Abstract
Purpose
To characterize glaucoma-induced damage following injections of plastic microbeads into the anterior chamber of mice.
Methods
Mice were divided into three groups: a single plastic microbeads injection (n = 21); two consecutive plastic microbead injections to the right eye at 1-week intervals, 4 of which with two consecutive saline injections in the left eye (n = 15); and an additional control group of two consecutive saline injections at 1-week intervals (n = 6). Intraocular pressure (IOP) was measured weekly. Retinal thickness, ganglion cells (RGCs) and axonal loss, inflammatory and gliosis reactions were measured at week four. Molecular analysis using qRT-PCR in the microbeads injection groups focused on expression levels of inflammation and glaucoma-related genes.
Results
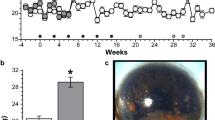
Mean IOP following single injection at 4 weeks was significantly elevated compared to baseline in injected eyes (14.5 ± 3.3 mmHg vs. 11.1 ± 2.5 mmHg, respectively, p = 0.003) and not in fellow eyes (13.2 ± 2.9 mmHg vs. 12.2 ± 2.9, respectively, NS). Six (35.3%) bead-injected eyes had IOP ≥ 17 mmHg compared with 2 (11.8%) saline-injected control eyes. Retinal thickness in injected and fellow eyes was 193.7 ± 15.5 µm and 223.9 ± 15.5 µm, respectively (p = 0.03). RGC loss in injected and fellow eyes was 16.0 ± 0.5 and 17.6 ± 0.7 cells per 200 µm, respectively (p = 0.005). Retinal gliosis, axonal loss and inflammatory cell infiltration to the bead-injected eyes were noted. Molecular analysis following double injection showed STAT3 expression decreased in the glaucoma-induced optic nerves (0.69 ± 0.3 vs. 1.16 ± 0.3, p = 0.04), but increased in the glaucoma-induced retinae (p = 0.05) versus saline; retinal IL-1β decreased significantly (0.04 ± 0.04 vs. 0.36 ± 0.2, p = 0.02). TNF-α, NFkB and SOD-1 expression did not change.
Conclusion
One/two injections of microbeads elevated IOP, with measurable neuronal damage. An inflammatory response was detected in the injured retina and optic nerve. The therapeutic significance of these findings should be explored.






Similar content being viewed by others
References
Tham YC, Li X, Wong TY, Quigley HA, Aung T, Cheng CY (2014) Global prevalence of glaucoma and projections of glaucoma burden through 2040: a systematic review and meta-analysis. Ophthalmology 121:2081–2090. https://doi.org/10.1016/j.ophtha.2014.05.013
Kingman S (2004) Glaucoma is second leading cause of blindness globally. Bull World Health Organ 82:887–888
Gordon MO, Beiser JA, Brandt JD et al (2002) The ocular hypertension treatment study: baseline factors that predict the onset of primary open-angle glaucoma. Arch Ophthalmol 120:714–720. https://doi.org/10.1001/archopht.120.6.714
Lichter PR, Musch DC, Gillespie BW et al; CIGTS Study Group (2001) Interim clinical outcomes in the collaborative initial glaucoma treatment study comparing initial treatment randomized to medications or surgery. Ophthalmology 108:1943‒1953. https://doi.org/10.1016/s0161-6420(01)00873-9
Zeng HL, Shi JM (2018) The role of microglia in the progression of glaucomatous neurodegeneration- a review. Int J Ophthalmol 11:143–149
Vohra R, Tsai JC, Kolko M (2013) The role of inflammation in the pathogenesis of glaucoma. Surv Ophthalmol 58:311–320. https://doi.org/10.1016/j.survophthal.2012.08.010
Ebneter A, Casson RJ, Wood JPM, Chidlow G (2010) Microglial activation in the visual pathway in experimental glaucoma: spatiotemporal characterization and correlation with axonal injury. Invest Ophthalmol Vis Sci 51:6448–6460. https://doi.org/10.1167/iovs.10-5284
Aires ID, Ambrósio AF, Santiago AR (2017) Modeling human glaucoma: lessons from the in vitro models. Ophthalmic Res 57:77–86. https://doi.org/10.1159/000448480
Savinova OV, Sugiyama F, Martin JE et al (2001) Intraocular pressure in genetically distinct mice: an update and strain survey. BMC Genet 2:12. https://doi.org/10.1186/1471-2156-2-12
Thanos S, Naskar R (2004) Correlation between retinal ganglion cell death and chronically developing inherited glaucoma in a new rat mutant. Exp Eye Res 79:119–129. https://doi.org/10.1016/j.exer.2004.02.005
Kuchtey J, Olson LM, Rinkoski T et al (2011) Mapping of the disease locus and identification of ADAMTS10 as a candidate gene in a canine model of primary open angle glaucoma. PLoS Genet 7:e1001306. https://doi.org/10.1371/journal.pgen.1001306
Snyder KC, Oikawa K, Williams J et al (2019) Imaging distal aqueous outflow pathways in a spontaneous model of congenital glaucoma. Transl Vis Sci Technol 8:22. https://doi.org/10.1167/tvst.8.5.22
Dawson WW, Brooks DE, Hope GM et al (1993) Primary open angle glaucomas in the rhesus monkey. Br J Ophthalmol 77:302–310. https://doi.org/10.1136/bjo.77.5.302
Cone FE, Gelman SE, Son JL, Pease ME, Quigley HA (2010) Differential susceptibility to experimental glaucoma among 3 mouse strains using bead and viscoelastic injection. Exp Eye Res 91:415–424. https://doi.org/10.1016/j.exer.2010.06.018
Johnson TV, Tomarev SI (2010) Rodent models of glaucoma. Brain Res Bull 81:349–358. https://doi.org/10.1016/j.brainresbull.2009.04.004
Goldenberg-Cohen N, Dadon-Bar-El S, Hasanreisoglu M et al (2009) Possible neuroprotective effect of brimonidine in a mouse model of ischaemic optic neuropathy. Clin Exp Ophthalmol 37:718–729. https://doi.org/10.1111/j.1442-9071.2009.02108.x
Goldenberg-Cohen N, Dratviman-Storobinsky O, El Dadon Bar S, Cheporko Y, Hochhauser E (2011) Protective effect of Bax ablation against cell loss in the retinal ganglion layer induced by optic nerve crush in transgenic mice. J Neuroophthalmol 31:331–338. https://doi.org/10.1097/WNO.0b013e318227e4fb
Adachi M, Takahashi K, Nishikawa M, Miki H, Uyama M (1996) High intraocular pressure-induced ischemia and reperfusion injury in the optic nerve and retina in rats. Graefe’s Arch Clin Exp Ophthalmol 234:445–451. https://doi.org/10.1007/BF02539411
Büchi ER (1992) Cell death in the rat retina after a pressure-induced ischaemia-reperfusion insult: an electron microscopic study. I. Ganglion cell layer and inner nuclear layer. Exp Eye Res 55:605–613. https://doi.org/10.1016/s0014-4835(05)80173-3
Wooff Y, Man SM, Aggio-Bruce R, Natoli R, Fernando N (2019) IL-1 family members mediate cell death, inflammation and angiogenesis in retinal degenerative diseases. Front Immunol 10:1618. https://doi.org/10.3389/fimmu.2019.01618
Markiewicz L, Pytel D, Mucha B et al (2015) Altered expression levels of MMP1, MMP9, MMP12, TIMP1, and IL-1β as a risk factor for the elevated IOP and optic nerve head damage in the primary open-angle glaucoma patients. Biomed Res Int 2015:812503. https://doi.org/10.1155/2015/812503
Wang N, Chintala SK, Fini ME, Schuman JS (2001) Activation of a tissue-specific stress response in the aqueous outflow pathway of the eye defines the glaucoma disease phenotype. Nat Med 7:304–309. https://doi.org/10.1038/85446
Benitez-Del-Castillo J, Cantu-Dibildox J, Sanz-González SM, Zanón-Moreno V, Pinazo-duran MD (2019) Cytokine expression in tears of patients with glaucoma or dry eye disease: a prospective, observational cohort study. Eur J Ophthalmol 29:437–443. https://doi.org/10.1177/1120672118795399
Chi W, Li F, Chen H et al (2014) Caspase-8 promotes NLRP1/NLRP3 inflammasome activation and IL-1β production in acute glaucoma. Proc Natl Acad Sci USA 111:11181–11186. https://doi.org/10.1073/pnas.1402819111
Mookherjee S, Banerjee D, Chakraborty S, Banerjee A, Mukhopadhyay I, Sen A, Ray K (2010) Association of IL1A and IL1B loci with primary open angle glaucoma. BMC Med Genet 11:99. https://doi.org/10.1186/1471-2350-11-99
Chua J, Vania M, Cheung CM, Ang M, Chee SP, Yang H, Li J, Wong TT (2012) Expression profile of inflammatory cytokines in aqueous from glaucomatous eyes. Mol Vis 18:431–438
Bareyre FM, Garzorz N, Lang C, Misgeld T, Büning H, Kerschensteiner M (2011) In vivo imaging reveals a phase-specific role of STAT3 during central and peripheral nervous system axon regeneration. Proc Natl Acad Sci U S A 108:6282–6287. https://doi.org/10.1073/pnas.1015239108
Luo X, Ribeiro M, Bray ER et al (2016) Enhanced transcriptional activity and mitochondrial localization of STAT3 Co-induce axon regrowth in the adult central nervous system. Cell Rep 15:398–410. https://doi.org/10.1016/j.celrep.2016.03.029
Sun D, Moore S, Jakobs TC (2017) Optic nerve astrocyte reactivity protects function in experimental glaucoma and other nerve injuries. J Exp Med 214:1411–1430. https://doi.org/10.1084/jem.20160412
Wang DY, Ray A, Rodgers K, Ergorul C, Hyman BT, Huang W, Grosskreutz CL (2010) Global gene expression changes in rat retinal ganglion cells in experimental glaucoma. I Invest Ophthalmol Vis Sci 51:4084–4095. https://doi.org/10.1167/IOVS.09-4864
Zhang S, Li W, Wang W, Zhang SS, Huang P, Zhang C (2013) Expression and activation of STAT3 in the astrocytes of optic nerve in a rat model of transient intraocular hypertension. PLoS ONE 8:e55683. https://doi.org/10.1371/journal.pone.0055683
Goebel U, Scheid S, Spassov S et al (2021) Argon reduces microglial activation and inflammatory cytokine expression in retinal ischemia/reperfusion injury. Neural Regen Res 16:192–198. https://doi.org/10.4103/1673-5374.290098
Nicou CM, Pillai A, Passaglia CL (2021) Effects of acute stress, general anesthetics, tonometry, and temperature on intraocular pressure in rats. Exp Eye Res 210:108727. https://doi.org/10.1016/J.EXER.2021.108727
Chae JJ, Prausnitz MR, Ethier CR (2021) Effects of general anesthesia on intraocular pressure in rabbits. J Am Assoc Lab Anim Sci 60:91–95
Jasien JV, Girkin CA, Downs JC (2019) Effect of anesthesia on intraocular pressure measured with continuous wireless telemetry in non-human primates. Invest Ophthalmol Vis Sci 60:3830–3834. https://doi.org/10.1167/IOVS.19-27758
Funding
This study was partially supported in part by the Zanvyl and Isabelle Krieger Fund, Baltimore, MD (N.G.-C.). The funding organization had no role in the design or conduct of this research.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Ethical approval
All protocols were conducted in accordance with the Association for Research in Vision and Ophthalmology (ARVO) Statement for the Use of Animals in Ophthalmic and Vision Research and National Institutes of Health guidelines, and were approved and monitored by the Institutional Animal Research Committee.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Presented in part at Israeli Society of Vision and Eye Research (ISVER) 2017, The Association for Research in Vision and Ophthalmology (ARVO), Baltimore, MD, June 2017.
Rights and permissions
About this article
Cite this article
Zahavi, A., Friedman Gohas, M., Sternfeld, A. et al. Histological and molecular characterization of glaucoma model induced by one or two injections of microbeads to the anterior chamber of mice. Int Ophthalmol 42, 3763–3775 (2022). https://doi.org/10.1007/s10792-022-02372-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10792-022-02372-9