Abstract
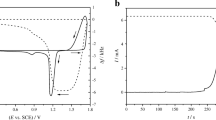
The kinetics of interaction of benzotriazole (C6H5N3, BTAH) with the surface of copper in salt water were studied using an electrochemical quartz crystal microbalance and X-ray photoelectron spectroscopy (XPS). Upon injecting BTAH into the electrolyte, three regions appear in the time response of the microbalance. Region I (at short time of few minutes), exhibits rapid linear growth of mass with time, which is attributed to the formation of a protective Cu(I)BTA complex. Region II reveals attachment of BTAH at a slower rate onto the inner Cu(I)BTA complex. Region III is a plateau indicating that the BTAH film attains an equilibrium mass and thickness, which increase with the concentration of BTAH. The intensity of the N1s peak in the XPS spectra increases with the time of immersion, indicating more BTAH on the surface. The results suggest a duplex inhibitor film composed of an inner thin layer of Cu(I)BTA and an outer layer of physically adsorbed BTAH which increases in thickness with time and BTAH concentration. They also offer an explanation for the much documented findings of simultaneous increase of the polarization resistance and decrease of double layer capacity with inhibitor concentration and time of immersion.





Similar content being viewed by others
References
Hashemi T, Hogarth CA (1988) Electrochim Acta 33(8):1123
Fox PG, Lewis G, Boden PJ (1979) Corros Sci 19(7):457
Cotton JB, Scholes IR (1967) Br Corros J London 2(1):1
Heakal FE, Haruyama S (1980) Corros Sci 20(7):887
Chan HYH, Weaver MJ (1998) Langmuir 15(9):3348
Polewska W, Vogt MR, Magnussen OM, Behm RJ (1999) J Phys Chem B 103(47):10440
Tromans D, Li G (2002) Electrochem Solid St Lett5(2):5
Schultz ZD, Biggin ME, White JO, Gewirth AA (2004) Anal Chem 76(3):604
Al-Hinai AT, Osseo-Asare K (2003) Electrochem Solid St 6(5):23
Hegde S, Babu SV (2003) Electrochem Solid St 6(10):G126
Tsai TH, Yen SC (2003) Appl Surf Sci 210(3, 4):190
Vogt BD, Lin EK, Wu WL, White CC (2004) J Phys Chem B 108(34):12685
Frignani A, Fonsati M, Monticelli C, Brunoro G (1999) Corros Sci 41(6):1217
Jin-Hua C, Zhi-Cheng L, Shu C, Li-Hua N, Shou-Zhuo Y (1997) Electrochim Acta 43(3, 4):265
Telegdi J, Shaban A, Kalman E (2000) Electrochim Acta 45(22):3639
Metikos-Hukovic M, Babic R, Marinovic A (1998) J Electrochem Soc 145(12):4045
Ravichandran R, Rajendran N (2005) Appl Surf Sci 239(2):182
Ravichandran R, Nanjundan S, Rajendran N (2004) J Appl Electrochem 34(11):1171
Papapanayiotou D, Deligianni H, Alkire RC (1998) J Electrochem Soc 145(9):3016
Tantavichet N, Pritzker M (2006) J Appl Electrochem 36(1):49
Scendo M, Malyszko J (2000) J Electrochem Soc 147(5):1758
Schmidt W, Alkire RC, Gewirth AA (1996) J Electrochem Soc 143(10):3122
Deshpande S, Kuiry SC, Klimov M, Obeng Y, Seal S (2004) J Electrochem Soc 151(11):G788; S. Deshpande, Kuiry SC, Klimov M, Seal S (2005) J Electrochem Solid St 8(4):G98
Li XJ, Guo DM, Ren RK, Jin ZJ (2006) In: Cai G, Xu X, Kang R (eds) Advances in grinding and abrasive technology XIII, key engineering materials, vol 304, 305. Trans Tech Publications, Switzerland, pp 350–354
Fang JY, Tsai MS, Dai BT, Wu SY, Feng MS (2005) Electrochem Solid St 8(5):G128
Babic-Samardzija K, Hackerman N (2005) J Solid St Electrochem 9(7):483
Sayed SY, El-Deab MS, El-Anadouli BE, Ateya BG (2003) J Phys Chem 107(23):5575
Cao PG, Yao JL, Zheng JW, Gu RA, Tian ZQ (2002) Langmuir 18(1):100
Brusic V, Frisch MA, Eldredge BN, Novak FP, Kanfman FB, Rush BM, Frankel GS (1991) J Electrochem Soc 138(8):2253
Fenelon AM, Breslin CB (2001) J Appl Electrochem 31(5):509
Walker R (1999) Br Corros J 34(4):304
Walsh JF, Dhariwal HS, Gutierrez-Sosa A, Finetti P, Muryn CA, Brookes NB, Oldman RJ, Thornton G (1998) Surf Sci 415(3):423
Jiang Y, Adams JB, Sun D (2004) J Phys Chem B 108(34):12851
Hegazy HS, Ashour EA, Ateya BG (2001) J Appl Electrochem 31(11):1261
Poling GW (1970) Corros Sci 10(5):359
Mansfield F, Smith T, Parry ET (1971) Corrosion 28(7):289
Clerc C, Alkire R (1991) J Electrochem Soc 138(1):25
Alkire R, Cangellari A (1989) J Electrochem Soc 136(4):913
Sauerbrey G (1959) Z Physik 155(206):206
Marx KA (2003) Biomacromolecules 4(5):1099
Zhou A, Xie B, Xie N (2000) Corros Sci 42(3):469
Mansikkamäki K, Ahonen P, Fabricius G, Murtomaki L, Kontturi K (2005) J Electrochem Soc 152(1):B12
Qafsaoui W, Blanc C, Pebere N, Takenouti H, Srhiri A, Mankowski G (2002) Electrochim Acta 47(27):4339
Hepel M, Cateforis E (2001) Electrochim Acta 46(24, 25):3801
Fonsati M, Zucchi F, Trabanelli G (1998) Electrochim Acta 44(2, 3):311
Kern P, Landolt D (2001) J Electrochem Soc 148(6):B228
Szocs E, Vastag Gy, Shaban A, Kalman E (2005) Corros Sci 47(4):893
Chmielova M, Seidlerova J, Weiss Z (2003) Corros Sci 45(5):883
Kear G, Barker BD, Walsh FC (2004) Corros Sci 46(1):109
Davies PR, Edwards D, Richards D (2004) Surf Sci 573(2):284
Warren MR, Madden JD (2006) Synthetic Met 156(9, 10):724
Al Kharafi FM, Ateya BG (2002) J Electrochem Soc 149(6):B206
Acknowledgments
The authors gratefully acknowledge support of this work by the Research Administration of Kuwait University, under Grant Numbers SC03/02 and GS01/01.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Al Kharafi, F.M., Abdullah, A.M. & Ateya, B.G. A quartz crystal microbalance study of the kinetics of interaction of benzotriazole with copper. J Appl Electrochem 37, 1177–1182 (2007). https://doi.org/10.1007/s10800-007-9383-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10800-007-9383-4