Abstract
Dietary algae have been reported to decrease HIV viral fusion/entry and replication and increase immune response, suggesting that regular consumption of algae by people in Japan, Korea, and Chad could be an important factor in their relatively low HIV/AIDS rates. Five antiretroviral-naïve people with HIV (three females, two males; five African Americans) living in Columbia SC participated in the phase I study of acute toxicity. Subjects were randomly assigned to 5 g day-1 brown seaweed (Undaria pinnatifida), Spirulina (Arthrospira platensis), or a combination of both. Endpoints included HIV viral load, complete blood count (CBC), metabolic and lipid panel, and quality of life questionnaire data. When no short-term toxicities were observed, six additional subjects (four females, two males; five African Americans, one Latina) were recruited to further evaluate short- and long-term toxicities (phase II). No adverse effects were observed for the 11 subjects in the phase I trial, and quality of life indicators improved at 3 weeks. No significant changes were observed in CBC, metabolic or lipid panel analyses. CD4 cells (milliliters) and HIV-1 viral load remained stable over the first 3-month phase II study period. One subject continued in the study for 13 months and had clinically significant improvement in CD4 (>100 cells mL−1) and decreased HIV viral load of 0.5 log10. Our pilot data suggest that Undaria, Spirulina, and a combination of both were nontoxic and over time may improve clinical endpoints of HIV/AIDS.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Dietary chemoprevention of HIV/AIDS is an attractive concept. In the best of worlds, everyone with AIDS would have access to pharmaceutical antiretroviral (ARV) medications, but in reality, only 5 million of 34 million people living with HIV/AIDS currently have access to ARV treatment (UNAIDS 2011). Additionally, ARV is not curative and is both expensive and associated with significant side effects including heart disease and dementia for long-time users (Payne et al. 2011), and drug resistance is an increasingly common problem.
Dietary algae may be an important factor in explaining geographic differences. Seaweeds are consumed daily by most people in Japan and Korea, but only infrequently in other countries of the world. Rates of HIV/AIDS in Japan and Korea have remained stable and extremely low over the last 30 years (<0.1%) compared to rates in the USA (0.6%) (UNAIDS 2011). Although seaweed is rarely consumed in Africa, some tribal groups in Chad have traditionally consumed the blue-green alga Arthrospira platensis (Spirulina) as part of their daily cuisine (Abdulqader et al. 2000). HIV/AIDS rates in Chad are high (5.1%) compared to Asia, but have remained lower than in several nearby countries, despite poverty and decades of civil unrest (UNAIDS 2011).
Algae contain a wide variety of bioactive compounds. Lacking an immune system, algae have developed elaborate chemical defense compounds against bacteria, viruses, and fungi, which pose significant threats to algae. It has been estimated that a milliliter of seawater contains 107 viruses, 106 bacteria, and 103 fungal cells (Rheinheimer 1992). Few in-depth studies have been conducted, but work of Kubanek and her colleagues have identified more than 20 different antimicrobial compounds from the surfaces of two common macroalga (Kubanek et al. 2003; Lane et al. 2009). These compounds have been shown to be effective against infectious and pathogenic microbes and show promise against some human diseases. The presence of similar compounds on other seaweeds may help explain the observed in vitro and in vivo anti-HIV activities of several algae. Possible mechanisms of action have been proposed for algal inhibition of HIV including inhibition of viral entry/fusion, reverse transcriptase activity, and integrase and protease activities (Ahn et al. 2004; Baba et al. 1990; Ghosh et al. 2009; McReynolds and Gervay-Hague 2007; Schaeffer and Krylov 2000; Witvrouw and De Clercq 1997). Previously, we reported that a high fucoidan (75%) supplement significantly amplified the CXCR4+ hematopoietic progenitor cell population in the circulation probably by competing with the C-X-C chemokine receptor type 4 (CXCR4) antagonist stromal cell-derived factor-1 (SDF-1). The CXCR4–fucoidan complex resulted in blocking the expression of SDF-1 and led to the increase of the free circulating SDF-1 that mobilized the hematopoietic progenitor cells (Irhimeh et al. 2007). Co-expression of CD4 and CXCR4 on a cell allows HIV isolates to fuse with and infect the cell. We postulated that fucoidan seaweed, which is characterized by its relative large molecular weight, could bind to the T cell L-selectins leading to the blockage of the CXCR4 receptor, thus decreasing the possibility for HIV to infect the cell. Our results support this possible mechanism, providing evidence that HIV concentrations would remain stable and gradually decrease over time.
Because seaweed and Spirulina are not common foods in the USA, there is some question of whether it is even safe for Americans with HIV to eat, and so we used traditional drug-testing methods to demonstrate that dietary algae commonly eaten in other countries is safe in an algae-naïve American population of people with HIV. The use of the combination of a brown seaweed and a blue-green alga was based on the improved efficacy of combination drug ARV therapy compared to monotherapy against HIV. To test this hypothesis, we used a pharmaceutical proof of concept test model. Proof of Concept forms the basis for phase I early clinical testing of drugs and is usually restricted to short (1 to 2 weeks) clinical study of up to six subjects. The endpoints are safety and any indication of efficacy. Phase II clinical testing is used to investigate long-term toxicities, and a minimum of 12 subjects per treatment arm has been suggested (Julious 2005). Phase III trials are the widely known “gold standard” of randomized placebo-controlled clinical trials used to evaluate efficacy compared to standard treatment, and almost always follow phase I/II testing of the treatment.
Our dose of algae was based on the use of algae as whole foods with an established cultural history of dietary intake. The average daily intake of seaweed in Japan is about 5.5 g day−1 (Matsumura 2001); anthropological estimates of Spirulina intake in Chad are about 12 g day−1 (Abdulqader et al. 2000) However, we also had to estimate the maximum tolerance for number of pills/day people would be likely to consume, so we compromised on ten 500-mg capsules day−1 of Undaria pinnatifida sporophyll and ten 600-mg capsules day−1 of Spirulina. Our third arm combined both algae at half dose (2.5 g day−1 U. pinnatifida and 3 g day−1 Spirulina). Although imprecise, the dose reflected our concerns with adherence to a supplement regimen that would be accepted by our study subjects and still give us an indication of toxicity issues, possible efficacy, and impact on quality of life.
Materials and methods
Subjects
This study was conducted according to the guidelines described in the Declaration of Helsinki, and all procedures involving human subjects/patients were approved by the Institutional Review Boards of the University of South Carolina and Palmetto Health Hospital. Written informed consent was obtained from all the subjects. The trial is registered with the National Institutes of Health Clinical Trials (NCT01195077) (Teas 2010). Subjects were given an honorarium of $2/day to take the supplements and $15/blood draw to cover the cost of transportation to the clinic.
Inclusion criteria
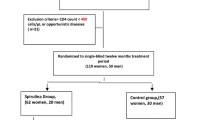
Subjects were all treatment naïve, and were referred by physicians to our study because of declining CD4 cell counts or increasing viral loads. Other study criteria included no allergies to seaweed, Spirulina, shellfish, or iodine and an interest in participating in the study. Participants were instructed to continue to eat their normal diets and vitamins, supplements, and medications during the study. None of the subjects consumed seaweed or Spirulina in either their normal diets or as supplements. Subjects were randomly assigned to a supplement treatment arm, and arms were balanced by supplement type. At each clinic visit, supplements were provided in prefilled 7-day plastic containers with separate compartments for morning and evening doses.
Blood collection and handling
Blood samples were drawn from fasting participants between 0630 and 1030 hours by venipuncture at a consistent time for each subject at baseline and at the end of each treatment period. Blood samples were analyzed by the Richland Palmetto Hospital lab. Metabolic panel included: glucose, blood urea nitrogen (BUN), creatinine, BUN/creatinine ratio, sodium, potassium, chloride, carbon dioxide, calcium, protein, albumin, globulin, A/G ratio, bilirubin, alkaline phosphatase (ALP), aspartate transaminase (AST), and alanine transaminase (ALT). Lipid panel included total cholesterol, triglycerides, high-density cholesterol (HDL), low-density cholesterol (LDL), and very low-density cholesterol (VLDL). Complete blood counts (CBC) with differential included: white blood cells (WBC), red blood cells (RBC), hemoglobin (Hb), hematocrit (Hct), mean cell volume (MCV), mean corpuscular hemoglobin (MCH), mean corpuscular hemoglobin concentration (MCHC), red blood cell distribution width (RDW), platelets, absolute lymphocytes, percent lymphocytes, percent neutrophils, and percent monocytes. HIV-specific measurements included percent CD4 positive of all lymphocytes, CD4 cells (microliters), HIV-1 RNA by PCR copies (milliliters), and log10 HIV-1 RNA.
Undaria and Spirulina supplements
Seaweeds are consumed regularly by millions of people, particularly in Japan and Korea. They have been approved by the United States Food and Drug Administration and are on the list of foods Generally Regarded as Safe (Food and Drug Administration 1982). Spirulina received a class A safety rating by the Dietary Supplements Information Expert Committee of the United States Pharmacopeial Convention (Marles et al. 2011).
Undaria pinnatifida (Harvey) Suringar was harvested from Bahia Bustamante on the Patagonian coast of Argentina (Soriano SA). The sporophylls were removed from the stipes, shade dried, and pulverized before encapsulation (Vicrofer SRL, Buenos Aires, Argentina) into 500-mg capsules. The powder was tested by Soriano SA, and the nutritional analysis is presented in Table 1. Independent testing of the powder for iodine was conducted at the Iodine Research Laboratory at the Boston University School of Medicine (Teas et al. 2009a, b); fucoidan content was determined following the Stevenson analytical method for determining algal sulfated galactans at the Industrial Research Limited, Crown Research Institute in Wellington, NZ (Stevenson and Furneaux 1991). Spirulina was cultivated and harvested by Earthrise (Calipatria, CA). It was spray dried and encapsulated into 600-mg capsules. Earthrise® nutrient composition and bioactive component data for dried Spirulina were based on the Earthrise® label. Nutrient compositions of the supplements are presented in Table 1.
Medical Outcomes Study-HIV quality of life questionnaire
The Medical Outcomes Study-HIV (MOS-HIV) quality of life questionnaire has 30 items clustered around 13 dimensions of quality of life for people with HIV and has been validated in several populations (Wu et al. 1997). The questionnaire scores were calculated using the specified scaling and scoring program (Mapi Research Trust, Lyon, France) and represent percent change from baseline. Subjects were interviewed by the same project coordinator who administered the MOS-HIV quality of life questionnaire while waiting for the phlebotomist. Any comments or health effects also were noted at this time.
Statistical analysis
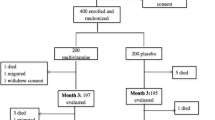
A total of 13 subjects were recruited; data from one subject were excluded because of inconsistent attendance at the clinic visits and complaints about the burden of taking ten capsules/day. Another subject dropped out of the study after the first 2 weeks, and her data also were excluded from analysis, leaving a final sample of 11 evaluable subjects, 5 in phase I and 6 in phase II. Nine of the subjects met the criteria of being healthy and not yet needing ARV; two people in the phase II trial just met the Centers for Disease Control and Prevention (CDC) criteria for ARV and had been placed on a waiting list for ARV. The referring physician felt that participation in our study would possibly benefit her patients until the medications were available. Subjects in phase II were followed for periods ranging from 5 to 14 months. Of these participants, one moved out of state after 13 months, one was lost to follow-up after 14 months, one chose a different treatment, and the two subjects who had CDC-defined AIDS were removed from the study when their CD4 cells again decreased to below 350 cells mL−1, and funding for ARV became available. Although we had hoped to recruit 12 subjects in each arm, the phase II study ended early because the referring physician left the practice and interest in our study ceased.
Although all the study participants had HIV-related lab tests performed, lab requests for metabolic panel, lipid profile, and CBC with differential counts were not standardized until later in the study. The number of evaluable patients for these additional lab results therefore varied. The number for each test is noted in Tables 2, 3, 4, 5, and 6. Lab values are reported as means ± standard error and compared with lab-specific reference values for each endpoint. Repeated measures analyses of variance with posttest were used to determine linear trend (GraphPad Prism 5, USA). Statistical tests were all two-tailed, and significance was set at p < 0.05.
Results
Both short- and long-term toxicity serum results suggest that algae are nontoxic for algae-naive Americans with HIV. Mean values for metabolic panel CBC with differential and lipid panel results remained within reference values (Tables 3, 4, and 5).
Data from the MOS-HIV quality of life questionnaire indicated that subjects had improved sense of well-being during the first 3 weeks of the study (Fig. 1). Although the sample size was too small to do more than estimate general trends, the magnitude of the improvement in quality of life, as measured by subjective feelings of physical health, was similar to that reported by subjects who were no longer limited in vigorous activity (Wu and Rubin 1992).
Efficacy is a minor concern in phase I/II trials; there is some evidence that algae might have decreased viral load and increased CD4 cell counts over time (Fig. 2). This was especially noticeable for subject 7 (Undaria plus Spirulina), who had an increase from 474 to 714 CD4 cells μL−1 over the course of 13 months. This subject met the CDC criteria for clinically significant improvement in CD4 counts (increase of more than 100 CD4 cells μL−1) and decreased viral load (decrease of at least 0.5 log10) from 3.3 to 2.8 log10. The changes fluctuated slightly and sustained trend over time. They reversed the declining trend seen for this subject before enrollment in our study.
Long-term HIV-1 viral load and CD4 cell count data. The total number of CD4 cells (microliters), the HIV-1 RNA by PCR copies (milliliters), and the HIV-1 RNA by PCR log10 are shown for the three study arms: a for subjects taking Spirulina, b for subjects taking Undaria, and c for subjects taking Spirulina and Undaria, highlighting the beneficial trend for subject 7
Discussion
Our clinical data further support existing in vitro and in vivo evidence of the safety of Undaria and Spirulina (Belay 2002; Beppu et al. 2009; Chung et al. 2010; Kim et al. 2010a, b). Our sample size was too small to make any generalizations about efficacy, but the two subjects followed for at least a year showed either improvement or stability. For the subject who remained stable, her life events during that time included unemployment, family losses, and homelessness, all thought to contribute to social stresses associated with declining health.
No previous studies have been conducted using seaweed supplements in people with HIV, but there have been a few clinical trials conducted using Undaria sp./Alaria sp. that support algae as nontoxic and potentially health-enhancing for humans. Hata and colleagues reported improvements in the blood pressure of 18 elderly Japanese patients receiving 3.3 g day−1 additional Undaria (Hata et al. 2001). Similarly, an 8-week study investigating the impact of consuming 4 or 6 g day−1 of Undaria in 27 men and women reported a significant decrease in systolic blood pressure (Teas et al. 2009a). Irhimeh compared the impact of 75% Undaria fucoidan (3 g day−1) on clotting time in a randomized single-blinded placebo-controlled (guar gum) trial in 20 healthy volunteers followed for 12 days (Irhimeh et al. 2009), and the trial reported normal clinical parameters.
Clinical data also are available for the closely related Alaria esculenta. In 30 healthy postmenopausal women who consumed placebo (maltodextrin) or 5 g day−1 Alaria for 8 weeks in conjunction with a soy supplement, there was a significant improvement in estrogen, phytoestrogen, and insulin-like growth factor metabolism, but no impact on thyroid function was observed (Teas et al. 2007, 2009b, 2011).
A clinical trial of 20 people with type 2 diabetes given half Undaria sp. and half Laminaria japonica supplement for 4 weeks improved glycemic control, lowered blood lipids, and increased serum antioxidant activities (Kim et al. 2008). Evidence that fucoidan has specific antiviral properties comes from two clinical trials. In the first, 21 patients with herpes infection consumed an Undaria high fucoidan supplement and reported increased rates of healing and decreased reactivation of viral symptoms (Cooper et al. 2002).
Perhaps of the most relevance to this study, Araya and colleagues conducted a trial using fucoidan in 13 patients with human T-lymphotropic virus type 1-associated neurological disease. Subjects were given 6 g day−1 fucoidan and followed for 6–13 months. Outcome variables were inhibition of cell-to-cell transmission of HTLV-1 in vitro and a 42% decrease in proviral serum concentration. The only toxicity observed was that four patients experienced diarrhea while on fucoidan which resolved when fucoidan treatment ceased. No effect was observed on the host immune cells.
Several studies have included Spirulina supplements to improve immune function in children with HIV (Simpore et al. 2005, 2007), and it has been determined that a minimum dose of 10 g day−1 was needed for children (Simpore et al. 2007). As our study only provided 5 g day−1 for adults, we almost certainly did not give enough Spirulina to properly test its effectiveness against HIV or its immune-enhancing properties in people with HIV. On the other hand, the dose (5 g day−1) of seaweed and the combined dose of seaweed plus Spirulina may have been synergistic and adequate to achieve stability of immune response and viral load. More research is needed before a therapeutic dose can be established, especially for Spirulina.
In studies that have used the MOS-HIV questionnaire, the same magnitude of differences (7% in general health and 5% in mental health) distinguished people with an AIDS-defining health event and those who remained free of opportunistic infection (Wu et al. 1997). Although we did not collect information on comorbidities, based on comments of participants, part of their perceived quality of life improvements were related to getting sick less often, and for subject 7, absence of esophageal reflux when taking the algae supplements.
Drug development is driven nearly exclusively by financial backing of pharmaceutical industry interests; the corollary is that a food that can be purchased in a grocery store has no patentable possibilities. Additionally, there are cultural values associated with algae that give it high value in some countries, notably Japan and Korea, but low value in other countries like the USA and Europe. Our intention had been to include 12 subjects in each treatment arm. However, our study was limited by lack of physician interest. Only one physician we approached had any real interest in nontoxic natural alternatives for her patients with HIV who showed signs of failing health but who did not yet qualify for ARV. When she left the community health clinic, none of her colleagues had any interest in non-pharmaceutical therapies. In the absence of such collaboration, we can only conclude that algal supplementation appeared to be safe and improved the quality of life of the subjects with HIV who did participate in our study, but there may be strong physician resistance to non-pharmaceutical alternatives for patients in resource-rich countries.
Dietary algae may offer immediate support for people with HIV who do not have access to ARV as well as provide new options for future drug development. Additionally, maintaining stable health indicators, especially for people with HIV who have declining health, may be a desirable objective leading to prolongation of the time between HIV infection and AIDS-defining illnesses. Based on our findings and those from other clinical trials using Undaria sp. as well as in vitro and in vivo evidence of nontoxicity, we propose that dietary Undaria and the combination of Undaria and Spirulina are safe to use in a phase III randomized placebo-controlled clinical trial to test for efficacy against HIV/AIDS and its associated opportunistic infections.
References
Abdulqader G, Barsanti L, Tredici MR (2000) Harvest of Arthrospira platensis from Lake Kossorom (Chad) and its household usage among the Kanembu. J Appl Phycol 12:493–498
Ahn MJ, Yoon KD, Min SY, Lee JS, Kim JH, Kim TG, Kim SH, Kim NG, Huh H, Kim J (2004) Inhibition of HIV-1 reverse transcriptase and protease by phlorotannins from the brown alga Ecklonia cava. Biol Pharm Bull 27:544–547
Baba M, Schols D, De Clercq E, Pauwels R, Nagy M, Györgyi-Edelényi J, Low M, Görög S (1990) Novel sulfated polymers as highly potent and selective inhibitors of human immunodeficiency virus replication and giant cell formation. Antimicrob Agents Chemother 34:134–138
Belay A (2002) The potential application of Spirulina (Arthropira) as a nutritional and therapeutic supplement in health management. J Am Nutraceut Assoc 5:27–48
Beppu F, Niwano Y, Sato E, Kohno M, Tsukui T, Hosokawa M, Miyashita K (2009) In vitro and in vivo evaluation of mutagenicity of fucoxanthin (FX) and its metabolite fucoxanthinol (FXOH). J Toxicol Sci 34:693–698
Chung H, Jeun J, Houng S, Jun H, Kweon D, Lee S (2010) Toxicological evaluation of fucoidan from Undaria pinnatifida in vitro and in vivo. Phytother Res 24:1078–1083
Cooper R, Dragar C, Elliot K, Fitton JH, Godwin J, Thompson K (2002) GFS, a preparation of Tasmanian Undaria pinnatifida is associated with healing and inhibition of reactivation of Herpes. BMC Complement Altern Med 2(1):11. doi:10.1186/1472-6882-2-11
Food and Drug Administration F (1982) GRAS status of certain red and brown algae and their extractives [Algae, brown (kelp), or red - NAT, GRAS, REG -184.1120, 184.1121 and 172.365]. Fed Regist 47(207):47373–47376
Ghosh T, Chattopadhyay K, Marschall M, Karmakar P, Mandal P, Ray B (2009) Focus on antivirally active sulfated polysaccharides: from structure–activity analysis to clinical evaluation. Glycobiology 19:2–15
Hata Y, Nakajima K, Uchida J, Hidaka H, Nakano T (2001) Clinical effects of brown seaweed, Undaria pinnatifida (wakame), on blood pressure in hypertensive subjects. J Clin Biochem Nutr 30:43–53
Irhimeh M, Fitton J, Lowenthal R (2007) Fucoidan ingestion increases the expression of CXCR4 on human CD34+ cells. Exp Hematol 35:989–994
Irhimeh M, Fitton J, Lowenthal R (2009) Pilot clinical study to evaluate the anticoagulant activity of fucoidan. Blood Coagul Fibrinolysis 20:607–610
Julious S (2005) Sample size of 12 per group rule of thumb for a pilot study. Pharm Stat 4:287–291
Kim M, Kim J, Choi W, Lee S (2008) Effects of seaweed supplementation on blood glucose concentration, lipid profile, and antioxidant enzyme activities in patients with type 2 diabetes mellitus. Nutr Res Pract 2:62–67
Kim KJ, Lee OH, Lee BY (2010a) Genotoxicity studies on fucoidan from sporophyll of Undaria pinnatifida. Food Chem Toxicol 48:1101–1104
Kim K-J, Lee O-H, Lee H-H, Lee B-Y (2010b) A 4-week repeated oral dose toxicity study of fucoidan from the sporophyll of Undaria pinnatifida in Sprague-Dawley rats. Toxicol 267:154–158
Kubanek J, Jensen P, Keifer P, Sullards MC, Collins D, Fenical W (2003) Seaweed resistance to microbial attack: a targeted chemical defense against marine fungi. PNAS 100:6916–6921
Lane A, Nyadong L, Galhena A, Shearer T, Stout E, Parry M, Kwasnik M, Wang M, Hay M, Fernandez F, Kubanek J (2009) Desorption electrospray ionization mass spectrometry reveals surface-mediated antifungal chemical defense of a tropical seaweed. PNAS 106:7314–7319
Marles R, Barrett M, Barnes J, Chavez M, Gardiner P, Ko R, Mahady G, Low Dog T, Sarma N, Giancaspro G, Sharaf M, Griffiths J (2011) United States pharmacopeia safety evaluation of Spirulina. Crit Rev Food Sci Nutr 51:593–604
Matsumura Y (2001) Nutrition trends in Japan. Asia Pac J Clin Nutr 10(Suppl):S40–S47
McReynolds K, Gervay-Hague J (2007) Chemotherapeutic interventions targeting HIV interactions with host-associated carbohydrates. Chem Rev 107:1533–1552
Payne B, Wilson I, Hateley C, Horvath R, Santibanez-Koref M, Samuels D, Price D, Chinnery P (2011) Mitochondrial aging is accelerated by anti-retroviral therapy through the clonal expansion of mtDNA mutations. Nat Genet 43(8):806–810
Rheinheimer G (1992) Aquatic microbiology, 3rd edn. Wiley, New York
Schaeffer DJ, Krylov VS (2000) Anti HIV activity of extracts and compounds from algae and cyanobacteria. Ecotoxicol Environ Saf 45:208–227
Simpore J, Zongo F, Kabore F, Dansou D, Bere A, Nikiema J-B, Pignatelli S, Biondi DM, Ruberto G, Musumeci S (2005) Nutrition rehabilitation of HIV-infected and HIV-negative undernourished children utilizing Spirulina. Ann Nutr Metab 49:373–380
Simpore J, Pignatelli S, Musumeci S (2007) The effects of Spiruline on the immune functions of HIV-infected undernourished children. J Infect Dev Ctries 1(2):112–117
Stevenson T, Furneaux R (1991) Chemical methods for the analysis of sulfated galactans from red algae. Carbohydr Res 210:277–298
Teas J (2010) Could dietary algae affect immunity and viral counts in people with HIV? C:\Users\teas\Documents\USC-SOM grant\clinical trial page.htm. Accessed 14 Nov 2011.
Teas J, Braverman L, Kurzer M, Pino S, Hurley T, Hebert J (2007) Seaweed and soy: companion foods in Asian cuisine and their effects on thyroid function in American women. J Med Food 10:90–100
Teas J, Baldeón M, Chiriboga D, Davis J, Sarriés A, Braverman L (2009a) Could dietary seaweed reverse metabolic syndrome? Asia Pac J Clin Nutr 18:145–154
Teas J, Hurley T, Hebert J, Franke A, Sepkovic D, Kurzer M (2009b) Dietary seaweed modifies estrogen and phytoestrogen metabolism in healthy postmenopausal women. J Nutr 139:939–944
Teas J, Irhimeh M, Druker S, Hurley T, Hébert J, Savarese T, Kurzer M (2011) Serum IGF-1 concentrations change with soy and seaweed supplements in healthy postmenopausal American women. Nutr Cancer 63:743–748
UNAIDS (2011) UNAIDS Joint United Nations Programme on HIV/AIDS. http://www.unaids.org/en/dataanalysis/epidemiology/2009aidsepidemicupdate/. Accessed 14 Nov 2011
Witvrouw M, De Clercq E (1997) Sulfated polysaccharides extracted from sea algae as potential antiviral drugs. Gen Pharmacol 29:497–511
Wu AW, Rubin HR (1992) Measuring health status and quality of life in HIV and AIDS. Psychol Health 6:251–264
Wu A, Revicki D, Jacobson D, Malitz F (1997) Evidence for reliability, validity and usefulness of the Medical Outcomes Study HIV health survey (MOS-HIV). Qual Life Res 6:481–493
Acknowledgments
We gratefully thank Drs. Ruth Falshaw and Susan Carnachan of the Industrial Research Limited, Crown Research Institute in Wellington, NZ for their glycoprotein analysis of the U. pinnatifida sporophyll; Drs. Lily Randolph, D. Daniels, and Rohit Talwani in Columbia, SC for referring subjects to our study; Drs. Greg Hand and Wesley Dudgeon at the University of South Carolina for assistance in subject interviews; Soriano S.A. for their donation of U. pinnatifida sporophyll and Earthrise Nutritionals for their donation of Spirulina; Dr. Amha Belay and the University of South Carolina Research Education Foundation for financial support.
Open Access
This article is distributed under the terms of the Creative Commons Attribution Noncommercial License which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This is an open access article distributed under the terms of the Creative Commons Attribution Noncommercial License (https://creativecommons.org/licenses/by-nc/2.0), which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
About this article
Cite this article
Teas, J., Irhimeh, M.R. Dietary algae and HIV/AIDS: proof of concept clinical data. J Appl Phycol 24, 575–582 (2012). https://doi.org/10.1007/s10811-011-9766-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10811-011-9766-0