Abstract
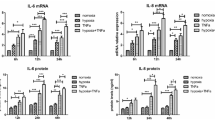
Synovial hypoxia-inducible factor 1α (HIF-1α) is a prospective therapeutic target for rheumatoid arthritis (RA). AMSP-30 m, a novel HIF-1α inhibitor, was reported to have notable anti-arthritic effects in rats with adjuvant-induced arthritis. However, its roles in inhibiting the pathogenic behaviors of fibroblast-like synoviocytes (FLS) and the involved mechanisms remain unknown. Here, AMSP-30 m inhibited proliferation and induced apoptosis in hypoxia-induced RA FLS (MH7A cell line), as evidenced by decreased cell viability, reduced Ki67-positive cells, G0/G1 phase arrest, lowered C-myc and Cyclin D1 protein levels, emergence of apoptotic nuclear fragmentation, raised apoptosis rates, and activation of caspase 3. Furthermore, AMSP-30 m prevented hypoxia-induced increases in pro-inflammatory factor production, MMP-2 activity, migration index, migrated/invasive cells, and actin cytoskeletal rearrangement. In vivo, AMSP-30 m alleviated the severity of rat collagen-induced arthritis (CIA). Mechanically, AMSP-30 m reduced HIF-1α expression and blocked sonic hedgehog (Shh) pathway activation in hypoxia-induced MH7A cells and CIA rat synovium, as shown by declines in pathway-related proteins (Shh, Smo, and Gli-1). Particularly, the combination of Shh pathway inhibitor cyclopamine enhanced AMSP-30 m’s inhibitory effects on the pathogenic behaviors of hypoxia-stimulated MH7A cells, whereas the combination of Shh pathway activator SAG canceled AMSP-30 m’s therapeutic effects in vitro and in CIA rats, implying a close involvement of Shh pathway inhibition in its anti-arthritic effects. We likewise confirmed AMSP-30 m’s anti-proliferative role in hypoxia-induced primary CIA FLS. Totally, AMSP-30 m suppressed hypoxia-induced proliferation, inflammation, migration, and invasion of MH7A cells and ameliorated the severity of rat CIA via inhibiting Shh signaling.









Similar content being viewed by others
Availability of Data and Materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Aletaha, D., and J.S. Smolen. 2018. Diagnosis and management of rheumatoid arthritis: A review. JAMA 320: 1360–1372.
Wu, Z., D. Ma, H. Yang, J. Gao, G. Zhang, K. Xu, and L. Zhang. 2021. Fibroblast-like synoviocytes in rheumatoid arthritis: Surface markers and phenotypes. International Immunopharmacology 93: 107392.
Masoumi, M., H. Bashiri, H. Khorramdelazad, K. Barzaman, N. Hashemi, H.A. Sereshki, A. Sahebkar, and J. Karami. 2021. Destructive roles of fibroblast-like synoviocytes in chronic inflammation and joint damage in rheumatoid arthritis. Inflammation 44: 466–479.
Bartok, B., and G.S. Firestein. 2010. Fibroblast-like synoviocytes: Key effector cells in rheumatoid arthritis. Immunological Reviews 233: 233–255.
Tsaltskan, V., and G.S. Firestein. 2022. Targeting fibroblast-like synoviocytes in rheumatoid arthritis. Current Opinion in Pharmacology 67: 102304.
Sabi, E.M., A. Singh, Z.M. Althafar, T. Behl, A. Sehgal, S. Singh, N. Sharma, S. Bhatia, A. Al-Harrasi, H.M. Alqahtani, and S. Bungau. 2022. Elucidating the role of hypoxia-inducible factor in rheumatoid arthritis. Inflammopharmacology 30: 737–748.
Gaber, T., R. Dziurla, R. Tripmacher, G.R. Burmester, and F. Buttgereit. 2005. Hypoxia inducible factor (HIF) in rheumatology: Low O2! See what HIF can do! Annals of the Rheumatic Diseases 64: 971–980.
Konisti, S., S. Kiriakidis, and E.M. Paleolog. 2012. Hypoxia–a key regulator of angiogenesis and inflammation in rheumatoid arthritis. Nature Reviews Rheumatology 8: 153–162.
Muz, B., M.N. Khan, S. Kiriakidis, and E.M. Paleolog. 2009. Hypoxia. The role of hypoxia and HIF-dependent signalling events in rheumatoid arthritis. Arthritis Research Therapy 11: 201.
Ahn, J.K., E.M. Koh, H.S. Cha, Y.S. Lee, J. Kim, E.K. Bae, and K.S. Ahn. 2008. Role of hypoxia-inducible factor-1alpha in hypoxia-induced expressions of IL-8, MMP-1 and MMP-3 in rheumatoid fibroblast-like synoviocytes. Rheumatology (Oxford) 47: 834–839.
Hu, F., R. Mu, J. Zhu, L. Shi, Y. Li, X. Liu, W. Shao, G. Li, M. Li, Y. Su, P.L. Cohen, X. Qiu, and Z. Li. 2014. Hypoxia and hypoxia-inducible factor-1alpha provoke toll-like receptor signalling-induced inflammation in rheumatoid arthritis. Annals of the Rheumatic Diseases 73: 928–936.
Hu, Y., T. Zhang, J. Chen, W. Cheng, J. Chen, Z. Zheng, J. Lin, G. Zhu, Y. Zhang, X. Bai, Y. Wang, B. Song, Q. Wang, L. Qin, and P. Zhang. 2020. Downregulation of hypoxia-inducible factor-1alpha by RNA interference alleviates the development of collagen-induced arthritis in rats. Molecular Therapy Nucleic acids 19: 1330–1342.
Li, G.F., Y.H. Qin, and P.Q. Du. 2015. Andrographolide inhibits the migration, invasion and matrix metalloproteinase expression of rheumatoid arthritis fibroblast-like synoviocytes via inhibition of HIF-1alpha signaling. Life Sciences 136: 67–72.
Hua, S., and T.H. Dias. 2016. Hypoxia-inducible factor (HIF) as a target for novel therapies in rheumatoid arthritis. Frontiers in Pharmacology 7: 184.
Liu, M., Y. Liang, Z. Zhu, J. Wang, X. Cheng, J. Cheng, B. Xu, R. Li, X. Liu, and Y. Wang. 2019. Discovery of novel aryl carboxamide derivatives as hypoxia-inducible factor 1alpha signaling inhibitors with potent activities of anticancer metastasis. Journal of Medicinal Chemistry 62: 9299–9314.
Meng, B., F.Y. Liu, M.M. Liu, L.C. Yu, W.T. Zhang, M.Y. Zhou, S.Y. Liu, R. Li, and L. Cai. 2022. AMSP-30 m as a novel HIF-1alpha inhibitor attenuates the development and severity of adjuvant-induced arthritis in rats: Impacts on synovial apoptosis, synovial angiogenesis and sonic hedgehog signaling pathway. International Immunopharmacology 103: 108467.
Mu, Y.R., M.Y. Zhou, L. Cai, M.M. Liu, and R. Li. 2020. Overexpression of aquaporin 1 in synovium aggravates rat collagen-induced arthritis through regulating beta-catenin signaling: An in vivo and in vitro study. Journal of Inflammation Research 13: 701–712.
Zhou, J., Y. Mao, X. Shi, Y. Zhang, X. Yu, X. Liu, L. Diao, X. Yang, C. Liu, D. Liu, X. Tan, and M. Liu. 2022. Peimine suppresses collagen-induced arthritis, activated fibroblast-like synoviocytes and TNFalpha-induced MAPK pathways. International Immunopharmacology 111: 109181.
Lampropoulos, C.E., P. Orfanos, V.K. Bournia, T. Karatsourakis, C. Mavragani, D. Pikazis, M.N. Manoussakis, A.G. Tzioufas, H.M. Moutsopoulos, and P.G. Vlachoyiannopoulos. 2015. Adverse events and infections in patients with rheumatoid arthritis treated with conventional drugs or biologic agents: A real world study. Clinical and Experimental Rheumatology 33: 216–224.
Ban, H.S., Y. Uto, and H. Nakamura. 2021. Hypoxia-inducible factor (HIF) inhibitors: A patent survey (2016–2020). Expert Opinion on Therapeutic Patents 31: 387–397.
Fearon, U., M. Canavan, M. Biniecka, and D.J. Veale. 2016. Hypoxia, mitochondrial dysfunction and synovial invasiveness in rheumatoid arthritis. Nature Reviews Rheumatology 12: 385–397.
Akhavani, M.A., L. Madden, I. Buysschaert, B. Sivakumar, N. Kang, and E.M. Paleolog. 2009. Hypoxia upregulates angiogenesis and synovial cell migration in rheumatoid arthritis. Arthritis Research & Therapy 11: R64.
Yu, S., Y. Lu, M. Zong, Q. Tan, and L. Fan. 2019. Hypoxia-induced miR-191-C/EBPbeta signaling regulates cell proliferation and apoptosis of fibroblast-like synoviocytes from patients with rheumatoid arthritis. Arthritis Research & Therapy 21: 78.
Zhang, Y., and B. Zhang. 2016. Trichostatin A, an inhibitor of histone deacetylase, inhibits the viability and invasiveness of hypoxic rheumatoid arthritis fibroblast-like synoviocytes via PI3K/Akt signaling. Journal of Biochemical and Molecular Toxicology 30: 163–169.
Noss, E.H., and M.B. Brenner. 2008. The role and therapeutic implications of fibroblast-like synoviocytes in inflammation and cartilage erosion in rheumatoid arthritis. Immunological Reviews 223: 252–270.
Sun, J., Y. Su, Y. Xu, D. Qin, Q. He, H. Qiu, J. Zhuo, and W. Li. 2022. CD36 deficiency inhibits proliferation by cell cycle control in skeletal muscle cells. Frontiers in Physiology 13: 947325.
Huang, X.Y., X.M. Zhang, F.H. Chen, L.L. Zhou, X.F. Deng, Y.J. Liu, and X.J. Li. 2014. Anti-proliferative effect of recombinant human endostatin on synovial fibroblasts in rats with adjuvant arthritis. European Journal of Pharmacology 723: 7–14.
Zhang, Q., J. Liu, M. Zhang, S. Wei, R. Li, Y. Gao, W. Peng, and C. Wu. 2019. Apoptosis induction of fibroblast-like synoviocytes is an important molecular-mechanism for herbal medicine along with its active components in treating rheumatoid arthritis. Biomolecules 9: 795.
Cai, L., P. Zong, M.Y. Zhou, F.Y. Liu, B. Meng, M.M. Liu, Z. Li, and R. Li. 2022. 7-Hydroxycoumarin mitigates the severity of collagen-induced arthritis in rats by inhibiting proliferation and inducing apoptosis of fibroblast-like synoviocytes via suppression of Wnt/beta-catenin signaling pathway. Phytomedicine 94: 153841.
Alam, J., I. Jantan, and S.N.A. Bukhari. 2017. Rheumatoid arthritis: Recent advances on its etiology, role of cytokines and pharmacotherapy. Biomedicine & Pharmacotherapy 92: 615–633.
Nygaard, G., and G.S. Firestein. 2020. Restoring synovial homeostasis in rheumatoid arthritis by targeting fibroblast-like synoviocytes. Nature Reviews Rheumatology 16: 316–333.
Bottini, N., and G.S. Firestein. 2013. Duality of fibroblast-like synoviocytes in RA: Passive responders and imprinted aggressors. Nature Reviews Rheumatology 9: 24–33.
Malemud, C.J. 2017. Matrix metalloproteinases and synovial joint pathology. Progress in Molecular Biology and Translational Science 148: 305–325.
Dominguez, R., and K.C. Holmes. 2011. Actin structure and function. Annual Review of Biophysics 40: 169–186.
Liu, F., X.X. Feng, S.L. Zhu, H.Y. Huang, Y.D. Chen, Y.F. Pan, R.R. June, S.G. Zheng, and J.L. Huang. 2018. Sonic hedgehog signaling pathway mediates proliferation and migration of fibroblast-like synoviocytes in rheumatoid arthritis via MAPK/ERK signaling pathway. Frontiers in Immunology 9: 2847.
Wang, M., S. Zhu, W. Peng, Q. Li, Z. Li, M. Luo, X. Feng, Z. Lin, and J. Huang. 2014. Sonic hedgehog signaling drives proliferation of synoviocytes in rheumatoid arthritis: A possible novel therapeutic target. Journal of Immunology Research 2014: 401903.
Zhu, S., J. Dang, Y. Shi, X. Feng, Y. Hu, L. Lin, and J. Huang. 2022. Sonic hedgehog promotes synovial inflammation and articular damage through p38 mitogen-activated protein kinase signaling in experimental arthritis. Journal of Autoimmunity 132: 102902.
Bhuria, V., J. Xing, T. Scholta, K.C. Bui, M.L.T. Nguyen, N.P. Malek, P. Bozko, and R.R. Plentz. 2019. Hypoxia induced sonic hedgehog signaling regulates cancer stemness, epithelial-to-mesenchymal transition and invasion in cholangiocarcinoma. Experimental Cell Research 385: 111671.
Bijlsma, M.F., A.P. Groot, J.P. Oduro, R.J. Franken, S. Schoenmakers, M.P. Peppelenbosch, and C.A. Spek. 2009. Hypoxia induces a hedgehog response mediated by HIF-1alpha. Journal of Cellular and Molecular Medicine 13: 2053–2060.
Wang, G., Z. Zhang, Z. Xu, H. Yin, L. Bai, Z. Ma, M.A. Decoster, G. Qian, and G. Wu. 2010. Activation of the sonic hedgehog signaling controls human pulmonary arterial smooth muscle cell proliferation in response to hypoxia. Biochimica et Biophysica Acta 1803: 1359–1367.
Miyazawa, K., A. Mori, and H. Okudaira. 1998. Establishment and characterization of a novel human rheumatoid fibroblast-like synoviocyte line, MH7A, immortalized with SV40 T antigen. Journal of Biochemistry 124: 1153–1162.
Chen, J., G. Zhu, Y. Sun, Y. Wu, B. Wu, W. Zheng, X. Ma, and Y. Zheng. 2022. 7-deacetyl-gedunin suppresses proliferation of human rheumatoid arthritis synovial fibroblast through activation of Nrf2/ARE signaling. International Immunopharmacology 107: 108557.
Gan, P., M. Sun, H. Wu, J. Ke, X. Dong, and F. Chen. 2022. A novel mechanism for inhibiting proliferation of rheumatoid arthritis fibroblast-like synoviocytes: Geniposide suppresses HIF-1alpha accumulation in the hypoxic microenvironment of synovium. Inflammation Research 71: 1375–1388.
Konishi, H., S.E. Kanou, R. Yukimatsu, M. Inui, M. Sato, N. Yamamoto, M. Nakano, and M. Koshiba. 2022. Adenosine inhibits TNFalpha-induced MMP-3 production in MH7A rheumatoid arthritis synoviocytes via A(2A) receptor signaling. Science and Reports 12: 6033.
Funding
This work was supported by the National Natural Science Foundation of China (81972040), Anhui Provincial Natural Science Foundation (2108085MH321), The University Synergy Innovation Program of Anhui Province (GXXT-2022–063), Scientific Research Level Promotion Program of Anhui Medical University (2022xkjT008), Open Project Program of Inflammation and Immune Mediated Diseases Laboratory of Anhui Province (IMMDL202001), Research Fund of Anhui Institute of Translational Medicine (2022zhyx-C14), and Undergraduate Innovation Training Program of Anhui Medical University (2021-ZQKY-156, 2022-ZQKY-134).
Author information
Authors and Affiliations
Contributions
Li Cai: investigation and writing—original draft. Bo Meng, Fei Jiang, Meng-qing Wang, Xin-jie Wu, and Ming-wang Hu: investigation. Wen-hao Shu, Xiao-hua Wang, and Yu-chen Yang: data curation and formal analysis. Xiang Ran: conceptualization, methodology, and visualization. Rong Li: conceptualization, methodology, supervision, writing—review and editing, and funding acquisition. All authors read and approved the manuscript.
Corresponding authors
Ethics declarations
Ethical Approval
This study was approved by the Ethical Committee on Animal Research of Anhui Medical University (approval number: LLSC20220564, approval date: July 26, 2022).
Consent for Publication
Not applicable.
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Li Cai, Bo Meng, and Fei Jiang contribute equally to this work
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Cai, L., Meng, B., Jiang, F. et al. Novel HIF-1α Inhibitor AMSP-30m Mitigates the Pathogenic Cellular Behaviors of Hypoxia-Stimulated Fibroblast-Like Synoviocytes and Alleviates Collagen-Induced Arthritis in Rats via Inhibiting Sonic Hedgehog Pathway. Inflammation 46, 2289–2305 (2023). https://doi.org/10.1007/s10753-023-01878-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10753-023-01878-3