Abstract
Objectives
To assess the relationship between orbital wall fractures connecting to paranasal sinuses (OWF-PNS) and SARS-CoV-2 ocular surface contamination (SARS-CoV-2-OSC) in asymptomatic COVID-19 patients.
Methods
This was a prospective case–control study enrolling two asymptomatic COVID-19 patient cohorts with vs. without OWF-PNS in the case–control ratio of 1:4. All subjects were treated in a German level 1 trauma center during a one-year interval. The main predictor variable was the presence of OWF-PNS (case/control); cases with preoperative conjunctival positivity of SARS-CoV-2 were excluded to rule out the possibility of viral dissemination via the lacrimal gland and/or the nasolacrimal system. The main outcome variable was laboratory-confirmed SARS-CoV-2-OSC (yes/no). Descriptive and bivariate statistics were computed with a statistically significant P ≤ 0.05.
Results
The samples comprised 11 cases and 44 controls (overall: 27.3% females; mean age, 52.7 ± 20.3 years [range, 19–85]). There was a significant association between OWF-PNS and SARS-CoV-2-OSC (P = 0.0001; odds ratio = 20.8; 95% confidence interval = 4.11–105.2; R-squared = 0.38; accuracy = 85.5%), regardless of orbital fracture location (orbital floor vs. medial wall versus both; P = 1.0).
Conclusions
Asymptomatic COVID-19 patients with OWF-PNS are associated with a considerable and almost 21-fold increase in the risk of SARS-CoV-2-OSC, in comparison with those without facial fracture. This could suggest that OWF-PNS is the viral source, requiring particular attention during manipulation of ocular/orbital tissue to prevent viral transmission.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Recently, the present investigators performed a research series pertaining to COVID-19 and craniomaxillofacial surgery. One of our results is that ocular surfaces amid the nasal and oral cavities can potentially be the virus reservoir in COVID-19 patients [1, 2, 3, 4]. It remains unknown whether the orbital fracture could cause contamination of the ocular surface and/or orbital tissue with SARS-CoV-2. If so, the manipulation (i.e., examination and/or surgery) of ocular/orbital tissue in COVID-19 patients requires particular attention to prevent the viral transmission. Moreover, this viral presence was found to be linked to delayed wound healing and/or postoperative infection [2, 3, 4].
The purpose of this study was to answer the following clinical research question: “Among asymptomatic COVID-19 patients (i.e., orbital surgery in symptomatic COVID-19 patients was usually postponed), does orbital wall fracture exposing paranasal sinuses (OWF-PNS, i.e., proximity of maxillary and ethmoid sinuses to the orbital floor and medial orbital wall, respectively) increase the risk of SARS-CoV-2 ocular surface contamination (SARS-CoV-2-OSC)?” The null hypothesis was that there would no significant association between OWF-PNS and SARS-CoV-2-OSC. Our specific aims were to (1) measure SARS-CoV-2-OSC in patients with vs. without OWF-PNS, (2) estimate the association between OWF-PNS and SARS-CoV-2-OSC, and (3) discuss the possible relevance. We supposed the 2011 Oxford Centre for Evidence-Based Medicine’s Level of Evidence 3 at the study end, and the results would be the “first” case–control research findings on this matter.
Materials and methods
Study design/population
To address the research purpose, a prospective case–control study was designed and implemented. The study population was composed of asymptomatic COVID-19 patients with/without fractured midface (FM) treated in a German level 1 trauma center in a “hot-spot” COVID-19 area (> 65,000 severely infected case) during a one-year period.
Concerning inclusion criteria, “case” subjects were those with (1) age ≥ 18 years, (2) willing to participate in the study, (3) no evidence of underlying infections, e.g., rhinosinusitis, (4) asymptomatic COVID-19 (confirmed using the methods we previously explained [2, 3, 4]), (5) radiologically (via computed tomography [CT]) and intraoperatively confirmed OWF-PNS, and (6) at least one “emergent/urgent” indication for operative treatments, e.g., extensive open wound, retrobulbar hematoma, intracranial injury, or polytrauma [2]. The list of unrelated asymptomatic COVID-19 “control” subjects with “no” facial trauma (e.g., radius or hip fracture) was retrieved from the hospital’s patient database, and 4:1 matched by gender and age to individual “cases.” Exclusion criteria were subjects who (1) denied participation in the study, (2) had an underlying sinus and/or ocular/orbital disease such as chronic rhinosinusitis.
The institutional review board approved this study, and the Helsinki Declaration and the STROBE statement were followed throughout the study. All patients gave written consent to participate in this study and to allow the use of their anonymous data.
Study variables
The predictor variable was the presence of OWF-PNS (yes/no) identified by the hospital’s database using the ICD (International Code of Disease), which reflected a complex combination of clinical and radiographic results for the definitive diagnosis. The outcome variable was RT-PCR-confirmed SARS-CoV-2-OSC (yes/no). The methods of RT-PCR for SARS-CoV-2 were described by Pitak-Arnnop et al. [2], and Atum et al. [6].
“Cases (with OWF-PNS)” were swabbed twice: (1) the first time was a preoperative conjunctival swab to rule out viral dissemination from the lacrimal gland and/or the nasolacrimal system and avoid the chance of intraoperative contamination, and (2) the second swab was performed intraoperatively at the OWF-PNS site directly and immediately when the fracture was exposed. In “control” subjects (without facial trauma), conjunctival swabbing was performed in the morning after they woke up because of probability of overnight microbial accumulation [5, 6, 7].
Other study variables were demographic parameters (age, gender).
Data collection and statistical analysis
Anonymous patient data were converted into a collection form in Microsoft Excel 2007 (Microsoft Corp., Redmond, WA/USA). Using a statistical software MedCalc® (MedCalc Software Ltd., Ostend/Belgium), descriptive statistics was computed for each of the study variables, and bivariate analyses were calculated to measure the association between the presence of OWF-PNS and RT-PCR-confirmed SARS-CoV-2-OSC. All evaluations were considered significant at P ≤ 0.05.
Results
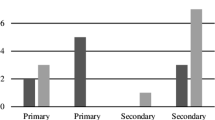
The study was conducted over 12 months including 55 patients (20% [or 11/55] had OWF-PNS; 80% [or 44/55] without facial fracture; 27.3% females) with a mean age of 52.7 ± 20.3 years (range, 19–85). Table 1 presents descriptive and bivariate analyses. SARS-CoV-2-OSC was found in 72% (or 8/11) and 11.4% (or 5/44; all reported multiple hand-eye contacts before the swab) of subjects with and without OWF-PNS, respectively (P = 0.0001; odds ratio = 20.8; 95% confidence interval [95% CI] = 4.11–105.2; R-squared = 0.38), regardless of the orbital fracture location (orbital floor vs. medial wall vs. both; P = 1.0).
With a prevalence of 0.236 (i.e., in this overall cohort of 55 patients, there were 13 patients with positive results from the conjunctival swab RT-PCR [or 23.6%]) and a probability of SARS-CoV-2-OSC cutoff point of 0.20 (i.e., it has generally been assumed and accepted that an adequate study power is 0.8 [80%], which would make β or chance of making a type II error to be 0.2 [20%]), the sensitivity, specificity, positive predictive value, negative predictive value and overall accuracy were 61.5%, 92.9%, 72.7%, 88.7%, and 85.5%, respectively. The secondary diagnostic test calculations demonstrated the posterior probabilities (odds) of positive and negative tests were 73% (95% CI, 45 to 90%) and 11% (95% CI, 6 to 20%). These results suggest that ~ 1 in every 1.4 patients with a positive test result is sick, and ~ 1 in every 1.1 with a negative test result is well (not infected).
Discussion
Although early studies discarded SARS-CoV-2-OSC detection of [6, 7, 8, 9], conjunctival and corneal tissue connects with the upper respiratory tract [1, 2, 3, 4] and contains angiotensin-converting enzyme 2 receptors (ACE-2-R) for virus entry into host cells before replication of new virus particles that infect more cells [6, 7, 8, 9, 10, 11, 12]. Our previous studies revealed conjunctivitis in 6–32% of COVID-19 patients [1], and most orbital retractors were contaminated with SARS-CoV-2 during midfacial fracture repair [2]. Other systematic reviews elucidated low ocular-tissue tropism of this virus (range, 1–36%), which depends on patient’s symptoms [7, 8, 9, 10, 11, 12]. This finding is consistent with our results, i.e., 11.4% in non-OWF-PNS patients, or approximately 1 in every 10 asymptomatic COVID-19 patients without facial fracture will have SARS-CoV-2-OSC. Eye-hand contamination may need clinical concern.
Despite no control of confounding factors in this study, e.g., hand-eye contact time and frequency, effects of antimicrobial agents in tears (e.g., immunoglobulin A, lactoferrin, lysozyme, and lipocalin), blinking, or washing by tears [8, 11, 12], 61.5% of our “cases” had SARS-CoV-2-OSC. In other words, ocular surfaces of approximately 6 in every 10 asymptomatic COVID-19 patients with orbital fracture are contaminated with SARS-CoV-2. The possible explanation is a link between SARS-CoV-2-OSC and ACE-2-R in conjunctival epithelial cells and fibroblasts, corneal epithelial and endothelial cells, retina, and aqueous humor [7, 8, 9, 12]. The virus in the paranasal sinuses can sneak out through the fracture line (i.e., airborne contagion), herniated orbital fat, or blood clot [13, 14, 15] or could be inoculated in the lacrimal gland or its drainage system [8, 9, 11]. Albeit quarantined, the mouth and nostrils are also continuous droplet and aerosol generators in daily life [9, 12]. All of these paths may cause the re-infection of SARS-CoV-2 and/or prolonged viral shedding. Recently, we found extended SARS-CoV-2 shedding in nasal fracture patients, even though the swab tests were negative twice [4].
Another concern in COVID-19 patients is orbital infection. Apart from the commonly mentioned rhino-orbital-cerebral mucormycosis [16], COVID-19-related orbital abscess has been sporadically reported in the literature [17]. Indeed, approximately 1–6% of OWF (especially those with sinusitis 1–2 weeks pre-trauma or as long as 4–5 weeks post-injury [14, 15]) develop orbital cellulitis, and 4% of which extend intracranially, e.g., in the form of cavernous sinus thrombosis, meningitis, or subdural or cerebral empyema [13, 14, 15, 16, 17, 18, 19]. In our study, there was no orbital cellulitis because patients with underlying rhinosinusitis and/or eye diseases were excluded. However, anaerobic cellulitis resulting from orbital tissue hypoperfusion due to the fracture [16], or a COVID-prothrombotic event [1, 15, 17] may be developed in COVID-19 patients.
Weaknesses of this study are a small cohort, and unknown viral shedding dynamics which may decrease with time [7]. False negatives due to SARS-CoV-2 mutations, or poor collection, transport, or handling are also possible [11]. After reviewing the literature, we found that many other studies monotonously focused on facial trauma epidemiology during the pandemic [20, 21, 22, 23, 24]. To the best of our knowledge, this is the first case–control study concerning SARS-CoV-2-OSC in asymptomatic COVID-19 patients with OWF-PNS. Despite meager R-squared, the significant P and high overall accuracy suggest the good predictability of OWF-PNS on SARS-CoV-2-OSC. This research has, anyhow, thrown up many questions in need of further investigations, e.g., re-infection, surgical site infection, and viral transmission. Strict preventive measures should, therefore, be applied until proven otherwise.
Conclusions
This study should be interpreted within the frame of the mentioned weaknesses. The results indicate that asymptomatic COVID-19 patients with any OWF-PNS locations (i.e., orbital floor, medial wall, or both) are almost 21 times more likely to have SARS-CoV-2-OSC than asymptomatic COVID-19 patients without the orbital/facial fracture. Although the case–control ratio and study design were likely to provide statistically relevant datasets, the present findings might not be representative of asymptomatic and/or symptomatic COVID-19 patients in other world regions. A large future study with more “cases” (which may be difficult because surgery for most of orbital fracture patients were postponed until the COVID-19 cures) will be beneficial in verifying or refuting the present findings. However, this work appears to be the very first look at the cause of SARS-CoV-2-OSC. OWF-PNS is the viral source and, thereby, should be manipulated cautiously.
Availability of data and materials
Deidentified individual participant data are not available. The datasets generated and analyzed during this study are available from the first author (P.P.) upon reasonable request.
Abbreviations
- ACE-2-R:
-
Angiotensin-converting enzyme 2 receptors
- 95% CI:
-
95% confidence interval
- COVID-19:
-
Coronavirus disease 2019
- CT:
-
Computed tomography
- FM:
-
Fractured midface
- OWF-PNS:
-
Orbital wall fractures in close proximity to paranasal sinuses
- RT-PCR:
-
Reverse transcription polymerase chain reaction
- SARS-CoV-2:
-
Severe acute respiratory syndrome coronavirus type 2
- SARS-CoV-2-OSC:
-
SARS-CoV-2 ocular surface contamination
References
Pitak-Arnnop P, Meningaud JP, Sirintawat N et al (2022) A German AWMF’s S2e/realist synthesis and meta-narrative snapshot of craniomaxillofacial manifestations in COVID-19 patients: rapid living update on 1 January 2021. J Stomatol Oral Maxillofac Surg 123:64–73. https://doi.org/10.1016/j.jormas.2021.01.012
Pitak-Arnnop P, Sirintawat N, Tangmanee C, Sukphopetch P, Meningaud JP, Neff A (2022) Inanimate surface contamination of SARS-CoV-2 during midfacial fracture repair in asymptomatic COVID-19 patients. J Stomatol Oral Maxillofac Surg. https://doi.org/10.1016/j.jormas.2022.01.006
Pitak-Arnnop P, Tangmanee C, Muangchan C, Meningaud JP, Neff A (2022) Asymptomatic or mildly symptomatic COVID-19 patients with craniomaxillofacial injuries have an increase risk of surgical site infection. Br J Oral Maxillofac Surg. https://doi.org/10.1016/j.bjoms.2022.05.009
Pitak-Arnnop P, Tangmanee C, Meningaud JP, Neff A (2022) Prolonged viral shedding identified from external splints and intranasal packings in immediately cured COVID-19 patients with nasal fractures: a retrospective study. J Stomatol Oral Maxillofac Surg 123:287–291. https://doi.org/10.1016/j.jormas.2022.04.003
Meltzer JA, Kunkov S, Crain EF (2010) Identifying children at low risk for bacterial conjunctivitis. Arch Pediatr Adolesc Med 164:263–267. https://doi.org/10.1001/archpediatrics.2009.289
Atum M, Boz AAE, Çakır B et al (2020) Evaluation of conjunctival swab PCR results in patients with SARS-CoV-2 Iinfection. Ocul Immunol Inflamm 28:745–748. https://doi.org/10.1080/09273948.2020.1775261
Dutescu RM, Banasik P, Schildgen O, Schrage N, Uthoff D (2021) Detection of coronavirus in tear samples of hospitalized patients with confirmed SARS-CoV-2 from oropharyngeal swabs. Cornea 40:348–350. https://doi.org/10.1097/ICO.0000000000002562
Cheong KX (2020) Systematic review of ocular involvement of SARS-CoV-2 in coronavirus disease 2019. Curr Ophthalmol Rep 8:185–194. https://doi.org/10.1007/s40135-020-00257-7
Al-Sharif E, Strianese D, AlMadhi NH et al (2021) Ocular tropism of coronavirus (CoVs): a comparison of the interaction between the animal-to-human transmitted coronaviruses (SARS-CoV-1, SARS-CoV-2, MERS-CoV, CoV-229E, NL63, OC43, HKU1) and the eye. Int Ophthalmol 41:349–362. https://doi.org/10.1007/s10792-020-01575-2
Moreira VM, Mascarenhas P, Machado V et al (2021) Diagnosis of SARS-Cov-2 infection by RT-PCR using specimens other than naso- and oropharyngeal swabs: a systematic review and meta-analysis. Diagnostics (Basel) 11:363. https://doi.org/10.3390/diagnostics11020363
Sopp NM, Sharda V (2021) An eye on COVID-19: a meta-analysis of positive conjunctival reverse transcriptase-polymerase chain reaction and SARS-CoV-2 conjunctivitis prevalence. Optom Vis Sci 98:429–436. https://doi.org/10.1097/OPX.0000000000001687
de Freitas SD, de Sousa LB, Câmara NOS, de Freitas D, de Oliveira LA (2021) SARS-COV-2 and ocular surface: from physiology to pathology, a route to understand transmission and disease. Front Physiol 12:612319. https://doi.org/10.3389/fphys.2021.612319
Bachelet JT, Landis BN, Scolozzi P (2019) Recurrent maxillary sinusitis and periorbital cellulitis revealing an unnoticed medial wall orbital fracture. J Craniofac Surg 30:2251–2252. https://doi.org/10.1097/SCS.0000000000005985
Byeon JY, Choi HJ (2017) Orbital cellulitis following orbital blow-out fracture. J Craniofac Surg 28:1777–1779. https://doi.org/10.1097/SCS.0000000000003732
Ben Simon GJ, Bush S, Selva D, McNab AA (2005) Orbital cellulitis: a rare complication after orbital blowout fracture. Ophthalmology 112:2030–2034. https://doi.org/10.1016/j.ophtha.2005.06.012
Hada M, Gupta P, Bagarhatta M et al (2022) Orbital magnetic resonance imaging profile and clinicoradiological correlation in COVID-19-associated rhino-orbital-cerebral mucormycosis: a single-center study of 270 patients from North India. Indian J Ophthalmol 70:641–648. https://doi.org/10.4103/ijo.IJO_1652_21
Heywood EG, Ul-Haq I, Pankhania M (2022) COVID-19-related orbital abscesses: a case report. Ann R Coll Surg Engl 104(6):e193–e195. https://doi.org/10.1308/rcsann.2021.0306
Takesue A, Asada Y, Ooya H, Yokoyama T (2021) Blowout fracture-associated orbital cellulitis progressing to panophthalmitis: a case report. BMC Ophthalmol 21:390. https://doi.org/10.1186/s12886-021-02153-5
Tu Y, Hong H, Wu W (2021) Orbital infection due to medial wall fracture: three cases of orbital complications caused by paranasal sinusitis secondary to medial orbital wall fracture. J Craniofac Surg 32:e712–e716. https://doi.org/10.1097/SCS.0000000000007714
Haapanen A, Furuholm J, Uittamo J, Snäll J (2022) Effect of social distancing during the COVID-19 pandemic on the occurrence of maxillofacial fractures in a Finnish tertiary trauma centre. Acta Odontol Scand 80:157–160. https://doi.org/10.1080/00016357.2021.1979643
Philip G, Dominic S, Poorna TA, Ek J (2022) Pattern of maxillofacial fractures in a tertiary referral centre in Central Kerala: a comparison between the Pre-COVID and COVID periods. J Oral Biol Craniofac Res 12:45–48. https://doi.org/10.1016/j.jobcr.2021.09.021
Canzi G, De Ponti E, Corradi F et al (2021) Epidemiology of maxillo-facial trauma during COVID-19 lockdown: reports from the hub trauma center in Milan. Craniomaxillofac Trauma Reconstr 14:277–283. https://doi.org/10.1177/1943387520983119
Visholm TM, Sandhu N, Dhariwal DK (2021) COVID-19: the oral and maxillofacial surgery experience, Oxford. UK Craniomaxillofac Trauma Reconstr 14:317–324. https://doi.org/10.1177/1943387521991267
Nhongo SS, Sklavos A, Lee K, Chan STF, Austin S (2020) The changing face of maxillofacial trauma during the COVID-19 lockdowns in Melbourne. Australia Oral Maxillofac Surg (in press). https://doi.org/10.1007/s10006-022-01041-6
Funding
Open Access funding enabled and organized by Projekt DEAL. The authors received no financial support for the research, authorship, and/or publication of this article.
Author information
Authors and Affiliations
Contributions
PP, CT contributed to acquisition of (blinded) data. PP, CT, J-PM, AN were involved in conception and study design, statistical analysis and interpretation, drafting the work, and final approval and agreement of all aspects of the work.
Corresponding author
Ethics declarations
Conflict of interest
Prof. Jean-Paul Meningaud is the Immediate Past President of the European Association for Cranio-Maxillo-Facial Surgery (EACMFS). Prof. Andreas Neff is the Head of the TMJ Section of the Strasbourg Osteosynthesis Research Group (S.O.R.G) and the Immediate Past President of the European Society of TMJ Surgeons (ESTMJS), as well as has received remunerations as a design surgeon for Medartis (Basel, Switzerland) for the development of midfacial and mandibular osteosynthesis systems. All of the authors indicate full freedom of investigation and manuscript preparation without potential conflict of interest as regards this study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Pitak-Arnnop, P., Tangmanee, C., Meningaud, JP. et al. Is orbital wall fracture associated with SARS-CoV-2 ocular surface contamination in asymptomatic COVID-19 patients?. Int Ophthalmol 43, 1369–1374 (2023). https://doi.org/10.1007/s10792-022-02535-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10792-022-02535-8