Abstract
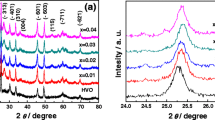
The present study synthesized Mo-doped δ-MnO2 powders with different doping ratios by implementing hydrothermal method. Various analyses, namely X-ray diffraction (XRD), scanning electron microscopy (SEM), transmission electron microscopy (TEM), the Brunauer–Emmett–Teller (BET) method, Raman spectroscopy, X-ray photoelectron spectroscopy (XPS), X-ray fluorescence spectrometer (XRF), and electrochemical measurements, were applied to characterize the dependence of the δ-MnO2 structure, morphology and electrochemical performance on Mo-doping. The experimental results indicated Mo6+ ions entered into the δ-MnO2 crystal lattice and occupied the Mn sites. Appropriate amount of Mo6+ ions doping decreases the charge transfer resistance and increases the Li+ ion diffusion coefficient, thus producing optimal electrochemical performance. The Mo 5% sample with Mo6+/Mn2+ molar ratio of 5:100 in the original solution presented a specific charge capacity of 476.8 mAh g−1 after 100 cycles at 1000 mA g−1 as well as capacity retention ratio of 112.7%.
Graphic abstract









Similar content being viewed by others
References
Zou B, Zhang Y, Wang J et al (2015) Hydrothermally enhanced MnO/reduced graphite oxide composite anode materials for high performance lithium-ion batteries. Electrochim Acta 167:25–31. https://doi.org/10.1016/j.electacta.2015.03.108
Wang J, Yang Y, Huang Z, Kang F (2015) MnO-carbon hybrid nanofiber composites as superior anode materials for lithium-ion batteries. Electrochim Acta 170:164–170. https://doi.org/10.1016/j.electacta.2015.04.157
Chen W, Qie L, Shao Q et al (2012) Controllable synthesis of hollow bipyramid β-MnO2 and its high electrochemical performance for lithium storage. ACS Appl Mater Interfaces 4:1–7
Sinha AK, Pradhan M, Pal T (2013) Morphological evolution of two-dimensional MnO2 nanosheets and their shape transformation to one-dimensional ultralong MnO2 nanowires for robust catalytic activity. J Phys Chem C 117:23976–23986
Zhang X, Yu P, Zhang H et al (2013) Rapid hydrothermal synthesis of hierarchical nanostructures assembled from ultrathin birnessite-type MnO2 nanosheets for supercapacitor applications. Electrochim Acta 89:523–529. https://doi.org/10.1016/j.electacta.2012.11.089
Zhao S, Liu T, Hou D et al (2015) Controlled synthesis of hierarchical birnessite-type MnO2 nanoflowers for supercapacitor applications. Appl Surf Sci 356:259–265. https://doi.org/10.1016/j.apsusc.2015.08.037
Yao W, Wang J, Li H, Lu Y (2014) Flexible α-MnO2 paper formed by millimeter-long nanowires for supercapacitor electrodes. J Power Sources 247:824–830. https://doi.org/10.1016/j.jpowsour.2013.09.039
Bian S, Zhao Y, Xian C (2020) Porous MnO2 hollow spheres constructed by nanosheets and their application in electrochemical capacitors. Mater Lett 111:75–77. https://doi.org/10.1016/j.matlet.2013.08.028
Lu X, Shen C, Zhang Z et al (2018) Core−shell composite fibers for high-performance flexible supercapacitor electrodes. ACS Appl Mater Interfaces 10:4041–4049. https://doi.org/10.1021/acsami.7b12997
Li Q, Wang Z, Li G et al (2012) Design and synthesis of MnO2/Mn/MnO2 sandwich-structured nanotube arrays with high supercapacitive performance for electrochemical energy storage. Nano Lett 12:3803–3807
Li Y, Shi B, Liu W et al (2018) Hollow polypyrrole@MnO2 spheres as nano-sulfur hosts for improved lithium-sulfur batteries. Electrochim Acta 260:912–920. https://doi.org/10.1016/j.electacta.2017.12.068
Hassan S, Suzuki M, Mori S, El-moneim AA (2013) MnO2/carbon nanowalls composite electrode for supercapacitor application. J Power Sources 249:21–27. https://doi.org/10.1016/j.jpowsour.2013.10.097
Zhang X, Wang T, Jiang C et al (2016) Manganese dioxide/cabon nanotubes composite with optimized microstructure via room temperature solution approach for high performance lithium-ion battery anodes. Electrochim Acta 187:465–472. https://doi.org/10.1016/j.electacta.2015.11.084
Dubal DP, Caban-Huertas Z, Holze R, Gomez-Romero P (2016) Growth of polypyrrole nanostructures through reactive templates for energy storage applications. Electrochim Acta 191:346–354. https://doi.org/10.1016/j.electacta.2016.01.078
Liu D, Choi WM (2019) Hierarchical hollow urchin-like structured MnO2 microsphere/carbon nanofiber composites as anode materials for Li-ion batteries. Curr Appl Phys 19:768–774. https://doi.org/10.1016/j.cap.2019.04.005
Feng L, Zhang Y, Wang R et al (2017) Preparation of PPy-coated MnO2 hybrid micromaterials and their improved cyclic performance as anode for lithium-ion batteries. Nanoscale Res Lett. https://doi.org/10.1186/s11671-017-2286-3
Wang C, Liu H, Jiang M et al (2017) Ammonium vanadate @ polypyrrole @ manganese dioxide nanowire arrays with enhanced reversible lithium storage. Appl Surf Sci 416:402–410. https://doi.org/10.1016/j.apsusc.2017.04.069
Xu S, Lu L, Liu L et al (2017) As cathode material for lithium ion battery applications. J Nanosci Nanotechnol 17:2109–2115. https://doi.org/10.1166/jnn.2017.12929
Zeng J, Wang S, Yu J (2014) Al and / or Ni-doped nanomanganese dioxide with anisotropic expansion and their electrochemical characterisation in primary Li–MnO2 batteries. J Solid State Electrochem 18:1585–1591. https://doi.org/10.1007/s10008-013-2372-0
Hu Z, Xiao X, Chen C et al (2014) Al-doped α-MnO2 for high mass-loading pseudocapacitor with excellent cycling stability. Nano Energy 11:226–234. https://doi.org/10.1016/j.nanoen.2014.10.015
Wang X, Yin M, Xue H et al (2018) Simple microwave synthesis and improved electrochemical performance of Nb-doped MnO2/reduced graphene oxide composite as anode material for lithium-ion batteries. Ionics 24:2583–2590. https://doi.org/10.1007/s11581-017-2401-6
Zhao K, Sun C, Yu Y et al (2018) Surface gradient Ti-doped MnO2 nanowires for high-rate and long-life lithium battery. ACS Appl Mater Interfaces 10:44376–44384. https://doi.org/10.1021/acsami.8b13376
Hashem AM, Abuzeid HM, Narayanan N et al (2011) Synthesis, structure, magnetic, electrical and electrochemical properties of Al, Cu and Mg doped MnO2. Mater Chem Phys 130:33–38. https://doi.org/10.1016/j.matchemphys.2011.04.074
Poonguzhali R, Shanmugam N, Gobi R et al (2015) Effect of Fe doping on the electrochemical capacitor behavior of MnO2 nanocrystals. J Power Sources 293:790–798. https://doi.org/10.1016/j.jpowsour.2015.06.021
Gao Q, Wang J, Ke B et al (2018) Fe doped δ-MnO2 nanoneedles as advanced supercapacitor electrodes. Ceram Int 44:18770–18775. https://doi.org/10.1016/j.ceramint.2018.07.108
Poonguzhali R, Shanmugam N, Gobi R et al (2015) Influence of Zn doping on the electrochemical capacitor behavior of MnO2 nanocrystals. RSC Adv 5:45407–45415. https://doi.org/10.1039/c5ra01326g
Yang Y, Huang X, Yang Y et al (2019) Improving the rate performance of manganese dioxide by doping with Cu2+, Co2+ and Ni2+ ions. Int J Electrochem Sci 14:3673–3683. https://doi.org/10.20964/2019.04.30
Liu Y, Zhou H, Cao R et al (2019) Different behaviors of birnessite-type MnO2 modified by Ce and Mo for removing carcinogenic airborne benzene. Mater Chem Phys 221:457–466. https://doi.org/10.1016/j.matchemphys.2018.09.077
Radhamani AV, Surendra MK, Rao MSR (2018) Applied Surface Science Zn doped δ-MnO2 nano flakes : An efficient electrode material for aqueous and solid state asymmetric supercapacitors. Appl Surf Sci 450:209–218. https://doi.org/10.1016/j.apsusc.2018.04.081
Zhu AL, Wang J, Rong S (2017) Cerium modified birnessite-type MnO2 for gaseous formaldehyde oxidation at low temperature. Appl Catal B 211:212–221. https://doi.org/10.1016/j.apcatb.2017.04.025
Nagaraju G, Ko YH, Cha SM et al (2016) A facile one-step approach to hierarchically assembled core–shell-like MnO2@MnO2 nanoarchitectures on carbon fibers: an efficient and flexible electrode material to. Nano Res 9:1507–1522. https://doi.org/10.1007/s12274-016-1047-4
Xu Z, Wang D, Zhao J et al (2017) Snowflake-like core-shell α-MnO2@δ-MnO2 for high performance asymmetric supercapacitor. Electrochim Acta 251:344–354. https://doi.org/10.1016/j.electacta.2017.08.146
Yan D, Zhang H, Li S et al (2014) Formation of ultrafine three-dimensional hierarchical birnessite-type MnO2 nanoflowers for supercapacitor. J Alloy Compd 607:245–250. https://doi.org/10.1016/j.jallcom.2014.04.077
Wang G, Ma Z, Zhang G et al (2015) Cerium-doped porous K-birnessite manganese oxides microspheres as pseudocapacitor electrode material with improved electrochemical capacitance. Electrochim Acta 182:1070–1077. https://doi.org/10.1016/j.electacta.2015.10.028
Yao K, Xu Z, Huang J et al (2019) Bundled defect-rich MoS2 for a high-rate and long-life sodium-ion battery: achieving 3D diffusion of sodium ion by vacancies to improve kinetics. Small 15:1–7. https://doi.org/10.1002/smll.201805405
Zhu L, Wang J, Rong S et al (2017) Applied catalysis B: environmental cerium modified birnessite-type MnO2 for gaseous formaldehyde oxidation at low temperature. Appl Catal B 211:212–221. https://doi.org/10.1016/j.apcatb.2017.04.025
Sun L, Cao Q, Hu B et al (2011) Applied catalysis A: general Synthesis, characterization and catalytic activities of vanadium—cryptomelane manganese oxides in low-temperature NO reduction with NH3. Appl Catal A 393:323–330. https://doi.org/10.1016/j.apcata.2010.12.012
Xiao X, Sun S, Mcbride MB, Lemley AT (2013) Degradation of ciprofloxacin by cryptomelane-type manganese (III/IV) oxides. Electron Suppl Mater 20:10–21. https://doi.org/10.1007/s11356-012-1026-6
Zhang L, Tu J, Lyu L, Hu C (2016) Enhanced catalytic degradation of ciprofloxacin over Ce-doped OMS-2 microspheres. Appl Catal B 181:561–569. https://doi.org/10.1016/j.apcatb.2015.08.029
Xia Y, Zhang W, Huang H et al (2011) Synthesis and electrochemical properties of Nb-doped Li3V2(PO4)3/C cathode materials for lithium-ion batteries. Mater Sci Eng B 176:633–639. https://doi.org/10.1016/j.mseb.2011.02.006
Qiao YQ, Wang XL, Xiang JY et al (2011) Electrochemical performance of Li3V2( PO4)3/C cathode materials using stearic acid as a carbon source. Electrochim Acta 56:2269–2275. https://doi.org/10.1016/j.electacta.2010.11.073
Yue J, Gu X, Jiang X et al (2015) Coaxial manganese dioxide@N-doped carbon nanotubes as superior anodes for lithium ion batteries. Electrochim Acta 182:676–681. https://doi.org/10.1016/j.electacta.2015.09.150
Liu H, Hu Z, Tian L et al (2016) Reduced graphene oxide anchored with δ-MnO2 nanoscrolls as anode materials for enhanced Li-ion storage. Ceram Int 42:13519–13524. https://doi.org/10.1016/j.ceramint.2016.05.144
Zhang Y, Liu H, Zhu Z et al (2013) Electrochimica Acta A green hydrothermal approach for the preparation of graphene/α-MnO2 3D network as anode for lithium ion battery. Electrochim Acta 108:465–471. https://doi.org/10.1016/j.electacta.2013.07.002
Chen J, Wang Y, He X et al (2014) Electrochimica Acta Electrochemical properties of MnO2 nanorods as anode materials for lithium ion batteries. Electrochim Acta 142:152–156. https://doi.org/10.1016/j.electacta.2014.07.089
Zhang W, Zhang B, Jin H et al (2018) Waste eggshell as bio-template to synthesize high capacity δ-MnO2 nanoplatelets anode for lithium ion battery. Ceram Int 44:20441–20448. https://doi.org/10.1016/j.ceramint.2018.08.038
Zhang B, Wan J, Zhang B, Wan J (2019) Waste utilization method for -MnO2 anode composited with MWCNT and graphene by embedded on conductive paper for lithium ion battery. NANO. https://doi.org/10.1142/S1793292019500516
Fan Y, Clavel G, Pinna N (2018) Effect of passivating Al2O3 thin films on MnO2/carbon nanotube composite lithium-ion battery anodes. J Nanopart Res 20:216
Acknowledgements
The present study was supported by the China Postdoctoral Science Foundation (2016M592746) and the Doctor Initiation Funding Scheme of the Shaanxi University of Science & Technology (BJ15-04).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Xia, A., Zhao, C., Yu, W. et al. Mo-doped δ-MnO2 anode material synthesis and electrochemical performance for lithium-ion batteries. J Appl Electrochem 50, 733–744 (2020). https://doi.org/10.1007/s10800-020-01431-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10800-020-01431-2