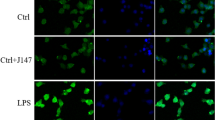
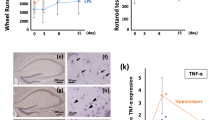
Abstract
It has been proven that neuroinflammation triggered by microglial activation is the pathogenesis of depression associated with sepsis. An endogenous lipid mediator known as resolvin D1 (RvD1) is known to have anti-inflammatory effects in a sepsis model. However, it remains unknown if the effects of RvD1 on inflammatory responses are regulated by microglial autophagy. The current study investigated the role of RvD1-induced microglial autophagy in neuroinflammation. The findings showed that RvD1 reverses LPS-induced autophagy inhibition in microglia. RvD1 treatment significantly inhibits inflammatory responses by preventing NF-κB nuclear translocation and microglial M1 phenotypic transition. RvD1 exhibits an attenuation of neurotoxicity in both in vivo and in vitro models of sepsis. Following RvD1 injection, depressive-like behaviors in SAE mice were significantly improved. Notably, the aforesaid effects of RvD1 were eliminated by 3-MA, demonstrating that microglial autophagy was modulated. In conclusion, our findings shed new light on the involvement of microglial autophagy in SAE and emphasize the potential benefits of RvD1 as a promising therapeutic agent in the treatment of depression.








Similar content being viewed by others
Data availability
All the raw data of this study are available from the corresponding author, Zhongliang Dai, upon reasonable request.
Abbreviations
- BECN1:
-
Beclin1
- Casp.3:
-
Cleaved-caspase3
- C-PARP:
-
Cleaved poly ADP-ribose polymerase
- FST:
-
Forced swim test
- LPS:
-
Lipopolysaccharides
- NF-κB:
-
Nuclear factor-kappa-B
- OFT:
-
Open field test
- RvD1:
-
Resolvin D1
- SAE:
-
Sepsis-associated encephalopathy
- TST:
-
Tail suspension test
- TEM:
-
Transmission electron microscopy
References
Ali T, Rahman SU, Hao Q, Li W, Liu Z, Ali Shah F, Murtaza I, Zhang Z, Yang X, Liu G, Li S (2020) Melatonin prevents neuroinflammation and relieves depression by attenuating autophagy impairment through FOXO3a regulation. J Pineal Res 69:e12667. https://doi.org/10.1111/jpi.12667
Bisicchia E, Sasso V, Catanzaro G, Leuti A, Besharat ZM, Chiacchiarini M, Molinari M, Ferretti E, Viscomi MT, Chiurchiù V (2018) Resolvin D1 halts remote neuroinflammation and improves functional recovery after focal brain damage Via ALX/FPR2 receptor-regulated microRNAs. Mol Neurobiol 55:6894–6905. https://doi.org/10.1007/s12035-018-0889-z
Catarina AV, Branchini G, Bettoni L, De Oliveira JR, Nunes FB (2021) Sepsis-associated encephalopathy: from pathophysiology to progress in experimental studies. Mol Neurobiol 58:2770–2779. https://doi.org/10.1007/s12035-021-02303-2
Choi Y, Bowman JW, Jung JU (2018) Autophagy during viral infection - a double-edged sword. Nat Rev Microbiol 16:341–354. https://doi.org/10.1038/s41579-018-0003-6
Deretic V (2021) Autophagy in inflammation, infection, and immunometabolism. Immunity 54:437–453. https://doi.org/10.1016/j.immuni.2021.01.018
Fan Z, Ma H, Li Y, Wu Y, Wang J, Xiong L, Fang Z, Zhang X (2022) Neuronal MD2 induces long-term mental impairments in septic mice by facilitating necroptosis and apoptosis. Front Pharmacol 13:884821. https://doi.org/10.3389/fphar.2022.884821
Han H-E, Kim T-K, Son H-J, Park WJ, Han P-L (2013) Activation of autophagy pathway suppresses the expression of iNOS, IL6 and cell death of lps-stimulated microglia cells. Biomol Ther (seoul) 21:21–28. https://doi.org/10.4062/biomolther.2012.089
He Y, She H, Zhang T, Xu H, Cheng L, Yepes M, Zhao Y, Mao Z (2018) p38 MAPK inhibits autophagy and promotes microglial inflammatory responses by phosphorylating ULK1. J Cell Biol 217:315–328. https://doi.org/10.1083/jcb.201701049
Hu X, Leak RK, Shi Y, Suenaga J, Gao Y, Zheng P, Chen J (2015) Microglial and macrophage polarization—new prospects for brain repair. Nat Rev Neurol 11:56–64. https://doi.org/10.1038/nrneurol.2014.207
Ji J, Xue T-F, Guo X-D, Yang J, Guo R-B, Wang J, Huang J-Y, Zhao X-J, Sun X-L (2018) Antagonizing peroxisome proliferator-activated receptor γ facilitates M1-to-M2 shift of microglia by enhancing autophagy via the LKB1-AMPK signaling pathway. Aging Cell 17:e12774. https://doi.org/10.1111/acel.12774
Karunakaran I, Alam S, Jayagopi S, Frohberger SJ, Hansen JN, Kuehlwein J, Hölbling BV, Schumak B, Hübner MP, Gräler MH, Halle A, van Echten-Deckert G (2019) Neural sphingosine 1-phosphate accumulation activates microglia and links impaired autophagy and inflammation. Glia 67:1859–1872. https://doi.org/10.1002/glia.23663
Krashia P, Cordella A, Nobili A, La Barbera L, Federici M, Leuti A, Campanelli F, Natale G, Marino G, Calabrese V, Vedele F, Ghiglieri V, Picconi B, Di Lazzaro G, Schirinzi T, Sancesario G, Casadei N, Riess O, Bernardini S, Pisani A, Calabresi P, Viscomi MT, Serhan CN, Chiurchiù V, D’Amelio M, Mercuri NB (2019) Blunting neuroinflammation with resolvin D1 prevents early pathology in a rat model of Parkinson’s disease. Nat Commun 10:3945. https://doi.org/10.1038/s41467-019-11928-w
Li L, Wu Y, Wang Y, Wu J, Song L, Xian W, Yuan S, Pei L, Shang Y (2014) Resolvin D1 promotes the interleukin-4-induced alternative activation in BV-2 microglial cells. J Neuroinflammation 11:72. https://doi.org/10.1186/1742-2094-11-72
Li M, Li C, Yu H, Cai X, Shen X, Sun X, Wang J, Zhang Y, Wang C (2017) Lentivirus-mediated interleukin-1β (IL-1β) knock-down in the hippocampus alleviates lipopolysaccharide (LPS)-induced memory deficits and anxiety- and depression-like behaviors in mice. J Neuroinflammation 14:190. https://doi.org/10.1186/s12974-017-0964-9
Li Y, Yin L, Fan Z, Su B, Chen Y, Ma Y, Zhong Y, Hou W, Fang Z, Zhang X (2020) Microglia: a potential therapeutic target for sepsis-associated encephalopathy and sepsis-associated chronic pain. Front Pharmacol 11:600421. https://doi.org/10.3389/fphar.2020.600421
Lin S-P, Wei J-X, Hu J-S, Bu J-Y, Zhu L-D, Li Q, Liao H-J, Lin P-Y, Ye S, Chen S-Q, Chen X-H (2021) Artemisinin improves neurocognitive deficits associated with sepsis by activating the AMPK axis in microglia. Acta Pharmacol Sin 42:1069–1079. https://doi.org/10.1038/s41401-021-00634-3
Liu G-J, Tao T, Wang H, Zhou Y, Gao X, Gao Y-Y, Hang C-H, Li W (2020) Functions of resolvin D1-ALX/FPR2 receptor interaction in the hemoglobin-induced microglial inflammatory response and neuronal injury. J Neuroinflammation 17:239. https://doi.org/10.1186/s12974-020-01918-x
Netea-Maier RT, Plantinga TS, van de Veerdonk FL, Smit JW, Netea MG (2016) Modulation of inflammation by autophagy: consequences for human disease. Autophagy 12:245–260. https://doi.org/10.1080/15548627.2015.1071759
Olah M, Biber K, Vinet J, Boddeke HWGM (2011) Microglia phenotype diversity. CNS Neurol Disord Drug Targets 10:108–118
Orihuela R, McPherson CA, Harry GJ (2016) Microglial M1/M2 polarization and metabolic states. Br J Pharmacol 173:649–665. https://doi.org/10.1111/bph.13139
Pang Y, Wu L, Tang C, Wang H, Wei Y (2022) Autophagy-inflammation interplay during infection: balancing pathogen clearance and host inflammation. Front Pharmacol 13:832750. https://doi.org/10.3389/fphar.2022.832750
Plaza-Zabala A, Sierra-Torre V, Sierra A (2017) Autophagy and microglia: novel partners in neurodegeneration and aging. Int J Mol Sci. https://doi.org/10.3390/ijms18030598
Prieto P, Rosales-Mendoza CE, Terrón V, Toledano V, Cuadrado A, López-Collazo E, Bannenberg G, Martín-Sanz P, Fernández-Velasco M, Boscá L (2015) Activation of autophagy in macrophages by pro-resolving lipid mediators. Autophagy 11:1729–1744. https://doi.org/10.1080/15548627.2015.1078958
Qin C, Liu Q, Hu Z-W, Zhou L-Q, Shang K, Bosco DB, Wu L-J, Tian D-S, Wang W (2018) Microglial TLR4-dependent autophagy induces ischemic white matter damage STAT1/6 pathway. Theranostics 8:5434–5451. https://doi.org/10.7150/thno.27882
Qu W, Cheng Y, Peng W, Wu Y, Rui T, Luo C, Zhang J (2022) Targeting iNOS alleviates early brain injury after experimental subarachnoid hemorrhage via promoting ferroptosis of M1 microglia and reducing neuroinflammation. Mol Neurobiol 59:3124–3139. https://doi.org/10.1007/s12035-022-02788-5
Rey C, Nadjar A, Buaud B, Vaysse C, Aubert A, Pallet V, Layé S, Joffre C (2016) Resolvin D1 and E1 promote resolution of inflammation in microglial cells in vitro. Brain Behav Immun 55:249–259. https://doi.org/10.1016/j.bbi.2015.12.013
Rudd KE, Johnson SC, Agesa KM, Shackelford KA, Tsoi D, Kievlan DR, Colombara DV, Ikuta KS, Kissoon N, Finfer S, Fleischmann-Struzek C, Machado FR, Reinhart KK, Rowan K, Seymour CW, Watson RS, West TE, Marinho F, Hay SI, Lozano R, Lopez AD, Angus DC, Murray CJL, Naghavi M (2020) Global, regional, and national sepsis incidence and mortality, 1990–2017: analysis for the Global Burden of Disease Study. Lancet 395:200–211. https://doi.org/10.1016/S0140-6736(19)32989-7
Serhan CN, Levy BD (2018) Resolvins in inflammation: emergence of the pro-resolving superfamily of mediators. J Clin Invest 128:2657–2669. https://doi.org/10.1172/JCI97943
Shen Y, Zhang Y, Du J, Jiang B, Shan T, Li H, Bao H, Si Y (2021) CXCR5 down-regulation alleviates cognitive dysfunction in a mouse model of sepsis-associated encephalopathy: potential role of microglial autophagy and the p38MAPK/NF-κB/STAT3 signaling pathway. J Neuroinflammation 18:246. https://doi.org/10.1186/s12974-021-02300-1
Singer M, Deutschman CS, Seymour CW, Shankar-Hari M, Annane D, Bauer M, Bellomo R, Bernard GR, Chiche J-D, Coopersmith CM, Hotchkiss RS, Levy MM, Marshall JC, Martin GS, Opal SM, Rubenfeld GD, van der Poll T, Vincent J-L, Angus DC (2016) The third international consensus definitions for sepsis and septic shock (sepsis-3). JAMA 315:801–810. https://doi.org/10.1001/jama.2016.0287
Sun Q, Yan H, Chen F, Jiang F, Chen W, Li D, Guo Y (2021) Restoration of proresolution pathway with exogenous resolvin D1 prevents sevoflurane-induced cognitive decline by attenuating neuroinflammation in the hippocampus in rats with type 2 diabetes mellitus. Front Pharmacol 12:720249. https://doi.org/10.3389/fphar.2021.720249
Tauber SC, Djukic M, Gossner J, Eiffert H, Brück W, Nau R (2021) Sepsis-associated encephalopathy and septic encephalitis: an update. Expert Rev Anti Infect Ther 19:215–231. https://doi.org/10.1080/14787210.2020.1812384
Wang HE, Kabeto MM, Gray M, Wadley VG, Muntner P, Judd SE, Safford MM, Kempker J, Levine DA (2021) Trajectory of cognitive decline after sepsis. Crit Care Med 49:1083–1094. https://doi.org/10.1097/CCM.0000000000004897
Westhoff D, Engelen-Lee JY, Hoogland ICM, Aronica EMA, van Westerloo DJ, van de Beek D, van Gool WA (2019) Systemic infection and microglia activation: a prospective postmortem study in sepsis patients. Immun Ageing 16:18. https://doi.org/10.1186/s12979-019-0158-7
Wu Y, Qiu A, Yang Z, Wu J, Li X, Bao K, Wang M, Wu B (2019) Malva sylvestris extract alleviates the astrogliosis and inflammatory stress in LPS-induced depression mice. J Neuroimmunol 336:577029. https://doi.org/10.1016/j.jneuroim.2019.577029
Xu B, Li M, Cheng T, Xia J, Deng X, Hou J (2022) Resolvin D1 protects against sepsis-associated encephalopathy in mice by inhibiting neuro-inflammation induced by microglia. Am J Transl Res 14:6737–6750
Yan X, Yang K, Xiao Q, Hou R, Pan X, Zhu X (2022) Central role of microglia in sepsis-associated encephalopathy: From mechanism to therapy. Front Immunol 13:929316. https://doi.org/10.3389/fimmu.2022.929316
Yirmiya R, Rimmerman N, Reshef R (2015) Depression as a microglial disease. Trends Neurosci 38:637–658. https://doi.org/10.1016/j.tins.2015.08.001
You M, Miao Z, Tian J, Hu F (2020) Trans-10-hydroxy-2-decenoic acid protects against LPS-induced neuroinflammation through FOXO1-mediated activation of autophagy. Eur J Nutr 59:2875–2892. https://doi.org/10.1007/s00394-019-02128-9
Zhang Y-Y, Ning B-T (2021) Signaling pathways and intervention therapies in sepsis. Signal Transduct Target Ther 6:407. https://doi.org/10.1038/s41392-021-00816-9
Zhao X, Cao F, Liu Q, Li X, Xu G, Liu G, Zhang Y, Yang X, Yi S, Xu F, Fan K, Ma J (2019) Behavioral, inflammatory and neurochemical disturbances in LPS and UCMS-induced mouse models of depression. Behav Brain Res 364:494–502. https://doi.org/10.1016/j.bbr.2017.05.064
Zhong X, Xie L, Yang X, Liang F, Yang Y, Tong J, Zhong Y, Zhao K, Tang Y, Yuan C (2020) Ethyl pyruvate protects against sepsis-associated encephalopathy through inhibiting the NLRP3 inflammasome. Mol Med 26:55. https://doi.org/10.1186/s10020-020-00181-3
Zhu W, Cao F-S, Feng J, Chen H-W, Wan J-R, Lu Q, Wang J (2017) NLRP3 inflammasome activation contributes to long-term behavioral alterations in mice injected with lipopolysaccharide. Neuroscience 343:77–84. https://doi.org/10.1016/j.neuroscience.2016.11.037
Funding
This study was supported by the grants from the National Natural Science Foundation of China (81902016), the Natural Science Foundation of Guangdong (2022A1515012129) and the Shenzhen Municipal Science and Technology Foundation (JCYJ20220530152615035).
Author information
Authors and Affiliations
Contributions
WX: Investigation, visualization, writing-original draft preparation. HW: Investigation. HZ: Investigation. YX: Methodology. WG: Investigation. LC: Software. LC: Data Curation. ZD: Writing-reviewing and editing, conceptualization, supervision.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Ethics approval
All experiments procedures involving animals were granted permission by the committee of experimental animals of Tongji Medical College in Huazhong University of Science and Technology (Director: Shunchang Zhou; Date: Mar 5th, 2019; Certificate number: 2019-S1547).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Xiong, W., Wang, H., Zhang, H. et al. Resolvin D1 attenuates depressive-like behavior in LPS-challenged mice by promoting microglial autophagy. Inflammopharmacol 31, 2061–2075 (2023). https://doi.org/10.1007/s10787-023-01234-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10787-023-01234-9