Abstract
Purpose
The purpose of our study was to evaluate the interocular symmetry and distribution of peripapillary vessel density in young myopic eyes.
Methods
A cross-sectional observational study was designed. A total of 174 eyes of 87 young myopic patients were recruited in this study. According to spherical equivalent (SE), 48 eyes were classified as mild myopia with a mean SE of − 2.12D (SD 0.66D), 66 as moderate myopia with a mean SE of − 4.50D (SD 0.87D), and 60 as high myopia with a mean SE of − 7.39D (SD 1.30D). Optical coherence tomography angiography (OCTA) was used to measure the vessel density. The distribution and interocular symmetry of peripapillary vessel densities were analyzed.
Results
The vessel densities in the whole image, peripapillary, superior and inferior sectors were significantly lower in the high myopia group than in the mild or moderate myopia group (All P < 0.001), and the density in the nasal sector was significantly lower in the high myopia group than in the mild group. And most interesting, the vessel densities in the inside disc and temporal sector showed no difference among the three myopic groups (All P > 0.05). By Pearson correlation analysis, the vessel densities in the whole image, peripapillary, superior, inferior and nasal sectors were negatively correlated with axial length (AL) and SE (All P < 0.001), but vessel densities in the inside disc and temporal sector did not show this correlation (All P > 0.05). Interocular symmetry was observed in all the vascular parameters through paired-samples t-tests (All P > 0.05), intraclass correlation coefficient (ICC) and Pearson correlation analysis (All P < 0.001).
Conclusion
The density of radial peripapillary capillaries decreased in the myopic eye with axial elongation, and optical vascular parameters showed significant interocular symmetry among young myopic eyes.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Myopia has become a major health problem around the world, particularly in East Asia, due to its complications that may cause blindness [1]. In myopic eyes, with the changes in the optic disc, parapapillary zone and ocular elongation axis, the retina may thin and less oxygen, leading to decreased blood circulation and retinal microvascular attenuation [2, 3]. So, retinal vascular morphology should be timely evaluated in myopic patients.
Interocular symmetry is an indicator to judge the pathological changes in eyes [4]. Asymmetry may occur between organ pairs during the progression of diseases [5]. Indeed, asymmetry between eyes is valuable for identifying pathological conditions, such as early glaucomatous damage [6]. Interocular asymmetry between retinal biometric parameters may be indicative of disease [7, 8]. However, the interocular symmetry of vessel density (VD) in myopia is not well characterized. Shahlaee et al. [9] found no significant differences in foveal avascular zone area (FAZA) between myopic patients and healthy controls. Hou et al. [6] reported that interocular asymmetry of vessel density can be quantified by OCT-A measurement, and glaucoma patients demonstrated significant vessel density asymmetry compared with healthy eyes. Therefore, interocular vessel density asymmetry can help identify pathological conditions.
Radial peripapillary capillaries (RPCs), comprising a unique network of capillary beds within a retinal nerve fiber layer (RNFL), provide nutrition for retinal ganglion cell axons. However, information is limited regarding interocular symmetry of peripapillary vessel density and distribution in myopia. Therefore, the current study was performed to evaluate the interocular symmetry and distribution of peripapillary vessel density in young myopic eyes.
Materials and methods
Subjects
We performed a cross-sectional observational study on myopic eyes of young Chinese. The study was approved by the Ethics Committee of Shanghai Tenth People’s Hospital (No: 20KT168) and was registered at Chinese Clinical Trial Registry (no: ChiCTR2000035768). A total of 174 eyes of 87 consecutive myopic candidates examined at Shanghai Tenth People’s Hospital were enrolled from September 2020 to August 2021. The median age was 25 years (range, 18–35 years). The average spherical equivalent (SE) was − 4.84 D (SD 2.31D, range − 0.5D to − 10.625D), and the average axial length (AL) was 25.56 ± 1.21 mm (range, 22.86 to 28.49 mm). According to SE, the subjects were divided into three groups: mild myopia group (− 0.5 D ≤ SE < − 3.0 D), moderate myopia group (− 3 D ≤ SE < -6.0 D and high myopia group (SE ≥ − 6.0 D). Myopia was mild in 48 eyes with a mean SE of − 2.12D (SD 0.66D), moderate in 66 eyes with a mean SE of − 4.50D (SD 0.87D) and high in 60 eyes with a mean SE of − 7.39D (SD 1.30D). None had a history of ocular surgery, ocular trauma, or systemic or ocular health abnormality. The demographic and ophthalmic characteristics of the three groups are summarized in Table 1. The study adhered to the tenets of the Declaration of Helsinki, and written informed consent was obtained by all subjects.
All eligible subjects were invited to undergo a comprehensive eye examination. Visual acuity, refraction and intraocular pressure (IOP) were examined by a noncontact tonometer; AL in each eye by the IOL Master 700® (Carl Zeiss Meditec AG, Jena, Germany); anterior segment by a slit lamp; fundus evaluated by slit-lamp biomicroscopy using Goldmann three-mirror contact lens and wide-field laser ophthalmoscopy (Optos, Marlborough, MA, UK). Exclusion criteria included a history of prior vitreous or retinal surgery, anisometropic eye, pathologic myopia, the glaucoma subject and glaucoma suspect, evidence of retinal disease affecting the retina or optic nerves by examination and systemic diseases potentially affecting the eyes. Measurements were performed from 8:00 to 11:30 to minimize the potential impact of diurnal variation.
Optical coherence tomography angiography
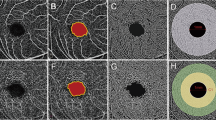
The Optovue Angiovue software (Optovue, Inc. version 2015.1.1.98) was used for characterizing the vascular structures of the retina at the capillary level. The optic disc OCTA scan was performed as volumetric scans generally covering an area of 4.5 × 4.5 mm2 centered around the optic disc. Whole enface image vessel density in optic nerve head (ONH) was measured in the entire 4.5 × 4.5 mm2 image, and circumpapillary vessel density was calculated in a 750-µm-wide elliptical annulus extending from the optic disc boundary. All the measurements were taken by one well-trained investigator (author LL). Studies have reported that vessel density was lower on OCTA images with weaker signals [10,11,12], so only images with scan quality of 6 or greater and without significant motion artifacts were included in the study. The measurements were made for each patient in the OCTA according to the mean of values acquired for two times.
Statistical analysis
Statistical analyses were performed using SPSS V.17.0 software (SPSS Inc, Chicago, IL, USA).
The non-normally distributed data were analyzed by one-sample K–S test. The continuous variables (SE, AL and IOP) were compared among three groups with one-way analysis of variance (except gender compared by Chi-square tests). One-way analysis of variance with the post hoc Bonferroni correction was performed to compare vessel density parameters among three groups and differences between each pair of groups. The correlation between vessel density and AL/SE was measured by the Pearson correlation. Interocular symmetry was evaluated using paired-samples t-test, Pearson’s correlation coefficient and ICC values. P value of less than 0.05 was considered statistically significant.
Results
Basic demographic and ophthalmic characteristics
A total of 184 eyes of 92 subjects were initially enrolled. Five subjects were excluded due to poor-quality OCTA images. The remaining 174 eyes of 87 subjects were included, consisting of 48 eyes from 24 subjects in the mild myopia group (− 0.5 ≤ SE < -3.0 D; mean − 2.12 ± 0.66D), 66 eyes from 33 subjects in the moderate myopia group (− 3.0 ≤ SE < -6.0 D; mean − 4.50 ± 0.87 D) and 60 eyes from 30 subjects in the high myopia group (SE ≥ -6.0 D; mean − 7.39 ± 1.30 D). The basic demographics and ocular characteristics are shown in Table 1. There were significant differences among three groups in AL and SE (all P < 0.01), but not in age, gender and IOP (all P value > 0.05). The high myopia group had a longer AL (24.44 ± 0.69 mm, 25.48 ± 0.77 mm and 26.56 ± 0.11 mm in the mild myopia, moderate myopia and high myopia groups, respectively, P < 0.01).
Vessel density and distribution in three groups
The vessel densities in optic nerve heads are presented in Table 2. The whole-image RPC density in the high myopia group was 47.88%, significantly lower than those in mild myopia group (50.14%) and moderate myopia group (49.26%). The whole-image RPC density tended to decrease with the aggravation of myopia.
We further clarified the distribution of vessel density around the optic disc. The vessel densities in the peripapillary, superior and inferior sectors were significantly lower in the high myopia group compared with those in the mild or moderate myopia group, and the vessel density in the nasal sector was significantly lower in the high myopia group than in the mild group (All P value < 0.001). Surprisingly, the vessel density in the inside disc and temporal sector showed no difference among the three groups (All P value > 0.05) (Table 2, Fig. 1).
Correlations of vascular parameters with AL and SE
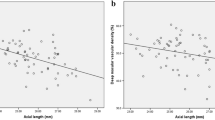
Pearson’s correlation analyses were conducted to investigate the associations between vascular parameters and ocular variables in all study eyes. By analysis, the whole and peripapillary vessel densities were negatively correlated with AL (r = − 0.506, P < 0.001, r = − 0.423, P < 0.001; respectively), while the vessel density in the inside disc did not show this correlation (r = − 0.068, P = 0.374). Among different sectors, a significantly negative correlation was observed between AL and peripapillary vessel density in the superior sector (r = − 0.392, P < 0.001), inferior sector (r = − 0.447, P < 0.001) and nasal sector (r = − 0.334, P < 0.001). In contrast, AL showed no association with the vessel density in the temporal sector (r = 0.110, P = 0.149) (Table 3).
Similar results were found between SE and vessel density. The whole and peripapillary vessel densities were negatively correlated with SE (r = − 0.453, P < 0.001, r = − 0.411, P < 0.001; respectively). There was no correlation between the vessel density in the inside disc and SE (r = − 0.029, P = 0.706). As expected, the SE was strongly and inversely associated with the peripapillary vessel density in the superior sector (r = − 0.410, P < 0.001), inferior sector (r = − 0.393, P < 0.001) and nasal sector (r = − 0.316, P < 0.001). However, we did not observe any correlation between the vessel density in the temporal sector and SE (r = 0.062, P = 0.415) (Table 3).
Interocular symmetry of vessel density parameters in optic nerve heads
Table 4 summarizes the interocular symmetry of SE, AL and VD. There was no significant interocular difference in all the three parameters among three groups through paired-samples t-tests (All P value > 0.05). Pearson’s correlation analysis showed high interocular symmetry of VD in three groups (All P value < 0.01). In addition, the interocular correlation coefficients (ICCs) verified the significant symmetry of the vessel density in the whole image, inside disc, peripapillary and four sectors (P < 0.001 in all ICC values). All the evidence suggests that vessel density parameters of optic nerve heads show interocular symmetric features.
Discussion
In our study, we evaluated the RPC density that directly supplies the retinal ganglion cells in young myopic individuals. To reduce the effect of AL and SE, anisometropic eyes were excluded. Vessel density displayed evident interocular symmetry in three myopic groups. Interocular vessel density asymmetry may serve as a complementary parameter to assess retinal disorders. OCTA allows noninvasive visualization and quantification of the retinal vasculature without the use of exogenous intravenous dye injection [13]. Reproducible quantitative tools have emerged to assess the vessel density around the optic disc, including in the optic nerve head and RPC layer [10, 14]. This quantification can provide detailed information about retinal vascular circulation in the treatment of subclinical retinal and optic diseases [15].
Previous studies using OCTA have revealed vascular changes in myopia may be related to axial elongation. Using OCTA, Wang et al. found a significant reduction in RPC flow density in severe myopic eyes, compared with that in emmetropic eyes [16]. Further, the peripapillary vessel density drops in myopia patients, and much in those with myopic glaucoma [2]. Yaprak AC et al. have reported that the whole and peripapillary vessel densities were significantly decreased in the high myopia group but did not observe any correlation between AL and inside-disc vessel density [17]. Consistently to previous reports, we found an obvious decrease in vessel density in all regions of the RPC area in high myopia group eyes than in the mild myopia group, except that in the inside disc and temporal sector. OCT angiography has shown that high myopia can decrease retinal perfusion in the peripapillary region, compared with the mild myopia.
In addition, we found that RPC vessel density was negatively related to the AL and myopia diopter, but in the inside disc and temporal sector, RPC vessel density was not markedly associated with refractive error and axial length. In other words, with the increase of AL or myopia diopter, RPC vessel density decreased in most sectors of the peripapillary region, except for the inside disc and temporal sector. These findings indicate that retinal vascular dropout in the myopic process may be related to the mechanical axial elongation.
Since the eyeball in a myope is prolate and may further stretch and thin the retina and shift the optic nerve. The optic nerve head deformation may lead to retinal structural alterations in myopic eyes [18]. Myopic eyeball elongation decreases retinal function and oxygen consumption, as shown by the low blood circulation and retinal capillary loss in high myopia [3, 19]. A decrease in the vessel density may lead to hypoxia and nutritional deficiency in the retina and optic nerves, which may help to explain the relationship between longer AL and various pathologic changes in high myopia.
Interestingly, in our study, the vessel densities in all regions decreased with AL and SE, except those in the inside optic disc and temporal sector. So, we consider that among all the vascular parameters, the flow in the inside optic disc and temporal sector flow is not associated with retinal capillary loss related to myopia. Therefore, myopia can lead to the density of retinal capillary decrease in these peripapillary parts (such as superior, inferior and nasal sectors). Any abnormal vessel density in the inside optic disc and temporal sector can indicate the presence of pathologic disorders. The reduced vessel density could be related to the reduced blood supply around the optic disc, primarily due to mechanical stretching of the corresponding regions. Various studies have investigated the impact of myopic tilted disc on structural change of optic nerve. Fan et al. found that eyes with torted disc exhibited thicker temporal retinal and RNFL and more temporally positioned superior peak of RNFL [20], and during the myopic shift, the tilting and rotation of optic disc may be accompanied by nasal bulging and kinking of retinal nerve fibers [21]. Therefore, it could result in vessel density of the inside optic disc and temporal quadrant does not descend.
Healthy organ pairs mostly show symmetric anatomic features. Interocular asymmetry is observed in the condition of clinical diseases [4]. For instance, glaucoma often shows asymmetric features, particularly in the early stage [4, 8]. Several researchers have reported the symmetry of the retinal structure measured by OCT. By swept-source optical coherence tomography (SS-OCT), Lee SY et al. have found significant asymmetry of macular inner retinal layers between the glaucoma and normal groups [4]. Kim et al. have detected the symmetrical choroidal thickness of both eyes [22]. Using spectral-domain optical coherence tomography (SD-OCT), Field MG et al. have found that interocular retinal and RNFL thickness asymmetry are efficient to predict early glaucomatous damage [23]. Using OCTA, Shahlaee et al. have found no interocular asymmetry of FAZA among healthy subjects [9]. Glaucoma can significantly increase interocular asymmetry of vessel density in optic nerve head [6]. Our data prove that there were no significant differences between right eyes and left eyes among three myopic group, and vessel density display a high degree of interocular symmetry in three myopic groups even among patients with high myopia. The founding has not been reported before. Hereby by that means, in the case of similar diopters, the peripapillary vessel density is abnormally decreased in one eye, pathological disease should be considered. However, if the changes occurred in both eye, other factors can first be eliminated, such as refractive factors, which could influence the measurements by the OCTA, especially among high myopia patients, inter-eye vessel density asymmetry values may serve as a complementary parameter that can be assessed with other clinical parameters.
The major limitation of this study was that the change in optic nerve head parameters were parallel in this study, and longitudinal studies are needed to better characterize the relationship of vessel density with the development and progression of myopia. Additionally, we sought to eliminate the effect of age, a high proportion of young myopes were included, further studies with a larger age spectrum should be performed to evaluate the changes in the retinal capillaries.
In conclusion, myopic eyes display a high degree of interocular symmetry of optical vascular parameters. Asymmetry analysis can be a potential ancillary tool for diagnosing unilateral pathology that results in subtle asymmetrical capillary dropout in young myopic patients.
Data availability
All data generated during this study are included in this article. Further enquiries can be directed to the corresponding author.
References
Ang M, Flanagan JL, Wong CW, Müller A, Davis A, Keys D, Resnikoff S, Jong M, Wong TY, Sankaridurg P (2020) Review: myopia control strategies recommendations from the 2018 WHO/IAPB/BHVI meeting on myopia. Br J Ophthalmol 104:1482–1487
Suwan Y, Fard MA, Geyman LS, Tantraworasin A, Chui TY, Rosen RB, Ritch R (2018) Association of myopia with peripapillary perfused capillary density in patients with glaucoma: an optical coherence tomography angiography study. JAMA Ophthalmol 136:507–513
Shimada N, Ohno-Matsui K, Harino S, Yoshida T, Yasuzumi K, Kojima A, Kobayashi K, Futagami S, Tokoro T, Mochizuki M (2004) Reduction of retinal blood flow in high myopia. Graefes Arch Clin Exp Ophthalmol 242:284–288
Lee SY, Lee EK, Park KH, Kim DM, Jeoung JW (2016) Asymmetry analysis of macular inner retinal layers for glaucoma diagnosis: swept-source optical coherence tomography study. PLoS ONE 11:e0164866
Lee SY, Jeoung JW, Park KH, Kim DM (2015) Macular ganglion cell imaging study: interocular symmetry of ganglion cell-inner plexiform layer thickness in normal healthy eyes. Am J Ophthalmol 159:315-323.e312
Hou H, Moghimi S, Zangwill LM, Shoji T, Ghahari E, Manalastas PIC, Penteado RC, Weinreb RN (2018) Inter-eye asymmetry of optical coherence tomography angiography vessel density in bilateral glaucoma, glaucoma suspect, and healthy eyes. Am J Ophthalmol 190:69–77
Huynh SC, Wang XY, Burlutsky G, Mitchell P (2007) Symmetry of optical coherence tomography retinal measurements in young children. Am J Ophthalmol 143:518–520
Weinreb RN, Khaw PT (2004) Primary open-angle glaucoma. Lancet 363:1711–1720
Shahlaee A, Pefkianaki M, Hsu J, Ho AC (2016) Measurement of foveal avascular zone dimensions and its reliability in healthy eyes using optical coherence tomography angiography. Am J Ophthalmol 161:50-55.e51
Venugopal JP, Rao HL, Weinreb RN, Pradhan ZS, Dasari S, Riyazuddin M, Puttiah NK, Rao DAS, Devi S, Mansouri K et al (2018) Repeatability of vessel density measurements of optical coherence tomography angiography in normal and glaucoma eyes. Br J Ophthalmol 102:352–357
Mansouri K, Rao HL, Hoskens K, D’Alessandro E, Flores-Reyes EM, Mermoud A, Weinreb RN (2018) Diurnal variations of peripapillary and macular vessel density in glaucomatous eyes using optical coherence tomography angiography. J Glaucoma 27:336–341
Rao HL, Pradhan ZS, Weinreb RN, Reddy HB, Riyazuddin M, Sachdeva S, Puttaiah NK, Jayadev C, Webers CAB (2017) Determinants of peripapillary and macular vessel densities measured by optical coherence tomography angiography in normal eyes. J Glaucoma 26:491–497
de Carlo TE, Romano A, Waheed NK, Duker JS (2015) A review of optical coherence tomography angiography (OCTA). Int J Retina Vitreous 1:5
Jia Y, Morrison JC, Tokayer J, Tan O, Lombardi L, Baumann B, Lu CD, Choi W, Fujimoto JG, Huang D (2012) Quantitative OCT angiography of optic nerve head blood flow. Biomed Opt Express 3:3127–3137
Chen FK, Menghini M, Hansen A, Mackey DA, Constable IJ, Sampson DM (2018) Intrasession repeatability and interocular symmetry of foveal avascular zone and retinal vessel density in OCT angiography. Transl Vis Sci Technol 7:6
Wang X, Kong X, Jiang C, Li M, Yu J, Sun X (2016) Is the peripapillary retinal perfusion related to myopia in healthy eyes? A prospective comparative study. BMJ Open 6:e010791
Yaprak AC, Yaprak L (2021) Retinal microvasculature and optic disc alterations in non-pathological high myopia with optical coherence tomography angiography. Graefes Arch Clin Exp Ophthalmol 259:3221–3227
Sung MS, Lee TH, Heo H, Park SW (2018) Association between optic nerve head deformation and retinal microvasculature in high myopia. Am J Ophthalmol 188:81–90
Man RE, Lamoureux EL, Taouk Y, Xie J, Sasongko MB, Best WJ, Noonan JE, Kawasaki R, Wang JJ, Luu CD (2013) Axial length, retinal function, and oxygen consumption: a potential mechanism for a lower risk of diabetic retinopathy in longer eyes. Invest Ophthalmol Vis Sci 54:7691–7698
Fan Y, Jonas J, Wang Y, Chen C, Wei W (2017) Horizontal and vertical optic disc rotation. Beijing Eye Study PLoS One 12:e0175749
Chan PP, Zhang Y, Pang CP (2023) Myopic tilted disc: mechanism, clinical significance, and public health implication. Front Med (Lausanne) 10:1094937
Kim MS, Lim HB, Lee WH, Won YK, Nam KY, Kim JY (2021) Wide-field swept-source OCT analysis of interocular symmetry of choroidal thickness in subjects with uncomplicated pachychoroid. J Clin Med 10:4253
Field MG, Alasil T, Baniasadi N, Que C, Simavli H, Sobeih D, Sola-Del Valle D, Best MJ, Chen TC (2016) Facilitating glaucoma diagnosis with intereye retinal nerve fiber layer asymmetry using spectral-domain optical coherence tomography. J Glaucoma 25:167–176
Acknowledgements
Thank you to everyone who has helped this study.
Funding
This study was supported by grants from the Clinical Research Plan of SHDC (No. SHDC2020CR1043B-009), and Clinical research project of Shanghai Tenth People’s Hospital (NO. SHYCS08), National Natural Science Foundation of China Grants (82171077) and The Clinical Research Shanghai Municipal Health Commission (202240131).
Author information
Authors and Affiliations
Contributions
FW helped in conception and design; JZ helped in conduct of the study; LL, M-LD and LF collected research data; LL and ML analyzed the data; LL wrote the paper; FW, DX and JZ reviewed and edited the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to declare.
Ethical approval
This study was performed in line with the principles of the Declaration of Helsinki. Approval was granted by the Ethics Committee of Shanghai Tenth People’s Hospital (No: 20kt168) and was registered at Chinese Clinical Trial Registry (No: ChiCTR2000035768).
Consent to participate
Informed consent was obtained from all individual participants included in the study.
Consent to publish
The participant has consented to the submission of the case report to the journal.
Human or animal rights
Our research involving human participants and no animals.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Liu, L., Deng, Ml., Li, M. et al. Assessment interocular symmetry of peripapillary vessel density in young myopes with optical coherence tomographic angiography. Int Ophthalmol 43, 3453–3460 (2023). https://doi.org/10.1007/s10792-023-02737-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10792-023-02737-8