounted for approximately 40% of tAs in U. lactuca. Moreover, the bioavailability of the elements would be interesting to investigate to determine if these values are of concern or not (National Food Institute, Technical University of Denmark et al. 2019).
Color vs. biomass quality and usage
Altogether, these results imply (visualized in the biplot, Fig. 5) that a darker colored biomass is related with a higher content of DM, CP and most of the elements investigated. On the other hand, a lighter colored biomass is related with a higher content of lipids and nutritional value in terms of relative amount of EAA. A more comprehensive study would be interesting to conduct to see if the same relations can be drawn with further sample outtakes at different time points, investigating different species and different locations of harvests as this could influence the biochemical composition.
For the application of processing/biorefining, aiming for food, a high content of protein would be preferable, suggesting that a darker biomass should be chosen in accordance with the results obtained in this study. High content of protein is important if an industry wants to extract the protein as an ingredient for human nutrition. However, a darker biomass in the color separation resulted in a higher content of ash, leading to a higher content of toxic elements that potentially could exceed the limit values allowed in food. Moreover, a high ash content is always problematic as it may affect the protein extraction yield and make it difficult to achieve a high protein digestibility (Juul et al. 2022b). For the use of Ulva for nutritional purposes, it is necessary to extract the proteins to enhance the protein digestibility (Juul et al. 2022b). Bikker et al. (2016) showed that upon extraction of protein, the toxic elements As, Cd, Pb and Hg were increased as well. Therefore, additional treatment might be necessary to use the darker biomass for food and investigation of the bioavailability of these elements would be needed. Instead, the biomass of green color might be the better option for food, as there is a medium amount of protein with a good amount of EAA and a lower ash content especially for the toxic element, As. Considering the lipid content, this is generally very low in Ulva, making it an extra added value in the protein extracts, rather than a potential to be extracted as a nutrient itself (Toth et al. 2020).
The color coding can be an advantageous tool to get a fast and cost-efficiently indication on the biochemical composition and hence help to harvest an optimal biomass for the intended purpose. Stedt et al. (2022a) showed that color image analysis could be used to create a model to estimate the nitrogen content in U. fenestrata, resulting in a color guide for Ulva producers. In the same manner, this could perhaps be developed for DM and lipid contents as well as nutritional values as AA and the elemental composition to get a better indication on the quality of the biomass. This could contribute to securing a more uniform biomass supply as well as to allocate different colors of biomass for different purposes. The light green and dark green/black biomass could for instance be used in other sectors, such as feed or biogas if either protein contents are too low or toxic elements are too high for the biomass to be used for food. Moreover, it would be interesting to investigate if the extractability of the proteins might change with the different colors related to a change of structure in the biomass.
Conclusion
This study investigated the biochemical composition of wild Ulva in relation to season (May, June, and August) and color (light green, green, and dark green/black), showing a tendency that color can be used as an indicator for the biochemical composition. A darker green biomass of Ulva was found to relate with a higher content of DM and CP, whereas a lighter green colored biomass indicated a higher content of lipids. The Ulva harvested in June was predominantly light in color, resembling the composition of the light green fraction from August. The Ulva harvested in May was predominantly dark in color, which also revealed a high DM and CP content. The darker green biomass also revealed a higher content of elements, where high concentrations of the toxic element As could be of concern. On the other hand, the lighter green biomass was related to a higher N-to-AA conversion factor as well as a higher fraction of EAA, reflecting a higher nutritional value of this fraction.
Availability of data and material
The authors declare that the data supporting the findings of this study are available within the paper. Should any raw data files be needed they are available from the corresponding author upon reasonable request.
References
Almela C, Algora S, Benito V, Clemente MJ, Devesa V, Súñer MA, Vélez D, Montoro R (2002) Heavy metal, total arsenic, and inorganic arsenic contents of algae food products. J Agric Food Chem 50:918–923
Angell AR, Mata L, de Nys R, Paul NA (2016) The protein content of seaweeds: a universal nitrogen-to-protein conversion factor of five. J Appl Phycol 28:511–524
Bikker P, van Krimpen MM, van Wikselaar P, Houweling-Tan B, Scaccia N, van Hal JW, Huijgen WJJ, Cone JW, Lopez-Contreras AM (2016) Biorefinery of the green seaweed Ulva lactuca to produce animal feed, chemicals and biofuels. J Appl Phycol 28:3511–3525
Bikker P, Stokvis L, van Krimpen MM, van Wikselaar PG, Cone JW (2020) Evaluation of seaweeds from marine waters in Northwestern Europe for application in animal nutrition. Anim Feed Sci Technol 263:114460–114474
Bleakley S, Hayes M (2017) Algal proteins: Extraction, application, and challenges concerning production. Foods 6:33–64
Brennan L, Owende P (2010) Biofuels from microalgae - a review of technologies for production, processing, and extractions of biofuels and co-products. Renew Sust Energ Rev 14:557–577
Bruhn A, Dahl J, Nielsen HB, Nikolaisen L, Rasmussen MB, Markager S, Olesen B, Arias C, Jensen PD (2011) Bioenergy potential of Ulva lactuca: biomass yield, methane production and combustion. Bioresour Technol 102:2595–604
Bruhn A, Rasmussen MB, Pedersen HB, Thomsen M (2020) Høst af eutrofieringsbetingede masseforekomster af søsalat – status på viden om miljøeffekter og økonomi. Aarhus Universitet, DCE – Nationalt Center for Miljø og Energi, Notat nr. 2020|20, pp 1–17. https://dce.au.dk/fileadmin/dce.au.dk/Udgivelser/Notatet_2020/N2020_20.pdf
Buapet P, Hiranpan R, Ritchie RJ, Prathep A (2008) Effect of nutrient inputs on growth, chlorophyll, and tissue nutrient concentration of Ulva reticulata from a tropical habitat. Sci Asia 34:245–252
Catsiki VA, Papathanassiou E (1993) The use of chlorophyte Ulva lactuca (L.) as indicator organism of metal pollution (Cost-48) Symposium of Sub Group III In: Rijstenbil, JW, Haritonidis S (eds) Macroalgae, eutrophication and trace metal cycling in estuaries and lagoons. Commission of the European Communities, Thessaloniki, pp 93–105
Cherry P, O’Hara C, Magee PJ, McSorley EM, Allsopp PJ (2019) Risks and benefits of consuming edible seaweeds. Nutr Rev 77:307–329
De Melo NSM, Cardoso LG, De Castro Nunes JM, Brito GB, Caires TA, De Souza CO, Portz L, Druzian JI (2021) Effects of dry and rainy seasons on the chemical composition of Ulva fasciata, Crassiphycus corneus, and Sargassum vulgare seaweeds in tropical environment. Rev Bras Bot 44:331–344
EFSA (2009a) Cadmium in food - Scientific opinion of the Panel on Contaminants in the Food Chain. EFSA J 980:1–139
EFSA (2009b) Scientific opinion on arsenic in food. EFSA J 7:1351–1550
EFSA (2010) Scientific opinion on lead in food. EFSA J 8:1570–1721
EFSA (2012) Scientific opinion on the risk for public health related to the presence of mercury and methylmercury in food. EFSA J 10:2985–3226
Fleurence J (1999) Seaweed proteins: biochemical, nutritional aspects and potential uses. Trends Food Sci Technol 10:25–28
Fleurence J, LeCoeur C, Mabeau S, Maurice M, Landrein A (1995) Comparison of different extractive procedures for proteins from the edible seaweeds Ulva rigida and Ulva rotundata. J Appl Phycol 7:577–582
Gao G, Clare AS, Rose C, Caldwell GS (2017) Eutrophication and warming-driven green tides (Ulva rigida) are predicted to increase under future climate change scenarios. Mar Pollut Bull 114:439–447
Gao G, Clare AS, Chatzidimitriou E, Rose C, Caldwell G (2018) Effects of ocean warming and acidification, combined with nutrient enrichment, on chemical composition and functional properties of Ulva rigida. Food Chem 258:71–78
Gordillo F, Jiménez C, Goutx M, Niell X (2001) Effects of CO2 and nitrogen supply on the biochemical composition of Ulva rigida with especial emphasis on lipid class analysis. J Plant Physiol 158:367–373
Guiry MD, Guiry GM (2020) AlgaeBase World-wide electronic publication. National University of Ireland, Galway http://www.algaebase.org accessed 30 January 2024
Hafting JT, Critchley AT, Cornish ML, Hubley SA, Archibald AF (2011) On-land cultivation of functional seaweed products for human usage. J Appl Phycol 24:385–392
Hansen JW, Høgslund S (eds) (2023) Marine områder 2021. NOVANA. Aarhus Universitet, DCE – Nationalt Center for Miljø og Energi, Videnskabelig rapport fra DCE nr. 529, pp 1–220. http://dce2.au.dk/pub/SR529.pdf
Haritonidis S, Malea P (1999) Bioaccumulation of metals by the green alga Ulva rigida from Thermaikos Gulf, Greece. Environ Pollut 104:365–372
Holdt SL, Kraan S (2011) Bioactive compounds in seaweed: functional food applications and legislation. J Appl Phycol 23:543–597
Hughey JR, Maggs CA, Mineur F, Jarvis C, Miller KA, Shabaka SH, Gabrielson PW (2019) Genetic analysis of the Linnaean Ulva lactuca (Ulvales, Chlorophyta) holotype and related type specimens reveals name misapplications, unexpected origins, and new synonymies. J Phycol 55:503–508
Jansen HM, Bernard MS, Nederlof MAJ, Van Der Meer IM, Van Der Werf A (2022) Seasonal variation in productivity, chemical composition and nutrient uptake of Ulva spp. (Chlorophyta) strains. J Appl Phycol 34:1649–1660
Juul L (2022) Extraction and evaluation of protein from Ulva sp. PhD thesis, Aarhus University, Denmark, p 91
Juul L, Steinhagen S, Bruhn A, Jensen SK, Undeland I, Dalsgaard TK (2022a) Combining pressing and alkaline extraction to increase protein yield from Ulva fenestrata biomass. Food Bioprod Process 134:80–85
Juul L, Stødkilde L, Ingerslev AK, Bruhn A, Jensen SK, Dalsgaard TK (2022b) Digestibility of seaweed protein from Ulva sp. and Saccharina latissima in rats. Algal Res 63:102644–102651
Magnusson M, Carl C, Mata L, de Nys R, Paul NA (2016) Seaweed salt from Ulva: A novel first step in a cascading biorefinery model. Algal Res 16:308–316
Magnusson M, Glasson CRK, Vucko MJ, Angell A, Neoh TL, de Nys R (2019) Enrichment processes for the production of high-protein feed from the green seaweed Ulva ohnoi. Algal Res 41:101555–101564
Malea P, Haritonidis S, Kevrekidis T (1995) Metal content of some green and brown seaweeds from Antikyra Gulf (Greece). Hydrobiologia 310:19–31
Mantri VA, Kazi MA, Balar NB, Gupta V, Gajaria T (2020) Concise review of green algal genus Ulva Linnaeus. J Appl Phycol 32:2725–2741
Martinez B, Rico J (2002) Seasonal variation of P content and major N pools in Palmaria palmata (Rhodophyta). J Phycol 38:1082–1089
National Food Institute, Technical University of Denmark, Sá Monteiro M, Sloth J, Holdt S, Hansen M (2019) Analysis and risk assessment of seaweed. EFSA J 17:e170915
Neveux N, Bolton JJ, Bruhn A, Roberts DA, Ras M (2018) The bioremediation potential of seaweeds: recycling nitrogen, phosphorus, and other waste products. In: Barre SL, Bates SS (eds) Blue Biotechnology: Production and Use of Marine Molecules. Wiley-VCH, Weinheim, pp 217–239
Nielsen MM, Bruhn A, Rasmussen MB, Olesen B, Larsen MM, Møller HB (2012) Cultivation of Ulva lactuca with manure for simultaneous bioremediation and biomass production. J Appl Phycol 24:449–458
Paiva L, Lima E, Neto AI, Marcone M, Baptista J (2017) Nutritional and functional bioactivity value of selected Azorean macroalgae: Ulva compressa, Ulva rigida, Gelidium microdon, and Pterocladiella capillacea. J Food Sci 82:1757–1764
Pallaoro MF, do Nascimento Vieira F, Hayashi L (2016) Ulva lactuca (Chlorophyta Ulvales) as co-feed for Pacific white shrimp. J Appl Phycol 28:3659–3665
Pedersen MF, Borum J (1996) Nutrient control of algal growth in estuarine waters. Nutrient limitation and the importance of nitrogen requirements and nitrogen storage among phytoplankton and species of macroalgae. Mar Ecol Prog Ser 142:261–272
Postma PR, Cerezo-Chinarro O, Akkerman RJ, Olivieri G, Wijffels RH, Brandenburg WA, Eppink MHM (2018) Biorefinery of the macroalgae Ulva lactuca: Extraction of proteins and carbohydrates by mild disintegration. J Appl Phycol 30:1281–1293
Queirós AS, Circuncisão AR, Pereira E, Válega M, Abreu MH, Silva AMS, Cardoso SM (2021) Valuable nutrients from Ulva rigida: Modulation by seasonal and cultivation factors. Appl Sci 11:6137–6152
R Core Team (2021) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna URL https://www.R-project.org/
Robertson-Andersson DV, Wilson DT, Bolton JJ, Anderson RJ, Maneveldt GW (2010) Rapid assessment of tissue nitrogen in cultivated Gracilaria gracilis (Rhodophyta) and Ulva lactuca (Chlorophyta). Afr J Aquat Sci 34:169–172
Rodolfi L, Chini Zittelli G, Bassi N, Padovani G, Biondi N, Bonini G, Tredici MR (2009) Microalgae for oil: Strain selection, induction of lipid synthesis and outdoor mass cultivation in a low-cost photobioreactor. Biotechnol Bioeng 102:100–112
Roleda MY, Lage S, Aluwini DF, Rebours C, Brurberg MB, Nitschke U, Gentili FG (2021) Chemical profiling of the Arctic sea lettuce Ulva lactuca (Chlorophyta) mass-cultivated on land under controlled conditions for food applications. Food Chem 341:127999–128011
Rouxel C, Bonnabeze E, Daniel A, Jérôme M, Etienne M, Fleurence J (2001) Identification by SDS PAGE of green seaweeds (Ulva and Enteromorpha) used in the food industry. J Appl Phycol 13:215–218
Shpigel M, Shauli L, Odintsov V, Ben-Ezra D, Neori A, Guttman L (2018) The sea urchin, Paracentrotus lividus, in an Integrated Multi-Trophic Aquaculture (IMTA) system with fish (Sparus aurata) and seaweed (Ulva lactuca): Nitrogen partitioning and proportional configurations. Aquaculture 490:260–269
Sode S, Bruhn A, Balsby TJS, Larsen MM, Gotfredsen A, Rasmussen MB (2013) Bioremediation of reject water from anaerobically digested waste water sludge with macroalgae (Ulva lactuca, Chlorophyta). Bioresour Technol 146:426–435
Stedt K, Toth GB, Davegård J, Pavia H, Steinhagen S (2022a) Determination of nitrogen content in Ulva fenestrata by color image analysis – a rapid and cost-efficient method to estimate nitrogen content in seaweeds. Front Mar Sci 9:1081870–1081879
Stedt K, Trigo JP, Steinhagen S, Nylund GM, Forghani B, Pavia H, Undeland I (2022b) Cultivation of seaweeds in food production process waters: Evaluation of growth and crude protein content. Algal Res 63:102647–102656
Toth GB, Harrysson H, Wahlström N, Olsson J, Oerbekke A, Steinhagen S, Kinnby A, White J, Albers E, Edlund U, Undeland I, Pavia H (2020) Effects of irradiance, temperature, nutrients, and pCO2 on the growth and biochemical composition of cultivated Ulva fenestrata. J Appl Phycol 32:3243–3254
Tumbo S, Wagner D, Heinemann P (2002) On-the-go sensing of chlorophyll status in corn. Trans ASABE 45:1207–1215
Unis R, Chemodanov A, Gnayem N, Gnaim R, Israel Á, Palatnik RR, Zilberman D, Gnaim J, Golberg A (2023) Effect of seasonality on the amino acid and monosaccharide profile from the green seaweed Ulva lactuca cultivated in plastic sleeves onshore (Mikhmoret, Israel). J Appl Phycol 35:1347–1363
Van Den Burg SWK, Van Duijn AP, Bartelings H, Van Krimpen MM, Poelman M (2016) The economic feasibility of seaweed production in the North Sea. Aqua Econ Manag 20:235–252
Vicente R, Morcuende R, Babiano J (2011) Differences in Rubisco and chlorophyll content among tissues and growth stages in two tomato (Lycopersicon esculentum Mill.) varieties. Agron Res 9:501–507
Villaresa R, Puentea X, Carballeirab A (2002) Seasonal variation and background levels of heavy metals in two green seaweeds. Environ Pollut 119:79–90
von Wettstein D, Gough S, Kannangara CG (1995) Chlorophyll biosynthesis. The Plant Cell 7:1039–1057
Walpole SC, Prieto-Merino D, Edwards P, Cleland J, Stevens G, Roberts I (2012) The weight of nations: an estimation of adult human biomass. BMC Public Health 12:439–445
Winter G, Todd CD, Trovato M, Forlani G, Funck D (2015) Physiological implications of arginine metabolism in plants. Front Plant Sci 6:534–548
Yaich H, Garna H, Besbes S, Paquot M, Blecker C, Attia H (2011) Chemical composition and functional properties of Ulva lactuca seaweed collected in Tunisia. Food Chem 128:895–901
Acknowledgement
Laboratory technicians Anna Marie Plejdrup and Sandra Drewes Fabricius at Department of Ecoscience, Aarhus University are acknowledged for performing the element analyzes. Thanks to HedeDanmark for harvesting of Ulva and to Skive Municipality for supporting the project with guidance and permission for test harvest.
Funding
Open access funding provided by Aarhus Universitet. Data in this work was generated through accessing research infrastructure at Aarhus University, including those funded by FOODHAY (Food and Health Open Innovation Laboratory, Danish Roadmap for Research Infrastructure). The study was funded by the Centre for Circular Bioeconomy at Aarhus University and GUDP with the project SeaSusProtein j. no 34009-19-1615.
Author information
Authors and Affiliations
Contributions
Signe H. Nissen: Conceptualization, Investigation, Formal analysis, Visualization, Writing – Original Draft.
Louise Juul: Conceptualization, Investigation, Writing – Review & Editing.
Annette Bruhn: Conceptualization, Supervision, Writing – Review & Editing.
Jens Søndergaard: Writing – Review & Editing.
Trine K. Dalsgaard: Conceptualization, Resources, Supervision, Writing – Review & Editing.
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Nissen, S.H., Juul, L., Bruhn, A. et al. The biochemical composition and its relation to color of Ulva spp. upon harvest time. J Appl Phycol 36, 2095–2107 (2024). https://doi.org/10.1007/s10811-024-03207-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10811-024-03207-z