Abstract
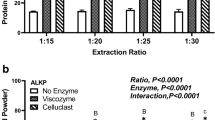
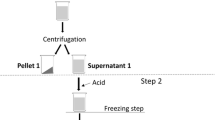
We compared protocols to isolate and concentrate protein from the green seaweed Ulva ohnoi. We quantified the effect of three factors on protein and essential amino acid yields and concentrations in protein isolates and residuals in a factorial experimental design. The three factors were starting material (as dry and milled or fresh and pulped), aqueous solvent-to-biomass ratio (20:1 or 5:1 v/w) and the incubation time in the aqueous solvent (incubated for 16 h at 30 °C or incubated for <1 min at ambient temperature). The protein isolation protocols increased the concentration of protein, total essential amino acids, methionine and lysine ~3 to 5-fold compared to whole U. ohnoi and were considerably more effective than the different protein concentrating combinations, which only increased protein and amino acid concentrations by 30–40 % in the residual biomass. The use of fresh and pulped biomass as the starting material, an incubation time of <1 min at ambient temperature and a low aqueous solution volume resulted in the highest protein isolate yield of 22 % of the protein found in seaweed. This study demonstrated that proteins from U. ohnoi were most effectively isolated by adopting protocols for terrestrial leaves compared to the protocols employed for seed crops as traditionally applied to seaweeds.




Similar content being viewed by others
References
Agboola S, Ng D, Mills D (2005) Characterisation and functional properties of Australian rice protein isolates. J Cereal Sci 41:283–290
Alves A, Sousa RA, Reis RL (2013) A practical perspective on ulvan extracted from green algae. J Appl Phycol 25:407–424
Anderson M, Gorley R (2007) PERMANOVA+ fo r PRIMER: guide to software and statistic al methods. PRIMER-E, Plymouth, England
Angell AR, Pirozzi I, de Nys R, Paul NA (2012) Feeding preferences and the nutritional value of tropical algae for the abalone Haliotis asinina. PLoS One 7(6):e38857
Angell AR, Mata L, de Nys R, Paul NA (2014) Variation in amino acid content and its relationship to nitrogen content and growth rate in Ulva ohnoi (Chlorophyta). J Phycol 50:216–226
Angell AR, Angell SF, de Nys R, Paul NA (2016) Seaweed as a protein source for mono-gastric livestock. Trends Food Sci Technol In press
Bals B, Dale BE (2011) Economic comparison of multiple techniques for recovering leaf protein in biomass processing. Biotechnol Bioeng 108:530–537
Barba FJ, Grimi N, Vorobiev E (2015) Evaluating the potential of cell disruption technologies for green selective extraction of antioxidant compounds from Stevia rebaudiana Bertoni leaves. J Food Eng 149:222–228
Barros FCN, da Silva DC, Sombra VG, Maciel JS, Feitosa JPA, Freitas ALP, de Paula RCM (2013) Structural characterization of polysaccharide obtained from red seaweed Gracilaria caudata (J Agardh). Carbohydr Polym 92:598–603
Berk Z (1992) Technology of production of edible flours and protein products from soybean. FAO Agricultural Services Bulletin 97, FAO, Rome
Bikker P, Krimpen MM, Wikselaar P, Houweling-Tan B, Scaccia N, Hal JW, Huijgen WJ, Cone JW, López-Contreras AM (2016) Biorefinery of the green seaweed Ulva lactuca to produce animal feed, chemicals and biofuels. J Appl Phycol. doi:10.1007/s10811-016-0842-3
BobinDubigeon C, Lahaye M, Guillon F, Barry JL, Gallant DJ (1997) Factors limiting the biodegradation of Ulva sp cell-wall polysaccharides. J Sci Food Agric 75:341–351
Bolton J, Robertson-Andersson D, Shuuluka D, Kandjengo L (2009) Growing Ulva (Chlorophyta) in integrated systems as a commercial crop for abalone feed in South Africa: a SWOT analysis. J Appl Phycol 21:575–583
Bosch L, Alegría A, Farré R (2006) Application of the 6-aminoquinolyl-N-hydroxysccinimidyl carbamate (AQC) reagent to the RP-HPLC determination of amino acids in infant foods. J Chromatogr B 831:176–183
Chiesa S, Gnansounou E (2011) Protein extraction from biomass in a bioethanol refinery–possible dietary applications: use as animal feed and potential extension to human consumption. Bioresour Technol 102:427–436
Cohen SA (2000) Amino acid analysis using precolumn derivatization with 6-aminoquinolyl-n-hydroxysuccinimidyl carbamate. In: Cooper C, Packer N, Williams K (eds) Amino Acid Analysis Protocols, vol 159. Methods in Molecular Biology. Humana Press, pp 39–47
Dierick N, Ovyn A, De Smet S (2009) Effect of feeding intact brown seaweed Ascophyllum nodosum on some digestive parameters and on iodine content in edible tissues in pigs. J Sci Food Agric 89:584–594
Ellis RJ (1979) The most abundant protein in the world. Trends Biochem Sci 4:241–244
Evans F, Critchley A (2014) Seaweeds for animal production use. J Appl Phycol 26:891–899
Fernández S, Padilla A, Mucciarelli S (1999) Protein extraction from Atriplex lampa leaves: potential use as forage for animals used for human diets. Plant Food Hum Nutr 54:251–259
Fleurence J, LeCoeur C, Mabeau S, Maurice M, Landrein A (1995) Comparison of different extractive procedures for proteins from the edible seaweeds Ulva rigida and Ulva rotundata. J Appl Phycol 7:577–582
Harnedy PA, FitzGerald RJ (2011) Bioactive proteins, peptides, and amino acids from macroalgae. J Phycol 47:218–232
Harnedy PA, FitzGerald RJ (2013) Extraction of protein from the macroalga Palmaria palmata. LWT-Food Sci Technol 51:375–382
Jazrawi C, Biller P, He Y, Montoya A, Ross AB, Maschmeyer T, Haynes BS (2015) Two-stage hydrothermal liquefaction of a high-protein microalga. Algal Res 8:15–22
Jordan P, Vilter H (1991) Extraction of proteins from material rich in anionic mucilages: partition and fractionation of vanadate-dependent bromoperoxidases from the brown algae Laminaria digitata and L. saccharina in aqueous polymer two-phase systems. Biochim Biophys Acta-Gen Subj 1073:98–106
Ju Z, Hettiarachchy N, Rath N (2001) Extraction, denaturation and hydrophobic properties of rice flour proteins. J Food Sci 66:229–232
Kadam SU, Tiwari BK, O’Donnell CP (2013) Application of novel extraction technologies for bioactives from marine algae. J Agric Food Chem 61:4667–4675
Kandasamy G, Karuppiah SK, Rao PVS (2012) Salt- and pH-induced functional changes in protein concentrate of edible green seaweed Enteromorpha species. Fisheries Sci 78:169–176
Katayama M, Fukuda T, Okamura T, Suzuki E, Tamura K, Shimizu Y, Suda Y, Suzuki K (2011) Effect of dietary addition of seaweed and licorice on the immune performance of pigs. Anim Sci J 82:274–281
Kolender AA, Matulewicz MC (2002) Sulfated polysaccharides from the red seaweed Georgiella confluens. Carbohydr Res 337:57–68
Kraan S (2012) Algal polysaccharides, novel applications and outlook. In: Chang C-F (ed) Carbohydrates—comprehensive studies on glycobiology and glycotechnology. InTech, Rijeka pp 489–532
Kumar KS, Ganesan K, Selvaraj K, Rao PVS (2014) Studies on the functional properties of protein concentrate of Kappaphycus alvarezii (Doty) Doty—an edible seaweed. Food Chem 153:353–360
Lawton RJ, Mata L, de Nys R, Paul NA (2013) Algal bioremediation of waste waters from land-based aquaculture using Ulva: selecting target species and strains. PLoS One 8(10):e77344
Le Guillard C, Bergé J-P, Donnay-Moreno C, Bruzac S, Ragon J-Y, Baron R, Fleurence J, Dumay J (2016) Soft liquefaction of the red seaweed Grateloupia turuturu Yamada by ultrasound-assisted enzymatic hydrolysis process. J Appl Phycol 28:2575–2585
Maciel JS, Chaves LS, Souza BWS, Teixeira DIA, Freitas ALP, Feitosa JPA, de Paula RCM (2008) Structural characterization of cold extracted fraction of soluble sulfated polysaccharide from red seaweed Gracilaria birdiae. Carbohydr Polym 71:559–565
Magnusson M, Carl C, Mata L, Rd N, Paul NA (2016) Seaweed salt from Ulva: a novel first step in a cascading biorefinery model. Algal Res 16:308–316
Masters DG, Benes SE, Norman HC (2007) Biosaline agriculture for forage and livestock production. Agric Ecosyst Environ 119:234–248
Mata L, Schuenhoff A, Santos R (2010) A direct comparison of the performance of the seaweed biofilters, Asparagopsis armata and Ulva rigida. J Appl Phycol 22:639–644
Mata L, Magnusson M, Paul NA, de Nys R (2016) The intensive land-based production of the green seaweeds Derbesia tenuissima and Ulva ohnoi: biomass and bioproducts. J Appl Phycol 28:365–375
McDermid KJ, Stuercke B (2003) Nutritional composition of edible Hawaiian seaweeds. J Appl Phycol 15:513–524
McDermid KJ, Stuercke B, Balazs GH (2007) Nutritional composition of marine plants in the diet of the green sea turtle (Chelonia mydas) in the Hawaiian islands. Bull Mar Sci 81:55–71
Melo MRS, Feitosa JPA, Freitas ALP, de Paula RCM (2002) Isolation and characterization of soluble sulfated polysaccharide from the red seaweed Gracilaria cornea. Carbohydr Polym 49:491–498
Neveux N, Yuen A, Jazrawi C, He Y, Magnusson M, Haynes B, Masters A, Montoya A, Paul N, Maschmeyer T (2014) Pre-and post-harvest treatment of macroalgae to improve the quality of feedstock for hydrothermal liquefaction. Algal Res 6:22–31
Nielsen MM, Bruhn A, Rasmussen MB, Olesen B, Larsen MM, Moller HB (2012) Cultivation of Ulva lactuca with manure for simultaneous bioremediation and biomass production. J Appl Phycol 24:449–458
Pirie N (1969) The production and use of leaf protein. Proc Nutr Soc 28:85–91
Ray B (2006) Polysaccharides from Enteromorpha compressa: isolation, purification and structural features. Carbohydr Polym 66:408–416
Šic Žlabur J, Voća S, Dobričević N, Pliestić S, Galić A, Boričević A, Borić N (2016) Ultrasound-assisted extraction of bioactive compounds from lemon balm and peppermint leaves. Int Agrophysics 30:95–104
Sinclair S (2009) Protein extraction from pasture. Literature review Part A: The plant fractionation bio-process and adaptability to farming systems. Milestone Report prepared for MAF SFF Grant C 8
Spreitzer RJ, Salvucci ME (2002) Rubisco: structure, regulatory interactions, and possibilities for a better enzyme. Plant Biol 53:449–475
Tan SH, Mailer RJ, Blanchard CL, Agboola SO (2011) Canola proteins for human consumption: extraction, profile, and functional properties. J Food Sci 76:R16–R28
Turhan K, Barbano D, Etzel M (2003) Fractionation of caseins by anion-exchange chromatography using food-grade buffers. J Food Sci 68:1578–1583
Wong KH, Cheung PCK (2001a) Influence of drying treatment on three Sargassum species 2. Protein extractability, in vitro protein digestibility and amino acid profile of protein concentrates. J Appl Phycol 13:51–58
Wong KH, Cheung PCK (2001b) Nutritional evaluation of some subtropical red and green seaweeds part II. In vitro protein digestibility and amino acid profiles of protein concentrates. Food Chem 72:11–17
Yamamoto M (1980) Physicochemical studies on sulfated polysaccharides extracted from seaweeds at various temperatures. Agric Biol Chem 44:589–593
Yu G, Zhang Y, Schideman L, Funk T, Wang Z (2011) Distributions of carbon and nitrogen in the products from hydrothermal liquefaction of low-lipid microalgae. Energy Environ Sci 4:4587–4595
Acknowledgments
This research is part of the MBD Energy Research and Development program for Biological Carbon Capture and Storage. The project is supported by the Advanced Manufacturing Cooperative Research Centre (AMCRC), funded through the Australian Government’s Cooperative Research Centre Scheme.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Angell, A.R., Paul, N.A. & de Nys, R. A comparison of protocols for isolating and concentrating protein from the green seaweed Ulva ohnoi . J Appl Phycol 29, 1011–1026 (2017). https://doi.org/10.1007/s10811-016-0972-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10811-016-0972-7