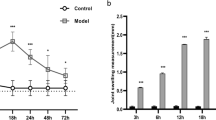
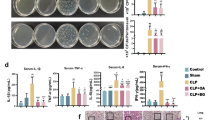
Abstract
Rheumatoid arthritis (RA) is a chronic, systemic autoimmune disease. Dendritic cells (DCs) are one of the most powerful antigen-presenting cells, and they play an important role in RA pathogenesis. Prostaglandin E2 (PGE2) is a potent lipid mediator that can regulate the maturation and activation of DCs, but the molecular mechanisms have not been elucidated. In this study, both in vitro and in an RA rat model, we investigated the mechanisms involved by focusing on PGE2-mediated signaling and using a novel anti-inflammatory compound, paeoniflorin-6′-O-benzene sulfonate (CP-25). PGE2 combined with tumor necrosis factor-α promoted DC maturation and activation through EP4-cAMP signaling. Treatment with CP-25 increased the endocytic capacity of DCs induced by PGE2. CP-25 inhibited the potency of DCs induced by the EP4 receptor agonist, CAY10598, to stimulate allogeneic T cells. Consistent with these findings, the CAY10598-induced upregulation of DC surface activation markers and production of IL-23 was significantly inhibited by CP-25 in a concentration-dependent manner. In vivo administration of CP-25 alleviated adjuvant arthritis (AA) in rats through inhibition of DC maturation and activation. Our results indicate that PGE2-EP4-cAMP signal hyperfunction can lead to abnormal activation of DC functions, which correlates with the course of disease in AA rats and provides a possible treatment target. The inhibition of DC maturation and activation by CP-25 interference of the PGE2-EP4 pathway may significantly contribute to the immunoregulatory profile of CP-25 when used to treat RA and other immune cell-mediated disorders.






Similar content being viewed by others
Abbreviations
- AA:
-
Rat adjuvant-induced arthritis
- BMDCs:
-
Bone marrow-derived DCs
- CFA:
-
Complete Freund’s adjuvant
- CP-25:
-
Paeoniflorin-6′-O-benzene sulfonate
- DC:
-
Dendritic cell
- DMAR:
-
Disease-modifying antirheumatic drugs
- ELISA:
-
Enzyme-linked immunosorbent assay
- FCS:
-
Fetal calf serum
- MLR:
-
Mixed lymphocyte reaction;
- MTX:
-
Methotrexate
- Pae:
-
Paeoniflorin
- PGE2:
-
Prostaglandin E2
- qRT-PCR:
-
Quantification via real-time PCR
- RA:
-
Rheumatoid arthritis
- TNFα:
-
Tumor necrosis factor-α
- TGP:
-
Total glucosides of peony
References
Aghdami N, Gharibdoost F, Moazzeni SM (2008) Experimental autoimmune encephalomyelitis (EAE) induced by antigen pulsed dendritic cells in the C57BL/6 mouse: Influence of injection route. Exp Anim 57:45–55
Ahmed MS, Bae YS (2016) Dendritic Cell-based Immunotherapy for Rheumatoid Arthritis: from Bench to Bedside. Immune Netw 16:44–51
Balanescu A, Nat R, Regalia T, Radu E, Bojinca V, Ionescu R, Predescu V, Popescu E, Predeţeanu D (2003) Correlation between the immunophenotypical presentation of dendritic cells and the clinical response to anti-rheumatic treatment in rheumatoid arthritis. Rom J Intern Med 41:255–267
Banchereau J, Steinman RM (1998) Dendritic cells and the control of immunity. Nature 392:245–252
Chang Y, Jia X, Wei F, Wang C, Sun X, Xu S, Yang X, Zhao Y, Chen J, Wu H, Zhang L, Wei W (2016) CP-25, a novel compound, protects against autoimmune arthritis by modulating immune mediators of inflammation and bone damage. Sci Rep 6:26239
Chen J, Wang Y, Wu H, Yan S, Chang Y, Wei W (2018) A modified compound from paeoniflorin, CP-25, suppressed immune responses and synovium inflammation in collagen-induced arthritis mice. Front Pharmacol 9:563
Firestein GS (2003) Evolving concepts of rheumatoid arthritis. Natur 423:356–361
Flores-Borja F, Mauri C, Ehrenstein MR (2008) Restoring the balance: harnessing regulatory T cells for therapy in rheumatoid arthritis. Eur J Immunol 38:934–937
Grauer O, Wohlleben G, Seubert S, Weishaupt A, Kämpgen E, Gold R (2002) Analysis of maturation states of rat bone marrow-derived dendritic cells using an improved culture technique. Histochem Cell Biol 117:351–362
Hackstein H, Thomson AW (2004) Dendritic cells: Emerging pharmacological targets of immunosuppressive drugs. Nat Rev Immunol 4:24–34
Harizi H, Grosset C, Gualde N (2003) Prostaglandin E2 modulates dendritic cell function via EP2 and EP4 receptor subtypes. J Leukoc Biol 73:756–763
Hayashi S, Miura Y, Tateishi K, Takahashi M, Kurosaka M (2010) Decoy receptor 3 is highly expressed in patients with rheumatoid arthritis. Mod Rheumatol 20:63–68
Van Helden SF, Krooshoop DJ, Broers KC, Raymakers RA, Figdor CG, van Leeuwen FN (2006) A critical role for prostaglandin E2 in podosome dissolution and induction of highspeed migration during dendritic cell maturation. J Immunol 177:1567–1574
Honda T, Segi-Nishida E, Miyachi Y, Narumiya S (2006) Prostacyclin-IP signaling and prostaglandin E2-EP2/EP4 signaling both mediate joint inflammation in mouse collagen-induced arthritis. J Exp Med 203:325–335
Jia XY, Chang Y, Sun XJ, Dai X, Wei W (2014) The role of prostaglandin E2 receptor signaling of dendritic cells in rheumatoid arthritis. Int Immunopharmacol 23:163–169
Jia X, Wei F, Sun X, Chang Y, Xu S, Yang X, Wang C, Wei W (2016) CP-25 attenuates the inflammatory response of fibroblast-like synoviocytes co-cultured with BAFF-activated CD4(+) T cells. J Ethnopharmacol 189:194–201
Jia XY, Chang Y, Wei F, Dai X, Wu YJ, Sun XJ, Xu S, Wu HX, Wang C, Yang XZ, Wei W (2019) CP-25 reverses prostaglandin E4 receptor desensitization-induced fibroblast-like synoviocyte dysfunction via the G protein-coupled receptor kinase 2 in autoimmune arthritis. Acta Pharmacol Sin. https://doi.org/10.1038/s41401-018-0196-2
Jiang GL, Nieves A, Im WB, Old DW, Dinh DT, Wheeler L (2007) The prevention of colitis by E Prostanoid receptor 4 agonist through enhancement of epithelium survival and regeneration. Pharmaol Exp Ther 320:22–28
Khayrullina T, Yen JH, Jing H, Ganea D (2008) In vitro differentiation of dendritic cells in the presence of prostaglandin E2 alters the IL-12/IL-23 balance and promotes differentiation of Th17 cells. J Immunol 181:721–735
Klareskog L, Catrina A, Paget S (2009) Rheumatoid arthritis. Lancet 373:659–672
Lebre MC, Jongbloed SL, Tas SW, Smeets TJ, McInnes IB, Tak PP (2008) Rheumatoid arthritis synovium contains two subsets of CD83-DC-LAMP- dendritic cells with distinct cytokine profiles. Am J Pathol 172:940–950
Liu Y, Wang S, Shen L, Xu Y (2010) Effects of simvastatin on the function of dendritic cells in patients with rheumatic arthritis. J Huazhong Univ Sci Technol Med Sci 30:741–745
Martel-Pelletier J, Pelletier JP, Fahmi H (2003) Cyclooxygenase-2 and prostaglandins in articular tissues. Semin Arthritis Rheum 33:155–167
McCoy JM, Wicks JR, Audoly LP (2002) The role of prostaglandin E2 receptors in the pathogenesis of rheumatoid arthritis. J Clin Invest 110:651–658
Rieser C, Böck G, Klocker H, Bartsch G, Thurnher M (1997) Prostaglandin E2 and tumor necrosis factor a cooperate to activate human dendritic cells: synergistic activation of interleukin 12 production. J Exp Med 186:1603–1608
Sallusto F, Cella M, Danieli C, Lanzavecchia A (1995) Dendritic cells use macropinocytosis and the mannose receptor to concentrate macromolecules in the major histocompatibility complex class II compartment: downregulation by cytokines and bacterial products. J Exp Med 182:389–400
Santiago-Schwarz F, Anand P, Liu S, Carsons SE (2001) Dendritic cells (DCs) in rheumatoid arthritis (RA): progenitor cells and soluble factors contained in RA synovial fluid yield a subset of myeloid DCs that preferentially activate Th1 inflammatory-type responses. J Immunol 167:1758–1768
Sarkar S, Fox DA (2005) Dendritic cells in rheumatoid arthritis. Front Biosci 10:656–665
Schirmer C, Klein C, von Bergen M, Simon JC, Saalbach A (2010) Human fibroblasts support the expansion of IL-17-producing T cells via up-regulation of IL-23 production by dendritic cells. Blood 16:1715–1725
Scott DL, Wolfe F, Huizinga TW (2010) Rheumatoid arthritis. Lancet 376:1094–1108
Sheibanie AF, Tadmori I, Jing H, Vassiliou E, Ganea D (2004) Prostaglandin E2 induces IL-23 production in bone marrow-derived dendritic cells. FASEB J 18:1318–1320
Singh P, Hoggatt J, Hu P, Speth JM, Fukuda S, Breyer RM, Pelus LM (2012) Blockade of prostaglandin E2 signaling through EP1 and EP3 receptors attenuates Flt3L-dependent dendritic cell development from hematopoietic progenitor cells. Blood 119:1671–1682
Swart JF, Wulffraat NM (2014) Mesenchymal stromal cells for treatment of arthritis. Best Pract Res Clin Rheumatol 28:589–603
Thomas R, MacDonald KP, Pettit AR, Cavanagh LL, Padmanabha J, Zehntner S (1999) Dendritic cells and the pathogenesis of rheumatoid arthritis. J Leukoc Biol 66:286–292
Trebino CE, Stock JL, Gibbons CP, Naiman BM, Wachtmann TS, Umland JP, Pandher K, Lapointe JM, Saha S, Roach ML, Carter D, Thomas NA, Durtschi BA, McNeish JD, Hambor JE, Jakobsson PJ, Carty TJ, Perez JR, Audoly LP (2003) Impaired inflammatory and pain responses in mice lacking an inducible prostaglandin E synthase. Proc Natl Acad Sci USA 100:9044–9049
Vassiliou E, Jing H, Ganea D (2003) Prostaglandin E2 inhibits TNF-α production in murine bone marrow-derived dendritic cells. Cell Immunol 223:120–132
Wang Y, Han CC, Cui D, Luo TT, Li Y, Zhang Y, Ma Y, Wei W (2018) Immunomodulatory effects of CP-25 on splenic T cells of rats with adjuvant arthritis. Inflammation 41:1049–1063
Wu H, Chen J, Song S, Yuan P, Liu L, Zhang Y, Zhou A, Chang Y, Zhang L, Wei W (2016) β2-adrenoceptor signaling reduction in dendritic cells is involved in the inflammatory response in adjuvant-induced arthritic rats. Sci Rep 6:24548
Wu YJ, Chen HS, Chen WS, Dong J, Dong XJ, Dai X, Huang Q, Wei W (2018) CP-25 Attenuates the Activation of CD4(+) T Cells Stimulated with Immunoglobulin D in Human. Front Pharmacol 9:4
Yamazaki S, Inaba K, Tarbell KV, Steinman RM (2006) Dendritic cells expand antigen-specific Foxp3+CD25+CD4+ regulatory T cells including suppressors of alloreactivity. Immunol Rev 212:314–329
Yang L, Yamagata N, Yadav R, Brandon S, Courtney RL, Morrow JD, Shyr Y, Boothby M, Joyce S, Carbone DP, Breyer RM (2003) Cancer-associated immunodeficiency and dendritic cell abnormalities mediated by the prostaglandin EP2 receptor. J Clin Invest 111:727–735
Yang X, Zhao Y, Jia X, Wang C, Wu Y, Zhang L, Chang Y, Wei W (2019) CP-25 combined with MTX/LEF ameliorates the progression of adjuvant-induced arthritis by the inhibition on GRK2 translocation. Biomed Pharmacother 110:834–843
Yen JH, Kocieda VP, Jing H, Ganea D (2011) Prostaglandin E2 induces matrix metalloproteinase 9 expression in dendritic cells through two independent signaling pathways leading to activator protein 1 (AP-1) activation. J Biol Chem 286:38913–38923
Zhang F, Shu JL, Li Y, Wu YJ, Zhang XZ, Han L, Tang XY, Wang C, Wang QT, Chen JY, Chang Y, Wu HX, Zhang LL, Wei W (2017) CP-25, a novel anti-inflammatory and immunomodulatory drug, inhibits the functions of activated human B cells through regulating BAFF and TNF-alpha signaling and comparative efficacy with biological agents. Front Pharmacol 8:933
Acknowledgements
This work was supported by the National Natural Science Foundation of China (81330081, 31200675, 81573443, 81603362), by the Anhui Province Nature Science Foundation in the University (KJ2015A317), by Anhui Province Natural Science Fund (outstanding youth) (170808J10).
Author information
Authors and Affiliations
Contributions
WW and YC conceived and designed the study. XYJ designed and performed most of the experiments, and wrote the manuscript. FW, XD, XJS, SX and XZY performed experiments. YJW carried out the flow cytometry assays and helped to revise the manuscript. HXW and CW participated in the design of the study and helped to revise the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there are no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Jia, Xy., Chang, Y., Sun, Xj. et al. Regulatory effects of paeoniflorin-6′-O-benzene sulfonate (CP-25) on dendritic cells maturation and activation via PGE2-EP4 signaling in adjuvant-induced arthritic rats. Inflammopharmacol 27, 997–1010 (2019). https://doi.org/10.1007/s10787-019-00575-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10787-019-00575-8