Abstract
Purpose
To present the efficacy and safety of surgical treatment of moderate to advanced stage of open-angle glaucoma using the surgical method of transscleral cyclophotocoagulation with MicroPulse® laser (Iridex, Silicon Valley, California, USA) and to compare these results with those of continuous transscleral cyclophotocoagulation with diode laser.
Methods
In a prospective observational clinical study 22 patients (30 eyes) with moderate and advanced open-angle glaucoma underwent transscleral cyclophotocoagulation. 15 eyes were treated with transscleral cyclophotocoagulation with MicroPulse® laser with 2500 mW (group A) and 15 eyes with continuous diode laser 810 nm and power between 2000 and 2300 mW (group B). The follow-up period of the study was 12 months.
Results
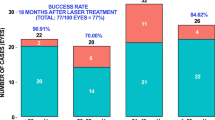
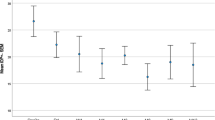
A reduction of the intraocular pressure > 30% was achieved in 53.3% of the group A and in 60% of the group B during the follow-up period of 12 months. A statistically significant decrease of the number of antiglaucoma agents was observed in group A. One eye (6.6%) of group A underwent additional antiglaucoma procedures to achieve target intraocular pressure. The corresponding percentage is 40% in group B. The postoperative inflammatory response was significantly more limited in group A compared to group B.
Conclusions
The transscleral cyclophotocoagulation with MicroPulse® diode laser with 2500 mW achieves a similar decrease of the intraocular pressure as cyclophotocoagulation with continuous diode laser for the first postoperative months, but more satisfactory reduction of the number of eye drops as well as more limited postoperative inflammatory response.





Similar content being viewed by others
Availability of data
All data generated or analyzed during this study are included in this published article.
Abbreviations
- AC:
-
Anterior chamber
- BCVA:
-
Best corrected visual acuity
- CCLRU:
-
Cornea and contact lens research unit
- cTS-CPC:
-
Continuous transscleral cyclophotocoagulation
- DMEK:
-
Descemet membrane endothelial keratoplasty
- ECD:
-
Corneal endothelial cell density
- GAT:
-
Goldmann applanation tonometry
- IOP:
-
Intraocular pressure
- MIGS:
-
Micro-invasive glaucoma surgery
- mTS-CPC:
-
MicroPulse® transscleral cyclophotocoagulation
- NRS:
-
Numeric rating scale
- OAG:
-
Open-angle glaucoma
- PEX:
-
Pseudoexfoliation
- POAG:
-
Primary open-angle glaucoma
References
Quigley HA, Broman AT (2006) The number of people with glaucoma worldwide in 2010 and 2020. Br J Ophthalmol 3:262–267
Weinreb RN, Khaw PT (2004) Primary open angle glaucoma. The Lancet 363:1711–1720
Gedde SJ, Schiffman JC, Feuer WJ, Herndon LW, Brandt JD, Budenz DL, Tube versus Trabeculectomy Study Group (2012) Treatment outcomes in the tube versustrabeculectomy (TVT) study after five years of follow-up. Am J Ophthalmol 153:789–803
Boland MV, Ervin A-M, Friedman DS, Jampel HD, Hawkins BS, Vollenweider D, Chelladurai Y, Ward D, Suarez-Cuervo C, Robinson KA (2013) Comparative effectiveness of treatments for open-angle glaucoma: a systematic review for the US Preventive Services Task Force. Ann Intern Med 158:271–279
Gavris MM, Olteanu I, Kantor E, Mateescu R, Belicioiu R (2017) IRIDEX MicroPulse® P3: innovative cyclophotocoagulation. Rom J Ophthalmol 61:107–111
Liu GJ, Mizukawa A, Okisaka S (1994) Mechanism of intraocular pressure decrease after contact transscleral continuous-waveNd:YAG laser cyclophotocoagulation. Ophthalmic Res 26:65–79
Sanchez FG, Peirano-Bonomi JC, Grippo TM (2018) MicroPulse® Transscleral Cyclophotocoagulation: A Hypothesis for the Ideal Parameters. Med Hypothesis Discov Innov Ophthalmol 7:94–100
Sarrafpour S, Saleh D, AyoubS, Radcliffe NM (2019) MicroPulse® Transscleral Cyclophotocoagulation: a look at long-term effectiveness and outcomes ophthalmology glaucoma 2:167–171
Yelenskiy A, Gillette TB, Arosemena A, Stern AG, Garris WJ, Young CT, Hoyt M, Worley N, Zurakowski D, Ayyala RS (2018) Patient Outcomes Following MicroPulse® Transscleral Cyclophotocoagulation: intermediate-term results. J Glaucoma 27:920–925
Varikuti VNV, Shah P, Rai O, Chaves AC, Miranda A, Lim BA, Dorairaj SK, Sieminski SF (2019) Outcomes of MicroPulse® Transscleral Cyclophotocoagulation in eyes with good central vision. J Glaucoma 28:901–905
Jabs DA1, Nussenblatt RB, Rosenbaum JT; Standardization of Uveitis Nomenclature (SUN) Working Group (2005) Standardization of uveitis nomenclature for reporting clinical data. Results of the First International Workshop. Am J Ophthalmol 140:509–516
Murphy PJ1, Lau JS, Sim MM, Woods RL, (2007) How red is a white eye? Clinical grading of normal conjunctival hyperaemia. Eye 21:633–638
Jensen MP, McFarland CA (1993) Increasing the reliability and validity of pain intensity measurement in chronic pain patients. Pain 55:195–203
Aquino MC, Barton K, Tan AM, Sng C, Li X, Loon SC, Chew PT (2015) MicroPulse® versus continuous wave transscleral diode cyclophotocoagulation in refractory glaucoma: a randomized exploratory study. Clin Exp Ophthalmol 43:40–46
Tan AM, Chockalingam M, Aquino MCD, Lim ZIL, See JLS, Chew PTK (2010) MicroPulse® trans-scleral cyclophotocoagulation in the treatment of refractory glaucoma. Clin Experiment Ophthalmol 38:266–272
Kuchar S, Moster MR, Reamer CB, Waisbourd M (2016) Treatmentoutcomes of MicroPulse® transscleral cyclophotocoagulationin advanced glaucoma. Lasers Med Sci 31:393–396
Gedde SJ, Schiffman JC, Feuer WJ, Herndon LW, Brandt JD, Budenz DL (2007) Treatment outcomes in the tube versus trabeculectomy study after one year of follow-up. Am J Ophthalmol 43:9–22
Emanuel ME, Grover DS, Fellman RL, Godfrey DG, Smith O, Butler MR, Kornmann HL, Feuer WJ, Goyal S (2017) MicroPulse® cyclophotocoagulation: initial results in refractory glaucoma. J Glaucoma 26:726–729
Williams AL, Moster MR, Rahmatnejad K, Resende AF, Horan T, Reynolds M, Yung E, Abramowitz B, Kuchar S, Waisbourd M (2018) clinical efficacy and safety profile of MicroPulse® Transscleral cyclophotocoagulation in refractory glaucoma. J Glaucoma 27:445–449
Bendel RE, Patterson MT (2017) Observational report: improved outcomes of transscleral cyclophotocoagulation for glaucoma patients. Medicine 96:e6946
Acknowledgements
There is no support, including pharmacological and industry support.
Funding
The authors have no relevant financial or non-financial interests to disclose. No funding was received for conducting this study.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Sofia Fili, Iraklis Vastardis, Georgios Perdikakis and Markus Kohlhaas. The first draft of the manuscript was written by Sofia Fili and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that there is no conflict of interest. All authors certify that they have no affiliations with or involvement in any organization or entity with any financial interest (such as honoraria; educational grants; participation in speakers' bureaus; membership, employment, consultancies, stock ownership, or other equity interest; and expert testimony or patent-licensing arrangements), or non-financial interest (such as personal or professional relationships, affiliations, knowledge or beliefs) in the subject matter or materials discussed in this manuscript.
Ethics approval
All procedures performed in this observatory study involving human participants were in accordance with the ethical standards of the institutional research committee of St-Johannes-Hospital in Dortmund, Germany and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Clinical trial registration
This is an observatory study and no clinical trial registration is necessary.
Consent to participate
Informed consent for participation was obtained from all individual participants included in the study.
Consent to publish
Informed consent for publication was obtained from all individual participants included in the study.
Animal research
No research is performed on animals in this study.
Plant reproducibility
No research is performed on animals in this study.
Image manipulation
No image manipulation is performed in this study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Fili, S., Vastardis, I., Perdikakis, G. et al. Transscleral cyclophotocoagulation with MicroPulse® laser versus cyclophotocoagulation with continuous diode laser in patients with open-angle glaucoma. Int Ophthalmol 42, 525–539 (2022). https://doi.org/10.1007/s10792-021-02023-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10792-021-02023-5