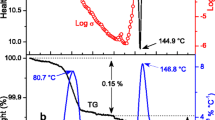
Abstract
Ionic semiclathrate hydrate is a crystalline compound mainly composed of water, which can dissociate at ordinary pressure and temperature. Ionic semiclathrate hydrates have been widely studied to develop prospective green and safety phase change materials utilized in a thermal energy storage system to cool batteries, in air conditioning, and in a cold chain system. The thermophysical properties, such as equilibrium temperature and dissociation heat, and the mass fraction dependence of such properties need to be carefully investigated to design such systems. In this study, the equilibrium temperature and the dissociation heat of tetrabutylphosphonium malonate ionic semiclathrate hydrates were experimentally determined at various mass fractions through an established method: the maximum value of the dissociation heat was 179.70 kJ·kg−1 at w = 0.357 and the equilibrium temperature was 8.9 °C at w = 0.357. By comparing tetrabutylphosphonium malonate ionic semiclathrate hydrates with other media in terms of the measured equilibrium temperature and the dissociation heat, the usability of the hydrates as thermal energy storage media for general air conditioning was discussed.





Similar content being viewed by others
References
P. Kinga, P. Krzysztof, Prog. Mater. Sci. 65, 67–123 (2014). https://doi.org/10.1016/j.pmatsci.2014.03.005
S. Jegadheeswaran, A. Sundaramahalingam, S.D. Pohekar, Int. J. Thermophys. 42, 171 (2021). https://doi.org/10.1007/s10765-021-02921-x
L. Patricia, S.P.T. Chandra, L. Jan, C. George, G. Christ, T. Jan, Int. J. Thermophys. 32, 913–924 (2011). https://doi.org/10.1007/s10765-011-0984-0
ASHRAE TC 9.9 2011, Thermal Guidelines for Data Processing Environments—Expanded Data Center Classes and Usage Guidance. (American Society of Heating, Refrigerating and Air-Conditioning Engineers Inc., 2011), https://www.ashrae.org/. Accessed 18 Oct 2022
K. Hayashi, S. Takao, H. Ogoshi, S. Matsumoto, in Workshop of Annex 10 ECES, International Energy Agency (2000)
J. Shimada, M. Yamada, A. Tani, T. Sugahara, K. Tsunashima, Y. Tsuchida, T. Hirata, J. Chem. Eng. Data 67, 67–73 (2022). https://doi.org/10.1021/acs.jced.1c00741
T. Iwai, I. Ohta, K. Hiraga, K. Kashima, A. Hotta, R. Ohmura, Fluid Phase Equilib. 562, 113561 (2022). https://doi.org/10.1016/j.fluid.2022.113561
T. Suginaka, H. Sakamoto, K. Iino, S. Takeya, M. Nakajima, R. Ohmura, Fluid Phase Equilib. 317, 25–28 (2012). https://doi.org/10.1016/j.fluid.2011.12.010
T. Miyamoto, R. Koyama, N. Kurokawa, A. Hotta, S. Alavi, R. Ohmura, Front. Chem. 8, 547 (2020). https://doi.org/10.3389/fchem.2020.00547
T. Iwai, T. Miyamoto, N. Kurokawa, A. Hotta, R. Ohmura, J. Energy Storage 52(Part A), 104801 (2022). https://doi.org/10.1016/j.est.2022.104801
K. Sato, H. Tokutomi, R. Ohmura, Fluid Phase Equilib. 337, 115–118 (2013). https://doi.org/10.1016/j.fluid.2012.09.016
Y. Arai, Y. Yamauchi, H. Tokutomi, F. Endo, A. Hotta, A. Saman, R. Ohmura, Int. J. Refrig. 88, 102–107 (2018). https://doi.org/10.1016/j.ijrefrig.2017.12.020
Y. Arai, R. Koyama, F. Endo, A. Hotta, R. Ohmura, J. Chem. Thermodyn. 131, 330–335 (2019). https://doi.org/10.1016/j.jct.2018.11.017
R. Koyama, A. Hotta, R. Ohmura, J. Chem. Thermodyn. 144, 106088 (2020). https://doi.org/10.1016/j.jct.2020.106088
Y. Yamauchi, T. Yamasaki, F. Endo, A. Hotta, R. Ohmura, Chem. Eng. Technol. 40, 1810–1816 (2017). https://doi.org/10.1002/ceat.201600459
T. Miyamoto, N. Kurokawa, I. Ota, A. Hotta, R. Ohmura, J. Energy Storage 45, 103773 (2022). https://doi.org/10.1016/j.est.2021.103773
H. Sakamoto, K. Sato, K. Shiraiwa, S. Takeya, M. Nakajima, R. Ohmura, RSC Adv. 1, 315–322 (2011). https://doi.org/10.1039/C1RA00108F
Y.A. Dyadin, K.A. Udachin, J. Incl. Phenom. 2, 61–72 (1984). https://doi.org/10.1007/BF00663240
G.A. Jefferey, Acc. Chem. Res. 2, 344–352 (1969). https://doi.org/10.1021/ar50023a004
H. Nakayama, K. Watanabe, Bull. Chem. Soc. Jpn. 51, 2518–2522 (1978). https://doi.org/10.1246/bcsj.51.2518
S. Muromachi, S. Taketa, Y. Yamamoto, R. Ohmura, CrystEngComm 16, 2056 (2014). https://doi.org/10.1039/c3ce41942h
L.G. Alan, D.S. Raymond, Ind. Eng. Chem. Fundam. 23, 420–425 (1984). https://doi.org/10.1021/i100016a005
R. Koyama, Y. Arai, Y. Yamauchi, S. Takeya, F. Endo, A. Hotta, R. Ohmura, J. Power Sources 427, 70–76 (2019). https://doi.org/10.1016/j.jpowsour.2019.04.055
M. Maruyama, R. Matsuura, R. Ohmura, Sci. Rep. 11, 11315 (2021). https://doi.org/10.1038/s41598-021-90802-6
M. Maruyama, R. Ohmura, Can. J. Chem. Eng. (2022). https://doi.org/10.1002/cjce.24493
H. Oyama, W. Shimada, T. Ebinuma, Y. Kamata, S. Takeya, T. Uchida, J. Nagao, H. Narita, Fluid Phase Equilib. 234, 131–135 (2005). https://doi.org/10.1016/j.fluid.2005.06.005
J. Shimada, M. Shimada, T. Sugahara, K. Tsunashima, A. Tani, Y. Tsuchida, M. Matsumiya, J. Chem. Eng. Data 63, 3615–3620 (2018). https://doi.org/10.1021/acs.jced.1c00741
A.A. Al-Abidi, S. Bin Mat, K. Sopian, M.Y. Sulaiman, C.H. Lim, A. Th, Renew. Sustain. Energy Rev. 16, 5802–5819 (2012). https://doi.org/10.1016/j.rser.2012.05.030
R. Feistel, W. Wagner, J. Phys. Chem. Ref. Data 35, 1021–1047 (2006). https://doi.org/10.1063/1.2183324
Acknowledgement
This study was supported by a Keirin-racing-based research-promotion fund from the JKA Foundation (2022M-270).
Author information
Authors and Affiliations
Contributions
Conceptualization: TI, RO; Methodology: TI, SO, IO, KH; Formal analysis and investigation: TI, SO, IO, KH, RO; Writing—original draft preparation: TI, SO; Writing—review and editing: AH, RO; Funding acquisition: RO; Resources: AH, RO; Supervision: RO.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Iwai, T., Oguro, S., Ota, I. et al. Thermophysical Properties of Ionic Semiclathrate Hydrate Formed with Tetrabutylphosphonium Malonate. Int J Thermophys 44, 28 (2023). https://doi.org/10.1007/s10765-022-03139-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10765-022-03139-1