Abstract
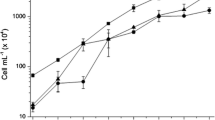
This study investigated the growth rate of chitosan-immobilized cells of the marine cyanobacterium Synechococcus elongatus and its potential application in the removal of nitrogen and phosphorus for wastewater treatment. Immobilized cell cultures had a lag phase of growth due to the immobilization method, and their growth rate was similar to that of free-living cell cultures. Ammonia removal was higher in free cells (54%) than in immobilized cells (29%), but nitrate removal was similar in immobilized (38%) and free cells (44%); phosphorus removal was more efficient in free cells (88%) than in immobilized cells (77%). Chlorophyll a and protein content were higher in immobilized cells. Our study demonstrates that S. elongatus immobilized into chitosan capsules can remove nutrients and is able to maintain a growth rate comparable to that of free cells in culture.




Similar content being viewed by others
References
Aguilar-May B, Sánchez-Saavedra MP, Lizardi J, Voltolina D (2007) Growth of Synechococcus sp. immobilized in chitosan with different times of contact with NaOH. J Appl Phycol 19:181–183 doi:10.1007/s10811-006-9132-9
Andersen RA, Berges JA, Harrison PJ, Watanabe MM (2005) Appendix A: recipes for freshwater and seawater media. In: Andersen RA (ed) Algal culturing techniques. Elsevier, Amsterdam, pp 429–538
Babel S, Kurniawan TA (2003) Low-cost adsorbents for heavy metals uptake from contaminated water: a review. J Hazard Mat B97:219–243 doi:10.1016/S0304-3894(02)00263-7
Bailliez C, Largeau C, Casadevall E (1985) Growth and hydrocarbon production of Botryococcus braunii immobilized in calcium alginate gel. Appl Microbiol Biotechnol 23:99–105
Becker EW (1986) Nutritional properties of microalgae: potentials and constraints. In: Richmond A (ed) Handbook of microalgal mass culture. CRC, Boca Raton, pp 339–420
Chen YC (2001) Immobilized microalga Scenedesmus quadricauda (Chlorophyta, Chlorococcales) for long-term storage and for application for water quality control in fish culture. Aquaculture 195:71–80 doi:10.1016/S0044–8486(00)00540–8
Cheng ZJ, Behnke KC, Dominy WG (2002) Effect of feather meal on growth and body composition of the juvenile Pacific white shrimp, Litopenaeus vannamei. J Appl Aquacult 12:57–70 doi:10.1300/J028v12n01_03
Chevalier P, de la Noüe J (1985) Efficiency of immobilized hyperconcentrated algae for ammonium and orthophosphorus removal from wastewater. Biotechnol Lett 7:395–400 doi:10.1007/BF01166210
Clifford HC (1994) Semi-intensive sensation—a case study in marine shrimp pond management. World Aquaculture 25(3):6–12
Cohen Y (2001) Biofiltration—the treatment of fluids by microorganisms immobilized into the filter bedding material: a review. Bioresour Technol 77:257–274 doi:10.1016/S0960-8524(00)00074-2
Crini G (2006) Non-conventional low-cost adsorbents for dye removal: a review. Bioresour Technol 97:1061–1085 doi:10.1016/j.biortech.2005.05.001
de la Noüe J, Proulx D (1988) Tertiary treatment of urban wastewaters by chitosan-immobilized Phormidium sp. In: Stadler T, Mollion J, Verdus MC (eds) Algal biotechnology. Elsevier, London, pp 159–168
de-Bashan LE, Bashan Y (2004) Recent advances in removing phosphorus from wastewater and its future use as fertilizer (1997–2003). Water Res 38:4222–4246
de-Bashan LE, Bashan Y, Moreno M, Lebsky VK, Bustillos JJ (2002) Increased pigment and lipid content, lipid variety and cell and population size pf the microalgae Chlorella spp. when co-immobilized in alginate beads with the microalgae-growth-promoting bacterium Azospirillum brasilense. Can J Microbiol 48:514–521
Donald K, Scanlan DJ, Carr NG, Mann NH, Joint I (1997) Comparative phosphorous nutrition of the marine cyanobaterium Synechococcus WH7803 and the marine diatom Thalassiosira weissflogii. J Plankt Res 19:1793–1813
Fierro-Reséndiz S, Sánchez-Saavedra MP, Copalcúa C (2008) Nitrate and phosphate removal by chitosan immobilized Scenedesmus. Bioresour Technol 99:1274–1279
Fogg GE, Thake BJ (1987) Algal growth in continuous culture. In: Fogg GE, Thake BJ (eds) Algal cultures and phytoplankton ecology. University of Wisconsin Press, Madison, USA, pp 57–80
Garbisu C, Hall DO, Serra JL (1992) Nitrate and nitrite uptake by free-living and immobilized N-starved cells of Phormidium laminosum. J Appl Phycol 4:139–148
Guillard RRL, Ryther JH (1962) Studies on marine planktonic diatoms I. Cyclotella nana Hustedt and Detonula confervacea (Cleve) Gran. Can J Microbiol 8:229–239
Hach Company (1997) Hach water analysis handbook. Hach, Loveland, USA
Han YH, Lee JS, Kwak JK, Lee EH, Cho MG (1999) High-cultivation of microalgae using microencapsulation. J Korean Fish Soc 32:186–191
Healey FP (1973) Inorganic nutrient uptake and deficiency in algae. Crit Rev Microbiol 3:69–113
Hernández JP, de-Bashan LE, Bashan Y (2006) Starvation enhances phosphorous removal from wastewater by the microalga Chlorella spp. co-immobilized with Asosphirillum brasilense. Enzyme Microbiol Tech 38:190–198
Hu Q, Westerhoff P, Vermaas W (2000) Removal of nitrate from groundwater by cyanobacteria: quantitative assessment of factor influencing nitrate uptake. Appl Environ Microbiol 66:133–139
Ikeya T, Ohki K, Takahashi M, Fujita Y (1997) Study on phosphate uptake of the marine cyanophyte Synechococcus sp. NIBB 1071 in relation to oligotrophic environments in the open ocean. Mar Biol 129:195–202
Joo DS, Cho MG, Lee JS, Park JH, Kwak JK, Han YH, Bucholz R (2001) New strategy for the cultivation of microalgae using microencapsulation. J Microencapsulation 18:567–576
Kaya VM, Picard G (1995) The viability of Scenedesmus bicellularis cells immobilized on alginate screens following nutrient starvation in air at 100% relative humidity. Biotechnol Bioeng 46:459–464
Kaya VM, Picard G (1996) Stability of chitosan gel as entrapment matrix of viable Scenedesmus bicellularis cells immobilized on screens for tertiary treatment of wastewater. Bioresour Technol 56:147–155
Kierstan MPJ, Coughlan MP (1985) Immobilization of cells and enzymes by gel entrapment. In: Woodward J (ed) Immobilized cells and enzymes: a practical approach. IRL, Oxford, pp 39–48
Kurita K (2006) Chitin and chitosan: functional biopolymers from marine crustaceans. Mar Biotechnol 8:203–226
Lau PS, Tam NFY, Wong YS (1997) Wastewater nutrients (N and P) removal by carrageenan and alginate immobilized Chlorella vulgaris. Environ Technol 18:945–951
Lau PS, Tam NFY, Wong YS (1998) Effect of carrageenan immobilization on the physiological activities of Chlorella vulgaris. Bioresour Technol 63:115–121
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
Mallick N (2002) Biotechnological potential of immobilized algae for wastewater N, P and metal removal: a review. BioMetals 15:377–390
Mallick N, Rai LC (1994) Removal of inorganic ions from wastewater by immobilized microalgae. World J Microbiol Biotechnol 10:439–443
Matsunaga T, Nakamura N, Tsuzaki N (1988) Selective production of glutamate by an immobilized marine blue-green alga, Synechococcus sp. Appl Microbiol Biotechnol 28:373–376
Mucott SE, Harfeman, DRF (1996) Method of drinking water treatment with natural cationic polymers. US Patent 268266 http://www.patentstorm.us/patents/5543056-description.html
Nieves M, Voltolina D, Piña P (2005) Growth and biomass production of Tetraselmis suecica and Dunaliella tertiolecta in a standard medium added with three products of zeolitic nature. Aquacult Eng 32:403–410
No HK, Meyers SP (1997) Preparation of chitin and chitosan. In: Muzzarelli RAA, Peter MG (eds) Chitin handbook. European Chitin Society, Grottammare, Italy, pp 475–487
Nübel U, Garcia-Pichel F, Muyzer G (1997) PCR primers to amplify 16S rRNA genes from cyanobacteria. Appl Environ Microbiol 63(8):3327–3332
Pane L, Feletti M, Bertino C, Carli A (1998) Viability of the marine microalga Tetraselmis suecica grown free and immobilized in alginate beads. Aquacult Int 6:411–420
Parsons TR, Maita Y, Lalli CM (1984) A manual of chemical and biological methods for seawater analysis. Pergamon, Oxford
Pastor de Abram A (2004) Quitina y quitosano: obtención, caracterización y aplicaciones. Ciencia y Tecnología para el Desarrollo (CYTED). Centro de Investigación en Alimentación y Desarrollo (CIAD). Resultados del proyecto CYTED IV.14: obtención de quitina y quitosano a partir de desechos de crustáceos. Fondo Editorial, Pontificia Universidad Católica del Perú, Lima, Perú
Perry MJ, Talbot MC, Alberte RS (1981) Photoadaptation in marine phytoplankton: response of the phytosynthetic unit. Mar Biol 62:91–110
Reyes JC, Chávez S, Muro-Pastor MI, Candau P, Florencio FJ (1993) Effect of glucose utilization on nitrite excretion by the unicellular cyanobacterium Synechocystis sp. strain PCC 6803. Appl Environ Microbiol 59:3161–3163
Rinaudo M, Milas M, Le Dung P (1993) Characterization of chitosan: influence of ionic strength and degree of acetylation on chain expansion. Int J Biol Macromol 15:281–285
Saeed A, Iqbal M (2006) Immobilization of blue green microalgae on loofa sponge to biosorb cadmium in repeated shake flask batch and continuous flow fixed bed column reactor system. World J Microbiol Biotechnol 22:775–782
Sasikala C, Ramana CV (1994) Growth and H2 production by Synechococcus spp. using organic/inorganic electron donors. World J Microbiol Biotechnol 10:531–533
Scanlan DJ (2003) Physiological diversity and niche adaptation in marine Synechococcus. Adv Microbiol Physiol 47:1–64
Sokal RR, Rohlf FJ (1979) Biometría: principios y métodos estadísticos en la investigación biológica. Blume, Madrid
Sorokin C (1973) Dry weight, packed cell volume and optical density. In: Stein JR (ed) Handbook of phycological methods. Culture methods and growth measurement. Cambridge University Press, New York, pp 321–343
StatSoft (2002) Statistica for windows. StatSoft, Tulsa, OK
Suzuki I, Horie N, Sugiyama T, Omata T (1995) Identification and characterization of two nitrogen-regulated genes of the cyanobacterium Synechococcus sp. strain PCC7942 required for maximum efficiency of nitrogen assimilation. J Bacteriol 177:290–296
Tam NFY, Wong YS (2000) Effect of immobilized microalgal bead concentrations on wastewater nutrient removal. Environ Pollut 107:145–151
Trevan MD, Mak AL (1988) Immobilized algae and their potential for use as biocatalysts. Trends Biotechnol 6:68–73
Vorlop KD, Klein J (1987) Entrapment of microbial cells in chitosan. Methods Enzymol 135:259–268
Waterbury JB, Watson SW, Guillard RRL, Brand LE (1979) Wide-spread occurrence of a unicellular, marine planktonic, cyanobacterium. Nature 277:293–294
Waterbury JB, Watson SW, Valois FW, Franks DG (1986) Biological and ecological characterization of the marine unicellular cyanobacterium Synechococcus. Can Bull Fish Aquat Sci 472:71–120
Wyman M, Bird C (2007) Lack of control of nitrite assimilation by ammonium in an oceanic picocyanobacterium, Synechococcus sp. Strain WH 8103. Appl Environ Microbiol 9:3028–3033
Yabur R, Basham Y, Hernández-Carmona G (2007) Alginate from the macroalgae Sargassum sinicola as a novel source for microbial immobilization material in wastewater treatment and plant promotion. J Appl Phycol 19:43–53
Yan-Tan EW, Lee VR (2002) Enzymatic hydrolysis of prawn shell waste for the purification of chitin. Department of Chemical Engineering, Loughborough University, Loughborough, UK
Acknowledgments
This work was supported by Consejo Nacional de Ciencia y Tecnología (CONACYT Project 45844) and by Centro de Investigación Científica y de Educación Superior de Ensenada (CICESE Project 7073). B. A.-M. acknowledges a CONACyT PhD scholarship. Alexei F. Licea-Navarro contributed to the molecular identification of S. elongatus. H. Gómez-Villa and C. Chávira-Ortega gave technical assistance. We thank N. Boston and Blue Pencil Science for editing and reviewing the grammar and style of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Aguilar-May, B., del Pilar Sánchez-Saavedra, M. Growth and removal of nitrogen and phosphorus by free-living and chitosan-immobilized cells of the marine cyanobacterium Synechococcus elongatus . J Appl Phycol 21, 353–360 (2009). https://doi.org/10.1007/s10811-008-9376-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10811-008-9376-7