Abstract
Purpose
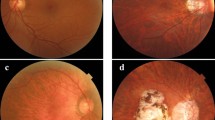
To predict exudative age-related macular degeneration (AMD), we combined a deep convolutional neural network (DCNN), a machine-learning algorithm, with Optos, an ultra-wide-field fundus imaging system.
Methods
First, to evaluate the diagnostic accuracy of DCNN, 364 photographic images (AMD: 137) were amplified and the area under the curve (AUC), sensitivity and specificity were examined. Furthermore, in order to compare the diagnostic abilities between DCNN and six ophthalmologists, we prepared yield 84 sheets comprising 50% of normal and wet-AMD data each, and calculated the correct answer rate, specificity, sensitivity, and response times.
Results
DCNN exhibited 100% sensitivity and 97.31% specificity for wet-AMD images, with an average AUC of 99.76%. Moreover, comparing the diagnostic abilities of DCNN versus six ophthalmologists, the average accuracy of the DCNN was 100%. On the other hand, the accuracy of ophthalmologists, determined only by Optos images without a fundus examination, was 81.9%.
Conclusion
A combination of DCNN with Optos images is not better than a medical examination; however, it can identify exudative AMD with a high level of accuracy. Our system is considered useful for screening and telemedicine.




Similar content being viewed by others
References
Lim LS, Mitchell P, Seddon JM, Holz FG, Wong TY (2012) Age-related macular degeneration. Lancet 379:1728–1738
Yasuda M, Kiyohara Y, Hata Y, Arakawa S, Yonemoto K, Doi Y, Iida M, Ishibashi T (2009) Nine-year incidence and risk factors for age-related macular degeneration in a defined Japanese population the Hisayama study. Ophthalmology 116:2135–2140
Rosenfeld PJ, Brown DM, Heier JS, Boyer DS, Kaiser PK, Chung CY, Kim RY, MARINA Study Group (2006) Ranibizumab for neovascular age-related macular degeneration. N Engl J Med 355:1419–1431
Brown DM, Michels M, Kaiser PK, Heier JS, Sy JP, Ianchulev T, ANCHOR Study Group (2009) Ranibizumab versus verteporfin photodynamic therapy for neovascular age-related macular degeneration: two-year results of the ANCHOR study. Ophthalmology 116:57–65
Comparison of Age-related Macular Degeneration Treatments Trials (CATT) Research Group, Maguire MG, Martin DF, Ying GS, Jaffe GJ, Daniel E, Grunwald JE, Toth CA, Ferris FL, Fine SL (2016) Five-year outcomes with anti-vascular endothelial growth factor treatment of neovascular age-related macular degeneration. Ophthalmology 123(8):1751–1761
Rofagha S, Bhisitkul RB, Boyer DS, Sadda SR, Zhang K, SEVEN-UP Study Group (2013) Seven-year outcomes in ranibizumab-treated patients in ANCHOR, MARINA, and HORIZON: a multicenter cohort study (SEVEN-UP). Ophthalmology 120(11):2292–2299
Rasmussen A, Bloch SB, Fuchs J, Hansen LH, Larsen M, Lacour M, Lund-Andersen H, Sander B (2013) A 4-year longitudinal study of 555 patients treated with ranibizumab for neovascular age-related macular degeneration. Ophthalmology 120:2630–2636
Witmer MT, Parlitsis G, Patel S, Kiss S (2013) Comparison of ultra-widefield fluorescein angiography with the Heidelberg Sp ectralis(®) noncontact ultra-widefield module versus the Optos(®) Optomap(®). Clin Ophthalmol 7:389–394
Nagiel A, Lalane RA, Sadda SR, Schwartz SD (2016) Ultra-widefield fundus imaging: a review of clinical applications and future trends. Retina 36:660–678
Ghasemi Falavarjani K, Tsui I, Sadda SR (2017) Ultra-wide-field imaging in diabetic retinopathy. Vis Res S0042–6989(17):30118–30119
Soliman AZ, Silva PS, Aiello LP, Sun JK (2012) Ultra-wide field retinal imaging in detection, classification, and management of diabetic retinopathy. Semin Ophthalmol 27(5–6):221–227
Witmer MT, Cho M, Favarone G, Chan RV, D’Amico DJ, Kiss S (2012) Ultra-wide-field autofluorescence imaging in non-traumatic rhegmatogenous retinal detachment. Eye (Lond) 26(9):1209–1216
LeCun Y, Bengio Y, Hinton G (2015) Deep learning. Nature 521:436–444
Liu S, Liu S, Cai W, Che H, Pujol S, Kikinis R, Feng D, Fulham MJ, ADNI (2015) Multimodal neuroimaging feature learning for multiclass diagnosis of Alzheimer’s disease. IEEE Trans Biomed Eng 62:1132–1140
Litjens G, Sánchez CI, Timofeeva N, Hermsen M, Nagtegaal I, Kovacs I, Hulsbergen-van de Kaa C, Bult P, van Ginneken B, van der Laak J (2016) Deep learning as a tool for increased accuracy and efficiency of histopathological diagnosis. Sci Rep 6:26286
Gulshan V, Peng L, Coram M, Stumpe MC, Wu D, Narayanaswamy A, Venugopalan S, Widner K, Madams T, Cuadros J, Kim R, Raman R, Nelson PC, Mega JL, Webster DR (2016) Development and validation of a deep learning algorithm for detection of diabetic retinopathy in retinal fundus photographs. JAMA 316:2402–2410
Pinaya WH, Gadelha A, Doyle OM, Noto C, Zugman A, Cordeiro Q, Jackowski AP, Bressan RA, Sato JR (2016) Using deep belief network modelling to characterize differences in brain morphometry in schizophrenia. Sci Rep 6:38897
Burlina P, Pacheco KD, Joshi N, Freund DE, Bressler NM (2017) Comparing humans and deep learning performance for grading AMD: a study in using universal deep features and transfer learning for automated AMD analysis. Comput Biol Med 82:80–86
Mrsnik M (2013) Global aging 2013: rising to the challenge. Standard and poor’s rating services network. https://www.nact.org/resources/2013_NACT_Global_Aging.pdf
Ohsugi H, Tabuchi H, Enno H, Ishitobi N (2017) Accuracy of deep learning, a machine-learning technology, using ultra-wide-field fundus ophthalmoscopy for detecting rhegmatogenous retinal detachment. Sci Rep 7:9425
Glorot X, Bordes A, Bengio Y (2011) Deep sparse rectifier neural networks. In: The 14th international conference on artificial intelligence and statistics, pp 315–323
Ioffe S, Szegedy C (2015) Batch normalization: accelerating deep network training by reducing internal covariate shift. In: International conference on machine learning
Srivastava N, Hinton G, Krizhevsky A, Sutskever I, Salakhutdinov R (2014) Dropout: a simple way to prevent neural networks from overfitting. J Mach Learn Res 15:1929–1958
Qian N (1999) On the momentum term in gradient descent learning algorithms. Neural Netw 12(1):145–151
Nesterov Y (1983) A method for unconstrained convex minimization problem with the rate of convergence O (1/k^ 2). Doklady AN USSR. 269
Schisterman EF, Faraggi D, Reiser B, Hu J (2008) Youden index and the optimal threshold for markers with mass at zero. Stat Med 27(2):297–315
Selvaraju RR, Cogswell M, Das A, Vedantam R, Parikh D, Batra D (2016) Grad-CAM: visual explanations from deep networks via gradient-based localization. https://arxiv.org/abs/1610.02391v3
Deng J, Dong W, Socher R, Li LJ, Li K, Li FF (2009) Imagenet: a large-scale hierarchical image database. In: Computer vision and pattern recognition, pp 248–255
Russakovsky O, Deng J, Su H, Krause J, Satheesh S, Ma S, Huang Z, Karpathy A, Khosla A, Bernstein M, Berg AC, Li FF (2014) Imagenet large scale visual recognition challenge. arXiv preprint arXiv:1409.0575
Lee CY, Xie S, Gallagher P, Zhang Z, Tu Z (2016) Deeply-supervised nets. arXiv preprint arXiv:1409.5185 (2014)
Gerding H (2016) Long-term results of intravitreal Anti-VEGF injections in wet AMD: a meta-analysis. Klin Monbl Augenheilkd 233:471–474
Gordois A, Cutler H, Pezzullo L, Gordon K, Cruess A, Winyard S, Hamilton W, Chua K (2012) An estimation of the worldwide economic and health burden of visual impairment. Glob Public Health 7(5):465–481
Acknowledgements
The authors thank Masayuki Miki and orthoptists of Tsukazaki Hospital for support in data collection.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Matsuba, S., Tabuchi, H., Ohsugi, H. et al. Accuracy of ultra-wide-field fundus ophthalmoscopy-assisted deep learning, a machine-learning technology, for detecting age-related macular degeneration. Int Ophthalmol 39, 1269–1275 (2019). https://doi.org/10.1007/s10792-018-0940-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10792-018-0940-0