Abstract
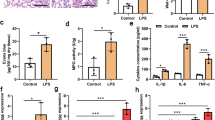
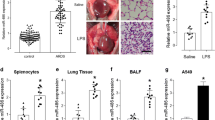
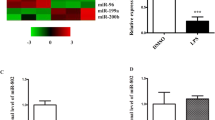
Exacerbated inflammation and apoptosis are considered upstream events associated with acute lung injury (ALI). microRNAs are critical regulators of genes responsible for inflammation and apoptosis and are considered potential therapeutic targets for ameliorating ALI. This study was undertaken to uncover the role of miR-7-5p in LPS-induced lung injury. A LPS-induced inflammation model was established using BEAS-2B cells and C57BL/6 mice. Bioinformatics analysis and the luciferase reporter assay confirmed that Raf-1 is a target of miR-7-5p and that its expression was inversely correlated with expression of proinflammatory markers and miR-7-5p, whereas miR-7-5p inhibition in vitro led to subsequent restoration of Raf-1 expression and prevention of apoptosis. Intranasal (i.n.) administration of antagomir using the C57BL/6 mouse model further confirmed that miR-7-5p inhibition suppresses LPS-induced inflammation and apoptosis via modulating the miR-7-5p/Raf-1 axis. Our findings indicate that blocking miR-7-5p expression by antagomir protects mice from LPS-induced lung injury by suppressing inflammation and activation of mitochondria-mediated survival signalling. In conclusion, our findings demonstrate a previously unknown pathophysiological role of miR-7-5p in the progression of ALI, and targeted i.n. administration of miR-7-5p antagomir could aid in the development of potential therapeutic strategies against lung injury.










Similar content being viewed by others
AVAILABILITY OF DATA AND MATERIALS
All data generated or analyzed during this study are included in this article.
Abbreviations
- COPD:
-
Chronic obstructive pulmonary disease
- ERK:
-
Extracellular signal–regulated protein kinases
- Bcl-2:
-
B-cell leukemia/lymphoma 2
- Bax:
-
Bcl-2-associated X protein
- MAPK:
-
Mitogen-activated protein kinase
- UTR:
-
Untranslated region
References
Luo, Q., J. Zhu, Q. Zhang, J. Xie, C. Yi, and T. Li. 2020. MicroRNA-486-5p promotes acute lung injury via inducing inflammation and apoptosis by targeting OTUD7B. Inflammation 43: 975–984.
Kosutova, P., P. Mikolka, M. Kolomaznik, S. Balentova, M. Adamkov, A. Calkovska, and D. Mokra. 2018. Reduction of lung inflammation, oxidative stress and apoptosis by the PDE4 inhibitor roflumilast in experimental model of acute lung injury. Physiological Research 67: S645–S654.
Khan, A.J., and R. Nanchal. 2007. Cotton dust lung diseases. Current Opinion in Pulmonary Medicine 13: 137–141. https://doi.org/10.1097/MCP.0b013e32802c7ceb.
Brass, D.M., J.W. Hollingsworth, M. Cinque, Z. Li, E. Potts, E. Toloza, W.M. Foster, and D.A. Schwartz. 2008. Chronic LPS inhalation causes emphysema-like changes in mouse lung that are associated with apoptosis. American Journal of Respiratory Cell and Molecular Biology 39: 584–590. https://doi.org/10.1165/rcmb.2007-0448OC.
Shen, W., X. Zhao, and S. Li. 2022. Exosomes derived from ADSCs attenuate sepsis-induced lung injury by delivery of circ-fryl and regulation of the miR-490-3p/SIRT3 Pathway. Inflammation 45: 331–342.
Luo, H., H. Liang, Y. Chen, S. Chen, Y. Xu, L. Xu, J. Liu, K. Zhou, J. Peng, and G. Guo. 2018. miR-7-5p overexpression suppresses cell proliferation and promotes apoptosis through inhibiting the ability of DNA damage repair of PARP-1 and BRCA1 in TK6 cells exposed to hydroquinone. Chemico-Biological Interactions 283: 84–90.
Liu, X., and J. Meng. 2018. Luteolin alleviates LPS-induced bronchopneumonia injury in vitro and in vivo by down-regulating microRNA-132 expression. Biomedicine & Pharmacotherapy 106: 1641–1649.
Pradhan, R., S. Chatterjee, K.C. Hembram, C. Sethy, M. Mandal, and C.N. Kundu. 2021. Nano formulated resveratrol inhibits metastasis and angiogenesis by reducing inflammatory cytokines in oral cancer cells by targeting tumor associated macrophages. Journal of Nutritional Biochemistry 92: 108624.
Livak, K.J., and T.D. Schmittgen. 2001. Analysis of relative gene expression data using real-time quantitative PCR and the 2- ΔΔCT method. Methods 25: 402–408. https://doi.org/10.1006/meth.2001.1262.
More, G.K., and R.T. Makola. 2020. In-vitro analysis of free radical scavenging activities and suppression of LPS-induced ROS production in macrophage cells by Solanum sisymbriifolium extracts. Scientific Reports 10: 1–9.
Zhu, J.H., A.M. Gusdon, H. Cimen, B. Van Houten, E. Koc, and C.T. Chu. 2012. Impaired mitochondrial biogenesis contributes to depletion of functional mitochondria in chronic MPP+ toxicity: Dual roles for ERK1/2. Cell Death & Disease 3: e312–e312. https://doi.org/10.1038/cddis.2012.46.
Plank, M.W., S. Maltby, H.L. Tay, J. Stewart, F. Eyers, P.M. Hansbro, and P.S. Foster. 2015. MicroRNA expression is altered in an ovalbumin-induced asthma model and targeting miR-155 with antagomirs reveals cellular specificity. PLoS ONE 10: e0144810.
Zhang, Y., J. Zhang, Y. Ren, T. Li, J. Bi, Z. Du, and R. Wu. 2021. Luteolin suppresses sepsis-induced cold-inducible RNA-binding protein production and lung injury in neonatal mice. Shock 55: 268–273. https://doi.org/10.1097/SHK.0000000000001624.
Zhang, X., X. Zhang, S. Hu, M. Zheng, J. Zhang, J. Zhao, X. Zhang, B. Yan, L. Jia, and J. Zhao. 2017. Identification of miRNA-7 by genome-wide analysis as a critical sensitizer for TRAIL-induced apoptosis in glioblastoma cells. Nucleic Acids Research 45: 5930–5944. https://doi.org/10.1093/nar/gkx317.
Shi, Y., X. Luo, P. Li, J. Tan, X. Wang, T. Xiang, and G. Ren. 2015. miR-7-5p suppresses cell proliferation and induces apoptosis of breast cancer cells mainly by targeting REGγ. Cancer Letters 35: 827–836. https://doi.org/10.1016/j.canlet.2014.12.014.
Fauconnier, J., D.C. Andersson, S.J. Zhang, J.T. Lanner, R. Wibom, A. Katz, J.D. Bruton, and H. Westerblad. 2007. Effects of palmitate on Ca2+ handling in adult control and ob/ob cardiomyocytes: Impact of mitochondrial reactive oxygen species. Diabetes 56: 1136–1142. https://doi.org/10.2337/db06-0739.
Martins, A.S., V.M. Shkryl, M.C. Nowycky, and N. Shirokova. 2008. Reactive oxygen species contribute to Ca2+ signals produced by osmotic stress in mouse skeletal muscle fibres. The Journal of Physiology 586: 197–210. https://doi.org/10.1113/jphysiol.2007.146571.
Liu, X., X. Zhao, X. Li, S. Lv, R. Ma, Y. Qi, A. Abulikemu, H. Duan, C. Guo, and Y. Li. 2020. PM2. 5. triggered apoptosis in lung epithelial cells through the mitochondrial apoptotic way mediated by a ROS-DRP1-mitochondrial fission axis. Journal of Hazardous Materials 397: 122608.
Leermakers, P.A., A. Schols, A.E.M. Kneppers, M. Kelders, C.C. de Theije, M. Lainscak, and H.R. Gosker. 2018. Molecular signalling towards mitochondrial breakdown is enhanced in skeletal muscle of patients with chronic obstructive pulmonary disease (COPD). Scientific Reports 8: 1–13. https://doi.org/10.1038/s41598-018-33471-2.
Zhong, J., J. Troppmair, and U.R. Rapp. 2001. Independent control of cell survival by Raf-1 and Bcl-2 at the mitochondria. Oncogene 20: 4807–4816. https://doi.org/10.1038/sj.onc.1204614.
Wang, H.G., U.R. Rapp, and J.C. Reed. 1996. Bcl-2 targets the protein kinase Raf-1 to mitochondria. Cell 87: 629–638. https://doi.org/10.1016/s0092-8674(00)81383-5.
Zhang, Z., Q. Nian, G. Chen, S. Cui, Y. Han, and J. Zhang. 2020. Klotho alleviates lung injury caused by paraquat via suppressing ROS/P38 MAPK-regulated inflammatory responses and apoptosis. Oxidative Medicine and Cellular Longevity 2020: 2020.
Polikepahad, S., J.M. Knight, A.O. Naghavi, T. Oplt, C.J. Creighton, C. Shaw, A.L. Benham, J. Kim, B. Soibam, R.A. Harris, et al. 2010. Proinflammatory role for let-7 microRNAS in experimental asthma. Journal of Biological Chemistry 285: 30139–30149. https://doi.org/10.1074/jbc.M110.145698.
Zhao, J., C. Chen, M. Guo, Y. Tao, P. Cui, Y. Zhou, N. Qin, J. Zheng, J. Zhang, and L. Xu. 2016. MicroRNA-7 deficiency ameliorates the pathologies of acute lung injury through elevating KLF4. Frontiers in Immunology 7: 389. https://doi.org/10.3389/fimmu.2016.00389.
Akbas, F., E. Coskunpinar, E. Aynacı, Y. Müsteri Oltulu, and P. Yildiz. 2012. Analysis of serum micro-RNAs as potential biomarker in chronic obstructive pulmonary disease. Experimental Lung Research 38: 286–294. https://doi.org/10.3109/01902148.2012.689088.
Liu, Z., Y. Liu, L. Li, Z. Xu, B. Bi, Y. Wang, and J.Y. Li. 2014. MiR-7-5p is frequently downregulated in glioblastoma microvasculature and inhibits vascular endothelial cell proliferation by targeting RAF1. Tumor Biology 35: 10177–10184. https://doi.org/10.1007/s13277-014-2318-x.
Kuznetsov, A.V., J. Smigelskaite, C. Doblander, M. Janakiraman, M. Hermann, M. Wurm, S.F. Scheidl, R. Sucher, A. Deutschmann, and J. Troppmair. 2008. Survival signaling by C-RAF: Mitochondrial reactive oxygen species and Ca2+ are critical targets. Molecular and Cellular Biology 28: 2304–2313. https://doi.org/10.1128/MCB.00683-07.
Alavi, A., J.D. Hood, R. Frausto, D.G. Stupack, and D.A. Cheresh. 2003. Role of Raf in vascular protection from distinct apoptotic stimuli. Science 301: 94–96. https://doi.org/10.1126/science.1082015.
Galmiche, A., and J. Fueller. 2007. RAF kinases and mitochondria. Biochimica et Biophysica Acta - Molecular Cell Research 1773: 1256–1262. https://doi.org/10.1016/j.bbamcr.2006.10.012.
Reimann, T., D. Büscher, R.A. Hipskind, S. Krautwald, M.L. Lohmann-Matthes, and M. Baccarini. 1994. Lipopolysaccharide induces activation of the Raf-1/MAP kinase pathway. A putative role for Raf-1 in the induction of the IL-1 beta and the TNF-alpha genes. Journal of Immunology. 153: 5740–5749.
Troppmair, J., and U.R. Rapp. 2003. Raf and the road to cell survival: A tale of bad spells, ring bearers and detours. Biochemical Pharmacology 66: 1341–1345. https://doi.org/10.1016/s0006-2952(03)00483-0.
Yang, G., D. Wu, J. Zhu, O. Jiang, Q. Shi, J. Tian, and Y. Weng. 2013. Upregulation of miR-195 increases the sensitivity of breast cancer cells to adriamycin treatment through inhibition of Raf-1. Oncology Reports 30: 877–889. https://doi.org/10.3892/or.2013.2532.
Gao, D., X. Qi, X. Zhang, K. Fang, Z. Guo, and L. Li. 2019. hsa_circRNA_0006528 as a competing endogenous RNA promotes human breast cancer progression by sponging miR-7-5p and activating the MAPK/ERK signaling pathway. Molecular Carcinogenesis 58: 554–564. https://doi.org/10.1002/mc.22950.
Chen, J., K. Fujii, L. Zhang, T. Roberts, and H. Fu. 2001. Raf-1 promotes cell survival by antagonizing apoptosis signal-regulating kinase 1 through a MEK–ERK independent mechanism. Proceedings of the National Academy of Sciences 98: 7783–7788. https://doi.org/10.1073/pnas.141224398.
Peruzzi, F., M. Prisco, M. Dews, P. Salomoni, E. Grassilli, G. Romano, B. Calabretta, and R. Baserga. 1999. Multiple signaling pathways of the insulin-like growth factor 1 receptor in protection from apoptosis. Molecular and Cellular Biology 19: 7203–7215. https://doi.org/10.1128/mcb.19.10.7203.
Salomoni, P., M.A. Wasik, R.F. Riedel, K. Reiss, J.K. Choi, T. Skorski, and B. Calabretta. 1998. Expression of constitutively active Raf-1 in the mitochondria restores antiapoptotic and leukemogenic potential of a transformation-deficient BCR/ABL mutant. Journal of Experimental Medicine 187: 1995–2007. https://doi.org/10.1084/jem.187.12.1995.
Patel, V.J, S. Biswas Roy, H.J. Mehta, M. Joo, and R.T. Sadikot. 2018. Alternative and natural therapies for acute lung injury and acute respiratory distress syndrome. BioMed Research International 2476824. https://doi.org/10.1155/2018/2476824.
Chen, Y., L. Qu, Y. Li, C. Chen, W. He, L. Shen, and R. Zhang. 2022. Glycyrrhizic acid alleviates lipopolysaccharide (LPS)-induced acute lung injury by regulating angiotensin-converting enzyme-2 (ACE2) and caveolin-1 signaling pathway. Inflammation 45: 253–266. https://doi.org/10.1007/s10753-021-01542-8.
ACKNOWLEDGEMENTS
D.P. acknowledges the Department of Science and Technology (DST-India) for the WOS-A grant. D.P. is grateful to Dr. Rajesh Thimmaluappa, JSS Medical College, Mysuru, for providing BEAS-2B cells.
Funding
This work was supported by grants from the Department of Science and Technology (DST-India) (No. SR/ WOS-A/LS-50/2018).
Author information
Authors and Affiliations
Contributions
Conceptualization, investigation and funding acquisition, methodology, writing original draft—D.P. Data curation, formal analysis and software—B.P. Validation, visualization, writing—review and editing—G.K. and R.J. Project administration, resources, supervision—V.B. and P.G.
Corresponding author
Ethics declarations
Ethics Approval
Ethics approval and all experiments were performed in accordance with the guidelines provided by Institute Animal Ethical Committee of CSIR–Central Food Technological Research Institute, Mysore, India (Approval No. CFT/IAEC/204/2020).
Consent for Publication
We guarantee the work described in the manuscript has not been published before or submitted for publication elsewhere.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Peethambaran, D., Puthusseri, B., Kumar, G. et al. miR-7-5p Antagomir Protects Against Inflammation-Mediated Apoptosis and Lung Injury via Targeting Raf-1 In Vitro and In Vivo. Inflammation 46, 941–962 (2023). https://doi.org/10.1007/s10753-023-01782-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10753-023-01782-w