Abstract
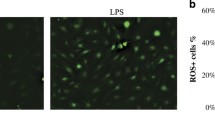
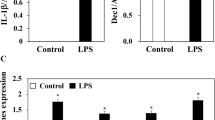
Periodontal disease is the pathological outcome of the overwhelming inflammation in periodontal tissue. Cellular senescence has been associated with chronic inflammation in several diseases. However, the role of cellular senescence in the pathogenesis of periodontal disease remained unclear. This study aimed to investigate the role and the mechanism of cellular senescence in periodontal disease. Using single-cell RNA sequencing, we first found the upregulated level of cellular senescence in fibroblasts and endothelial cells from inflamed gingival tissue. Subsequently, human gingival fibroblasts isolated from healthy and inflamed gingival tissues were labeled as H-GFs and I-GFs, respectively. Compared to H-GFs, I-GFs exhibited a distinct cellular senescence phenotype, including an increased proportion of senescence-associated β-galactosidase (SA-β-gal) positive cells, enlarged cell morphology, and significant upregulation of p16INK4A expression. We further observed increased cellular reactive oxygen species (ROS) activity, mitochondrial ROS, and DNA damage of I-GFs. These phenotypes could be reversed by ROS scavenger NAC, which suggested the cause of cellular senescence in I-GFs. The migration and proliferation assay showed the decreased activity of I-GFs while the gene expression of senescence-associated secretory phenotype (SASP) factors such as IL-1β, IL-6, TGF-β, and IL-8 was all significantly increased. Finally, we found that supernatants of I-GF culture induced more neutrophil extracellular trap (NET) formation and drove macrophage polarization toward the CD86-positive M1 pro-inflammatory phenotype. Altogether, our findings implicate that, in the inflamed gingiva, human gingival fibroblasts acquire a senescent phenotype due to oxidative stress-induced DNA and mitochondrial damage, which in turn activate neutrophils and macrophages through the secretion of SASP factors.







Similar content being viewed by others
Data Availability
Sequence data that support the findings of this study will be made available on request.
References
Hernandez-Segura, A., J. Nehme, and M. Demaria. 2018. Hallmarks of cellular senescence. Trends in Cell Biology 28: 436–453.
Yue, Z., L. Nie, P. Zhao, N. Ji, G. Liao, and Q. Wang. 2022. Senescence-associated secretory phenotype and its impact on oral immune homeostasis. Frontiers in Immunology 13: 1019313.
Gomez, C.R., V. Nomellini, D.E. Faunce, and E.J. Kovacs. 2008. Innate immunity and aging. Experimental Gerontology 43: 718–728.
Hajishengallis, G. 2022. Interconnection of periodontal disease and comorbidities: Evidence, mechanisms, and implications. Periodontology 2000 (89): 9–18.
Salminen, A. 2020. Activation of immunosuppressive network in the aging process. Ageing Research Reviews 57: 100998.
Singh, T., and A.B. Newman. 2011. Inflammatory markers in population studies of aging. Ageing Research Reviews 10: 319–329.
Rea, I.M., D.S. Gibson, V. McGilligan, S.E. McNerlan, H.D. Alexander, and O.A. Ross. 2018. Age and age-related diseases: Role of inflammation triggers and cytokines. Frontiers in Immunology 9: 586.
Tatsumi, M., M. Yanagita, M. Yamashita, S. Hasegawa, K. Ikegami, M. Kitamura, and S. Murakami. 2021. Long-term exposure to cigarette smoke influences characteristics in human gingival fibroblasts. Journal of Periodontal Research 56: 951–963.
Baima, G., M. Romandini, F. Citterio, F. Romano, and M. Aimetti. 2022. Periodontitis and accelerated biological aging: A geroscience approach. Journal of Dental Research 101: 125–132.
Kumari, R., and P. Jat. 2021. Mechanisms of cellular senescence: Cell cycle arrest and senescence associated secretory phenotype. Frontiers in Cell and Developmental Biology 9: 645593.
Coppé, J.P., C.K. Patil, F. Rodier, Y. Sun, D.P. Muñoz, J. Goldstein, P.S. Nelson, P.Y. Desprez, and J. Campisi. 2008. Senescence-associated secretory phenotypes reveal cell-nonautonomous functions of oncogenic RAS and the p53 tumor suppressor. PLoS Biology 6: 2853–2868.
Aquino-Martinez, R., B.A. Eckhardt, J.L. Rowsey, D.G. Fraser, S. Khosla, J.N. Farr, and D.G. Monroe. 2021. Senescent cells exacerbate chronic inflammation and contribute to periodontal disease progression in old mice. Journal of Periodontology 92: 1483–1495.
Di Micco, R., V. Krizhanovsky, D. Baker, and F. d’Adda di Fagagna. 2021. Cellular senescence in ageing: From mechanisms to therapeutic opportunities. Nature Reviews Molecular Cell Biology 22: 75–95.
Ikegami, K., M. Yamashita, M. Suzuki, T. Nakamura, K. Hashimoto, J. Kitagaki, M. Yanagita, M. Kitamura, and S. Murakami. 2023. Cellular senescence with SASP in periodontal ligament cells triggers inflammation in aging periodontal tissue. Aging (Albany NY) 15: 1279–1305.
An, J.Y., K.A. Kerns, A. Ouellette, L. Robinson, H.D. Morris, C. Kaczorowski, S.I. Park, T. Mekvanich, A. Kang, J.S. McLean, T.C. Cox, and M. Kaeberlein. 2020. Rapamycin rejuvenates oral health in aging mice. eLife 9: e54318.
Moiseeva, O., X. Deschênes-Simard, E. St-Germain, S. Igelmann, G. Huot, A.E. Cadar, V. Bourdeau, M.N. Pollak, and G. Ferbeyre. 2013. Metformin inhibits the senescence-associated secretory phenotype by interfering with IKK/NF-κB activation. Aging Cell 12: 489–498.
Demaria, M., N. Ohtani, S.A. Youssef, F. Rodier, W. Toussaint, J.R. Mitchell, R.M. Laberge, J. Vijg, H. Van Steeg, M.E. Dollé, J.H. Hoeijmakers, A. de Bruin, E. Hara, et al. 2014. An essential role for senescent cells in optimal wound healing through secretion of PDGF-AA. Developmental Cell 31: 722–733.
Jurdzinski, K.T., J. Potempa, and A.M. Grabiec. 2020. Epigenetic regulation of inflammation in periodontitis: Cellular mechanisms and therapeutic potential. Clinical Epigenetics 12: 186.
Wielento, A., K.B. Lagosz-Cwik, J. Potempa, and A.M. Grabiec. 2023. The role of gingival fibroblasts in the pathogenesis of periodontitis. Journal of Dental Research 102: 489–496.
Davidson, S., M. Coles, T. Thomas, G. Kollias, B. Ludewig, S. Turley, M. Brenner, and C.D. Buckley. 2021. Fibroblasts as immune regulators in infection, inflammation and cancer. Nature Reviews Immunology 21: 704–717.
Domon, H., K. Tabeta, T. Nakajima, and K. Yamazaki. 2014. Age-related alterations in gene expression of gingival fibroblasts stimulated with Porphyromonas gingivalis. Journal of Periodontal Research 49: 536–543.
Liu, J., X. Wang, M. Zheng, and Q. Luan. 2023. Oxidative stress in human gingival fibroblasts from periodontitis versus healthy counterparts. Oral Diseases 29: 1214–1225.
Williams, D.W., T. Greenwell-Wild, L. Brenchley, N. Dutzan, A. Overmiller, A.P. Sawaya, S. Webb, D. Martin, G. Hajishengallis, K. Divaris, M. Morasso, M. Haniffa, and N.M. Moutsopoulos. 2021. Human oral mucosa cell atlas reveals a stromal-neutrophil axis regulating tissue immunity. Cell 184: 4090-4104.e4015.
Chu, Z., Q. Huang, K. Ma, X. Liu, W. Zhang, S. Cui, Q. Wei, H. Gao, W. Hu, Z. Wang, S. Meng, L. Tian, H. Li, et al. 2023. Novel neutrophil extracellular trap-related mechanisms in diabetic wounds inspire a promising treatment strategy with hypoxia-challenged small extracellular vesicles. Bioactive Materials 27: 257–270.
Shao, Y., Z. Guo, Y. Yang, L. Liu, J. Huang, Y. Chen, L. Li, and B. Sun. 2022. Neutrophil extracellular traps contribute to myofibroblast differentiation and scar hyperplasia through the Toll-like receptor 9/nuclear factor Kappa-B/interleukin-6 pathway. Burns & Trauma 10: tkac044.
Chrysanthopoulou, A., I. Mitroulis, E. Apostolidou, S. Arelaki, D. Mikroulis, T. Konstantinidis, E. Sivridis, M. Koffa, A. Giatromanolaki, D.T. Boumpas, K. Ritis, and K. Kambas. 2014. Neutrophil extracellular traps promote differentiation and function of fibroblasts. Journal of Pathology 233: 294–307.
Tzach-Nahman, R., R. Nashef, O. Fleissig, A. Palmon, L. Shapira, A. Wilensky, and G. Nussbaum. 2017. Oral fibroblasts modulate the macrophage response to bacterial challenge. Scientific Reports 7: 11516.
Page, R.C., and P.I. Eke. 2007. Case definitions for use in population-based surveillance of periodontitis. Journal of Periodontology 78: 1387–1399.
Häkkinen, L., and H. Larjava. 1992. Characterization of fibroblast clones from periodontal granulation tissue in vitro. Journal of Dental Research 71: 1901–1907.
Saha, C., J. Li, X. Sun, X. Liu, and G. Huang. 2023. A novel role of Fas in delaying cellular senescence. Heliyon 9: e13451.
Avelar, R.A., J.G. Ortega, R. Tacutu, E.J. Tyler, D. Bennett, P. Binetti, A. Budovsky, K. Chatsirisupachai, E. Johnson, A. Murray, S. Shields, D. Tejada-Martinez, D. Thornton, et al. 2020. A multidimensional systems biology analysis of cellular senescence in aging and disease. Genome Biology 21: 91.
Chatsirisupachai, K., D. Palmer, S. Ferreira, and J.P. de Magalhães. 2019. A human tissue-specific transcriptomic analysis reveals a complex relationship between aging, cancer, and cellular senescence. Aging Cell 18: e13041.
Schopohl, P., P. Grüneberg, and M.F. Melzig. 2016. The influence of harpagoside and harpagide on TNFα-secretion and cell adhesion molecule mRNA-expression in IFNγ/LPS-stimulated THP-1 cells. Fitoterapia 110: 157–165.
Dave, J.R., S.S. Chandekar, S. Behera, K.U. Desai, P.M. Salve, N.B. Sapkal, S.T. Mhaske, A.M. Dewle, P.S. Pokare, M. Page, A. Jog, P.A. Chivte, R.K. Srivastava, et al. 2022. Human gingival mesenchymal stem cells retain their growth and immunomodulatory characteristics independent of donor age. Science Advances 8: eabm6504.
Wei, X., S. Zou, Z. Xie, Z. Wang, N. Huang, Z. Cen, Y. Hao, C. Zhang, Z. Chen, F. Zhao, Z. Hu, X. Teng, Y. Gui, et al. 2022. EDIL3 deficiency ameliorates adverse cardiac remodelling by neutrophil extracellular traps (NET)-mediated macrophage polarization. Cardiovascular Research 118: 2179–2195.
Bloom, S.I., M.T. Islam, L.A. Lesniewski, and A.J. Donato. 2023. Mechanisms and consequences of endothelial cell senescence. Nature Reviews Cardiology 20: 38–51.
Muñoz-Espín, D., and M. Serrano. 2014. Cellular senescence: From physiology to pathology. Nature Reviews Molecular Cell Biology 15: 482–496.
Silva, L.M., A.D. Doyle, T. Greenwell-Wild, N. Dutzan, C.L. Tran, L. Abusleme, L.J. Juang, J. Leung, E.M. Chun, A.G. Lum, C.S. Agler, C.E. Zuazo, M. Sibree, et al. 2021. Fibrin is a critical regulator of neutrophil effector function at the oral mucosal barrier. Science 374: eabl5450.
Okamura, D.M., and J. Himmelfarb. 2009. Tipping the redox balance of oxidative stress in fibrogenic pathways in chronic kidney disease. Pediatric Nephrology 24: 2309–2319.
Tchkonia, T., Y. Zhu, J. van Deursen, J. Campisi, and J.L. Kirkland. 2013. Cellular senescence and the senescent secretory phenotype: Therapeutic opportunities. Journal of Clinical Investigation 123: 966–972.
Ara, T., K. Kurata, K. Hirai, T. Uchihashi, T. Uematsu, Y. Imamura, K. Furusawa, S. Kurihara, and P.L. Wang. 2009. Human gingival fibroblasts are critical in sustaining inflammation in periodontal disease. Journal of Periodontal Research 44: 21–27.
Van Dyke, T.E., and C.N. Serhan. 2003. Resolution of inflammation: A new paradigm for the pathogenesis of periodontal diseases. Journal of Dental Research 82: 82–90.
Kondo, T., A. Gleason, H. Okawa, A. Hokugo, and I. Nishimura. 2023. Mouse gingival single-cell transcriptomic atlas identified a novel fibroblast subpopulation activated to guide oral barrier immunity in periodontitis. eLife 12: RP88183.
Han, N., Y. Liu, J. Du, J. Xu, L. Guo, and Y. Liu. 2023. Regulation of the host immune microenvironment in periodontitis and periodontal bone remodeling. International Journal of Molecular Sciences 24: 3158.
Xing, L., W. Dong, Y. Chen, W. Dai, X. Xiao, Z. Liu, X. Zhang, D. Bai, and H. Xu. 2023. Fibroblast ferroptosis is involved in periodontitis-induced tissue damage and bone loss. International Immunopharmacology 114: 109607.
Elsayed, R., M. Elashiry, Y. Liu, A. El-Awady, M. Hamrick, and C.W. Cutler. 2021. Porphyromonas gingivalis provokes exosome secretion and paracrine immune senescence in bystander dendritic cells. Frontiers in Cellular and Infection Microbiology 11: 669989.
Holliday, R.S., J. Campbell, and P.M. Preshaw. 2019. Effect of nicotine on human gingival, periodontal ligament and oral epithelial cells. A systematic review of the literature. Journal of Dentistry 86: 81–88.
Sonobe, S., S. Takahashi, S. Hatano, and K. Kuroda. 1986. Phosphorylation of Amoeba G-actin and its effect on actin polymerization. Journal of Biological Chemistry 261: 14837–14843.
Chen, Y., H. Wang, Q. Yang, W. Zhao, Y. Chen, Q. Ni, W. Li, J. Shi, W. Zhang, L. Li, Y. Xu, H. Zhang, D. Miao, et al. 2022. Single-cell RNA landscape of the osteoimmunology microenvironment in periodontitis. Theranostics 12: 1074–1096.
Herranz, N., and J. Gil. 2018. Mechanisms and functions of cellular senescence. Journal of Clinical Investigation 128: 1238–1246.
Paez, J., R. Hernandez, J. Espinoza, L. Rojas, C.E. Martinez, N. Tobar, J. Martinez, and P.C. Smith. 2020. Uncoupled inflammatory, proliferative, and cytoskeletal responses in senescent human gingival fibroblasts. Journal of Periodontal Research 55: 432–440.
Serrano, M., A.W. Lin, M.E. McCurrach, D. Beach, and S.W. Lowe. 1997. Oncogenic ras provokes premature cell senescence associated with accumulation of p53 and p16INK4a. Cell 88: 593–602.
Coppe, J.P., P.Y. Desprez, A. Krtolica, and J. Campisi. 2010. The senescence-associated secretory phenotype: The dark side of tumor suppression. Annual Review of Phytopathology 5: 99–118.
Cheng, R., D. Choudhury, C. Liu, S. Billet, T. Hu, and N.A. Bhowmick. 2015. Gingival fibroblasts resist apoptosis in response to oxidative stress in a model of periodontal diseases. Cell Death Discovery 1: 15046.
Gorąca, A., H. Huk-Kolega, P. Kleniewska, A. Piechota-Polańczyk, and B. Skibska. 2013. Effects of lipoic acid on spleen oxidative stress after LPS administration. Pharmacological Reports 65: 179–186.
Maruyama, T., T. Tomofuji, Y. Endo, K. Irie, T. Azuma, D. Ekuni, N. Tamaki, T. Yamamoto, and M. Morita. 2011. Supplementation of green tea catechins in dentifrices suppresses gingival oxidative stress and periodontal inflammation. Archives of Oral Biology 56: 48–53.
Sinha, K., J. Das, P.B. Pal, and P.C. Sil. 2013. Oxidative stress: The mitochondria-dependent and mitochondria-independent pathways of apoptosis. Archives of Toxicology 87: 1157–1180.
Naruishi, K., and T. Nagata. 2018. Biological effects of interleukin-6 on gingival fibroblasts: Cytokine regulation in periodontitis. Journal of Cellular Physiology 233: 6393–6400.
Morandini, A.C., C.R. Sipert, T.H. Gasparoto, S.L. Greghi, E. Passanezi, M.L. Rezende, and A. P. Sant’ana, A. P. Campanelli, G. P. Garlet, and C. F. Santos. 2010. Differential production of macrophage inflammatory protein-1alpha, stromal-derived factor-1, and IL-6 by human cultured periodontal ligament and gingival fibroblasts challenged with lipopolysaccharide from P. gingivalis. Journal of Periodontology 81: 310–317.
Boström, E.A., E. Kindstedt, R. Sulniute, P. Palmqvist, M. Majster, C.K. Holm, S. Zwicker, R. Clark, S. Önell, I. Johansson, U.H. Lerner, and P. Lundberg. 2015. Increased eotaxin and MCP-1 levels in serum from individuals with periodontitis and in human gingival fibroblasts exposed to pro-inflammatory cytokines. PLoS ONE 10: e0134608.
Liang, S., K.B. Hosur, H. Domon, and G. Hajishengallis. 2010. Periodontal inflammation and bone loss in aged mice. Journal of Periodontal Research 45: 574–578.
Miller, C.S., X. Ding, D.R. Dawson 3rd., and J.L. Ebersole. 2021. Salivary biomarkers for discriminating periodontitis in the presence of diabetes. Journal of Clinical Periodontology 48: 216–225.
Schalper, K.A., M. Carleton, M. Zhou, T. Chen, Y. Feng, S.P. Huang, A.M. Walsh, V. Baxi, D. Pandya, T. Baradet, D. Locke, Q. Wu, T.P. Reilly, et al. 2020. Elevated serum interleukin-8 is associated with enhanced intratumor neutrophils and reduced clinical benefit of immune-checkpoint inhibitors. Nature Medicine 26: 688–692.
Liao, Y., Q. Yan, T. Cheng, H. Yao, Y. Zhao, D. Fu, Y. Ji, and B. Shi. 2023. Sulforaphene inhibits periodontitis through regulating macrophage polarization via upregulating dendritic cell immunoreceptor. Journal of Agricultural and Food Chemistry 71: 15538–15552.
Li, C., T. Chen, J. Liu, Y. Wang, C. Zhang, L. Guo, D. Shi, T. Zhang, X. Wang, and J. Li. 2023. FGF19-induced inflammatory CAF promoted neutrophil extracellular trap formation in the liver metastasis of colorectal cancer. Advanced Science (Weinheim, Germany) 10: e2302613.
Qiu, W., Z. Wang, Z. Chen, Q. Sun, H. Wu, Z. Chen, K. Luan, Z. Liu, D. Ding, Q. Tu, J. Chen, B. Wu, and F. Fang. 2023. The adiponectin receptor agonist AdipoAI attenuates periodontitis in diabetic rats by inhibiting gingival fibroblast-induced macrophage migration. British Journal of Pharmacology 180: 2436–2451.
Funding
This work was supported by the National Natural Science Foundation of China (grant numbers 82071095 and 32370816). The authors assert that AI techniques should be utilized solely to enhance readability and language rather than replace critical research tasks.
Author information
Authors and Affiliations
Contributions
S.G. and L.F.: conceptualization, formal analysis, investigation, project administration, validation, visualization, and writing—original draft. C.Y. and W.S.: data curation, methodology, and resources. Q.S., L.C., and T.X.: investigation, resources, and validation. M.W. and H.X.: conceptualization, data curation, formal analysis, funding acquisition, project administration, supervision, and writing—review and editing.
Corresponding authors
Ethics declarations
Ethics Approval
This study was reviewed and approved by the Ethics Committee of Hospital of Stomatology Wuhan University (No. 2023/A52), and conducted in accordance with the revised Declaration of Helsinki 2013.
Consent to Participate
Informed consent was obtained from all individual participants included in the study.
Consent to Publication
Not applicable.
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Guo, S., Fu, L., Yin, C. et al. ROS-Induced Gingival Fibroblast Senescence: Implications in Exacerbating Inflammatory Responses in Periodontal Disease. Inflammation (2024). https://doi.org/10.1007/s10753-024-02014-5
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10753-024-02014-5