Abstract
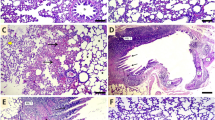
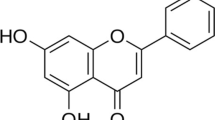
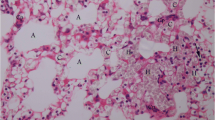
Vanillin, the main constituents of vanillin beans, has been reported to exhibit anti-inflammatory effects. However, the effects of vanillin on the cadmium-induced lung injury are still unclear. Therefore, we assay whether vanillin has potential preventive activity on cadmium-induced lung injury in mice. Mice were given vanillin (5, 10, 20 mg/kg) and treated with cadmium for 7 days. The detection data of vanillin on lung tissue changes were analyzed after the cadmium treatment. The results displayed that vanillin obviously decreased the lung histological alterations and myeloperoxidase (MPO) activity. Vanillin also suppressed the levels of TNF-α, IL-1β, and IL-6 in BALF. Furthermore, vanillin prevented cadmium-induced NF-κB activation and upregulation the expression of tight junction protein ZO-1 and occludin. In addition, vanillin significantly increased the expression of aryl hydrocarbon receptor (AhR), and inhibition of AhR by its agonist could reverse the protective effects of vanillin on cadmium-induced lung injury. To sum up, vanillin could be a potential drug for the treatment of cadmium-induced lung injury.







Similar content being viewed by others
References
Larson-Casey, J.L., L.L. Gu, O. Fiehn, and A.B. Carter. 2020. Cadmium-mediated lung injury is exacerbated by the persistence of classically activated macrophages. Journal of Biological Chemistry 295: 15754–15766.
Ng, C.S., S. Wan, A.A. Arifi, and A.P. Yim. 2006. Inflammatory response to pulmonary ischemia-reperfusion injury. Surgery Today 36: 205–214.
Tao, C., Y.X. Pei, L. Zhang, and Y.M. Zhang. 2020. Microbial communities respond to microenvironments in lungs of mice under simulated exposure to cadmium aerosols. Science of the Total Environment 710: 136300.
Abraham, E., M.A. Matthay, C.A. Dinarello, J.L. Vincent, J. Cohen, S.M. Opal, M. Glauser, P. Parsons, C.J. Fisher Jr., and J.E. Repine. 2000. Consensus conference definitions for sepsis, septic shock, acute lung injury, and acute respiratory distress syndrome: time for a reevaluation. Critical Care Medicine 28: 232–235.
Fein, A.M., and M.G. Calalang-Colucci. 2000. Acute lung injury and acute respiratory distress syndrome in sepsis and septic shock. Critical Care Clinics 16: 289–317.
Ware, L.B. 2006. Pathophysiology of acute lung injury and the acute respiratory distress syndrome. Seminars in Respiratory and Critical Care Medicine 27: 337–349.
Steinberg, K.P., L.D. Hudson, R.B. Goodman, C.L. Hough, P.N. Lanken, R. Hyzy, B.T. Thompson, M. Ancukiewicz, and L. National Heart. 2006. Blood Institute Acute Respiratory Distress Syndrome Clinical Trials N: Efficacy and safety of corticosteroids for persistent acute respiratory distress syndrome. The New England Journal of Medicine 354: 1671–1684.
Garibaldi, R.A. 1985. Epidemiology of community-acquired respiratory tract infections in adults. Incidence, etiology, and impact. The American Journal of Medicine 78: 32–37.
McCullers, J.A. 2014. The co-pathogenesis of influenza viruses with bacteria in the lung. Nature Reviews. Microbiology 12: 252–262.
Lu, Y.C., W.C. Yeh, and P.S. Ohashi. 2008. LPS/TLR4 signal transduction pathway. Cytokine 42: 145–151.
Bhatia, M., and S. Moochhala. 2004. Role of inflammatory mediators in the pathophysiology of acute respiratory distress syndrome. The Journal of Pathology 202: 145–156.
Ibrahim, M.N.M., C.S. Sipaut, and N.N.M. Yusof. 2009. Purification of vanillin by a molecular imprinting polymer technique. Separation and Purification Technology 66: 450–456.
Guo, W.J., B.R. Liu, G.Q. Hu, X.C. Kan, Y.W. Li, Q. Gong, D.W. Xu, H. Ma, Y. Cao, B.X. Huang, S. Fu, and J. Liu. 2019. Vanillin protects the blood-milk barrier and inhibits the inflammatory response in LPS-induced mastitis in mice. Toxicology and Applied Pharmacology 365: 9–18.
Yan, X., D.F. Liu, X.Y. Zhang, D. Liu, S.Y. Xu, G.X. Chen, B.X. Huang, W.Z. Ren, W. Wang, S.P. Fu, and J.X. Liu. 2017. Vanillin protects dopaminergic neurons against inflammation-mediated cell death by inhibiting ERK1/2, P38 and the NF-kappa B signaling pathway. International Journal of Molecular Sciences 18.
Wang, C., Z. Wei, Z. Han, J. Wang, X. Zhang, Y. Wang, Q. Liu, and Z. Yang. 2019. Neutrophil extracellular traps promote cadmium chloride-induced lung injury in mice. Environmental Pollution 254: 113021.
Black, P.H. 2003. The inflammatory response is an integral part of the stress response: implications for atherosclerosis, insulin resistance, type II diabetes and metabolic syndrome X. Brain, Behavior, and Immunity 17: 350–364.
Nathan, C., and A.H. Ding. 2010. Nonresolving Inflammation. Cell 140: 871–882.
Berger, M. 1991. Inflammation in the lung in cystic-fibrosis - a vicious cycle that does more harm than good. Clinical Reviews in Allergy 9: 119–142.
Hessle, C.C., B. Andersson, and A.E. Wold. 2005. Gram-positive and Gram-negative bacteria elicit different patterns of pro-inflammatory cytokines in human monocytes. Cytokine 30: 311–318.
Arnhold, J. 2004. Free radicals - friends or foes? - Properties, functions, and secretion of human myeloperoxidase. Biochemistry-Moscow 69: 4–9.
Wan, F.Y., and M.J. Lenardo. 2010. The nuclear signaling of NF-kappa B: current knowledge, new insights, and future perspectives. Cell Research 20: 24–33.
Silverman, N., R. Zhou, R.L. Erlich, M. Hunter, E. Bernstein, D. Schneider, and T. Maniatis. 2003. Immune activation of NF-kappa B and JNK requires Drosophila TAK1. Journal of Biological Chemistry 278: 48928–48934.
Wang, J.J., C.M. Guo, Z.K. Wei, X.X. He, J.H. Kou, E.S. Zhou, Z.T. Yang, and Y.H. Fu. 2016. Morin suppresses inflammatory cytokine expression by downregulation of nuclear factor-kappa B and mitogen-activated protein kinase (MAPK) signaling pathways in lipopolysaccharide-stimulated primary bovine mammary epithelial cells. Journal of Dairy Science 99: 3016–3022.
Hanada, T., and A. Yoshimura. 2002. Regulation of cytokine signaling and inflammation. Cytokine & Growth Factor Reviews 13: 413–421.
Appel, S., V. Mirakaj, A. Bringmann, M.M. Weck, F. Grunebach, and P. Brossart. 2005. PPAR-gamma agonists inhibit toll-like receptor-mediated activation of dendritic cells via the MAP kinase and NF-kappaB pathways. Blood 106: 3888–3894.
Availability of Data and Materials
The data used to support the findings of this study are available from the corresponding author upon request.
Author information
Authors and Affiliations
Contributions
Xueshibojie Liu, Wei Jiang, and Chengbi Xu designed the experiment; Xueshibojie Liu, Jinghui Yang, Jinqiu Li, and Wei Jiang did the experiment; Chengbi Xu analyzed the data; Xueshibojie Liu wrote the paper; Wei Jiang and Jinqiu Li revised the paper.
Corresponding author
Ethics declarations
Ethics Approval and Consent to Participate
All the experimental protocols in this study were approved by the Institutional Animal Care and Use Committee of Jilin University. All authors consent to participate this research.
Consent for Publication
All authors consent to publish this article.
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Liu, X., Yang, J., Li, J. et al. Vanillin Attenuates Cadmium-Induced Lung Injury Through Inhibition of Inflammation and Lung Barrier Dysfunction Through Activating AhR. Inflammation 44, 2193–2202 (2021). https://doi.org/10.1007/s10753-021-01492-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10753-021-01492-1