Abstract
Asthma is an inflammatory airway disease wherein bronchoconstriction, airway inflammation, and airway obstruction during asthma attacks are the main problems. It is recognized that imbalance of Th1/Th2 and Th17/Treg is a critical factor in asthma pathogenesis. Manipulation of these with signaling molecules such as mTOR, PI3K, Akt, and MyD88 can control asthma. Mouse model of allergic asthma was produced and treated with ketamine, metformin, metformin and ketamine, triciribine, LY294002, and torin2. MCh challenge test, BALf's Eos Count, the IL-4, 5, INF-γ, eicosanoid, total IgE levels were determined. The MUC5a, Foxp3, RORγt, PI3K, mTOR, Akt, PU.1, and MyD88 gene expressions and histopathology study were done. Asthma groups that were treated with all six components had reduced Penh value, total IgE, IL-4 and IL-5 levels, MUC5a, RORγt, MyD88 and mTOR expression, goblet cell hyperplasia, and mucus hyper-secretion. The eosinophil percentage and Cys-LT level were decreased by metformin and ketamine, triciribine, LY294002, and torin2. The level of IFN-γ was increased in triciribine, LY294002, and torin2. Metformin, metformin and ketamine, triciribine, LY294002, and torin2 reduced Akt and PI3K expression, peribronchial and perivascular inflammation, and increased expression of Foxp3. Torin2 had an effect on PU.1 expression. Inhibition of PI3K/AKT/mTOR and TLR4/MyD88/NF-κB signaling with targeted molecules can attenuate asthma pathology and play an important role in airways protection.
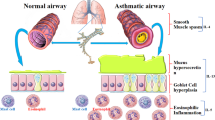
Graphical Abstract










Similar content being viewed by others
Data availability
Not Applicable.
Abbreviations
- AB:
-
Alcian blue
- AHR:
-
Airway hyperresponsiveness
- AKT:
-
A serine/threonine-specific protein kinase (protein kinase B (PKB))
- AMPK:
-
5-adenosine monophosphate-activated protein kinase
- ATF:
-
Transcription factor
- ATP:
-
Adenosine triphosphate
- BALf:
-
Bronchoalveolar lavage fluid
- Cys-LT:
-
Cysteinyl leukotriene
- DC:
-
Dendritic cell
- DNA:
-
Deoxyribonucleic acid
- EOS:
-
Eosinophil
- Foxp3:
-
Fork head/winged helix
- GAPDH:
-
Glyceraldehyde 3-phosphate dehydrogenase
- HDAC:
-
Histone deacetylase
- HDM:
-
House dust mite
- H&E:
-
Hematoxylin and Eosin
- HIF:
-
Hypoxia-induced factor
- HMGB:
-
High mobility group box
- HSF:
-
Heat shock factor
- Flt3L:
-
fms-like tyrosine 3 kinase ligand
- Ig:
-
Immunoglobulin
- IGF:
-
Insulin-like growth factor
- IGF1R:
-
IGF-1 receptor
- IL:
-
Interleukin
- IP:
-
Intraperitoneal
- IT:
-
Inhalation administration
- KLF:
-
Krüppel-like factor
- MCh:
-
Methacholine
- mTOR:
-
Serine/threonine-protein kinase mammalian target of rapamycin
- MyD88:
-
Myeloid differentiation primary response 88
- NF-κB:
-
Nuclear factor kappa-light-chain-enhancer of activated B cells
- OVA:
-
Ovalbumin
- PAS:
-
Periodic Acid Schiff
- PBS:
-
Phosphate-buffered saline
- PI3K:
-
Phosphatidylinositol-3-kinase
- Qrt:
-
Quantitative real-time
- RORγt:
-
Nuclear orphan receptor γt
- ROS:
-
Reactive oxygen species
- STAT:
-
Signal transducer and activator of transcription
- T-bet:
-
T-box expressed in T cells
- TGF:
-
Transforming growth factor
- Th:
-
Lymphocyte T helper
- TLR:
-
Toll-like receptor
- TNF:
-
Tumor necrosis factor
- Treg:
-
Lymphocyte T regulatory
References
Athari, S.S. 2019. Targeting cell signaling in allergic asthma. Signal Transduction and Targeted Therapy 4 (1): 1–19.
Mozaffarinya, M., A. Reza Shahriyari, M. Karim Bahadori, A. Ghazvini, S. Shamsadin Athari, and G. Vahedi. 2019. A data-mining algorithm to assess key factors in asthma diagnosis. Revue Française d'Allergologie 59 (7): 487–492.
Yu-sen Chai, Shi-hui Lin, Mu Zhang, Liangyong Deng, Yanqing Chen, Ke Xie, Chuan-jiang Wang, Fang Xu. IL-38 is a biomarker for acute respiratory distress syndrome in humans and down-regulates Th17 differentiation in vivo. Clinical Immunology 2020; 210:108315
Hu, Ying, Zhiqiang Chen, Jing Zeng, Shouyan Zheng, Liujuan Sun, Li Zhu, and Wei Liao. 2020. Th17/Treg imbalance is associated with reduced indoleamine 2,3 dioxygenase activity in childhood allergic asthma. Allergy, Asthma and Clinical Immunology 16: 61.
Yang, Zhao, Xiangsheng Li, Zhenzhen Xu, Lifang Hao, Yanfen Zhang, and Zhongcheng Liu. 2019. PI3K-AKT-mTOR signaling pathway: the intersection of allergic asthma and cataract. Pharmazie 74 (10): 598–600.
Shao, Youyou, Lei Chong, Peng Lin, Haiyan Li, Lili Zhu, Qiuping Wu, and Changchong Li. 2018. MicroRNA-133a alleviates airway remodeling in asthtama through PI3K/AKT/mTOR signaling pathway by targeting IGF1R. Journal of Cellular Physiology: 1–13.
Cheng, H., M. Shcherba, G. Pendurti, Y. Liang, B. Piperdi, and R. Perez-Soler. 2014. Targeting the PI3K/AKT/mTOR pathway: potential for lung cancer treatment. Lung Cancer Management 3: 67–75.
Putilin, Denis Anatolievich, Sergey Yuryevich Evchenko, Larisa Yaroslavivna Fedoniuk, Olexandr Stepanovich Tokarskyy, Oleksandr Mikhailovich Kamyshny, Liudmyla Mikhailivna Migenko, Serhiy Mikhailovich Andreychyn, Iryna Ihorivna Hanberher, and Tetyana Oleksandrivna Bezruk. 2020. The influence of metformin to the transcriptional activity of the mTOR and FOX3 genes in parapancreatic adipose tissue of streptozotocin-induced diabetic rats. Journal of Medicine and Life 13 (1): 50–55.
Zou, Hongyun, Li-Xia Wang, Muzi Wang, Cheng Cheng, Shuai Li, Qiying Shen, Lei Fang, and Rongyu Liu. 2019. MTOR-mediated autophagy is involved in the protective effect of ketamine on allergic airway inflammation. Journal of Immunology Research: 5879714, 11 pages.
Yanli Zhang, Ying Jing, Junying Qiao, Bin Luan, Xiufang Wang, Li Wang & Zhe Song. Activation of the mTOR signaling pathway is required for asthma onset. Scientific Reports 2017; 7:4532
Athari, Seyyed Shamsadin, Zahra Pourpak, Gert Folkerts, Johan Garssen, Mostafa Moin, Ian M. Adcock, Masoud Movassaghi, Mehdi Shafiee Ardestani, Seyed Mohammad Moazzeni, and Esmaeil Mortaz. 2016. Conjugated alpha-alumina nanoparticle with vasoactive intestinal peptide as a nano-drug in treatment of allergic asthma in mice. European Journal of Pharmacology 791: 811–820.
Ruan, Qingguo, Vasumathi Kameswaran, Yukiko Tone, Li Li, Hsiou-Chi Liou, Mark I. Greene, Masahide Tone, and Youhai H. Chen. 2009. Development of Foxp3+ regulatory t cells is driven by A c-Rel enhanceosome. Immunity. 31 (6): 932–940.
Ivanov, Ivaylo I., Brent S. McKenzie, Zhou Liang, Carlos E. Tadokoro, Alice Lepelley, Juan J. Lafaille, Daniel J. Cua, and Dan R. Littman. 2006. The orphan nuclear receptor RORγt directs the differentiation program of proinflammatory IL-17+ T Helper Cells. Cell 126 (6): 1121–1133.
Marinov, Marin, Algirdas Ziogas, Olivier E. Pardo, Liwen Terence Tan, Tony Dhillon, Francesco A. Mauri, Heidi A. Lane, Nicholas R. Lemoine, Uwe Zangemeister-Wittke, Michael J. Seckl, and Alexandre Arcaro. 2009. AKT/mTOR Pathway activation and BCL-2 family proteins modulate the sensitivity of human small cell lung cancer cells to RAD001. Clinical Cancer Research 15 (4): 1277–1287.
Liyuan, Liu, Yingxiong Wang, and Qiubo Yu. 2014 May. The PI3K/Akt signaling pathway exerts effects on the implantation of mouse embryos by regulating the expression of RhoA. International Journal of Molecular Medicine 33 (5): 1089–1096.
Liu, Qing, corresponding author Yongming Zhang, Songzhu Yang, Yanfang Wu, Jiantao Wang, Weiwei Yu, and Yanguo Liu. 2017. PU.1-deficient mice are resistant to thioacetamide-induced hepatic fibrosis: PU.1 finely regulates Sirt1 expression via transcriptional promotion of miR-34a and miR-29c in hepatic stellate cells. Bioscience Reports 37 (6): BSR20170926.
Into, Takeshi, Shumpei Niida, and Ken-ichiro Shibata. 2018. MyD88 signaling causes autoimmune sialadenitis through formation of high endothelial venules and upregulation of LTβ receptor-mediated signaling. Scientific Reports 8: 14272.
Thomson, Angus W., Heth R. Turnquist, and Giorgio Raimondi. 2009. Immunoregulatory functions of mTOR inhibition. Nature Reviews. Immunology 9 (5): 324–337.
Lamming, Dudley W. 2016. Inhibition of the mechanistic target of rapamycin (mTOR)-rapamycin and beyond. Cold Spring Harbor Perspectives in Medicine 6: a025924.
Fredriksson, Karin, Jill A. Fielhaber, Jonathan K. Lam, Xianglan Yao, Katharine S. Meyer, Karen J. Keeran, Gayle J. Zywicke, Qu Xuan, Zu-Xi Yu, Joel Moss, Arnold S. Kristof, and Stewart J. Levine. 2012. Paradoxical effects of rapamycin on experimental house dust mite-induced asthma. PLoS One 7 (5): e33984.
Hua, Wen, Hui Liu, Li-Xia Xia, Bao-Ping Tian, Hua-Qiong Huang, Zhi-Yang Chen, Zhen-Yu Ju Wen Li, Zhi-Hua Chen, and Hua-Hao Shen. 2015. Rapamycin inhibition of eosinophil differentiation attenuates allergic airway inflammation in mice. Respirology 20: 1055–1065.
Haczku, Angela, Andrew Alexander, Peter Brown, Basil Assoufi, Li Baiqing, A. Barry Kay, and Christopher Corrigan. 1994. The effect of dexamethasone, cyclosporine, and rapamycin on T-lymphocyte proliferation in vitro: comparison of cells from patients with glucocorticoid-sensitive and glucocorticoid-resistant chronic asthma. The Journal of Allergy and Clinical Immunology 93 (2): 510–519.
Elizabeth, M. 2011. Mushaben, Elizabeth L. Kramer, Eric B. Brandt, Gurjit K. Khurana Hershey, and Timothy D. Le Cras. Rapamycin attenuates airway hyperreactivity, goblet cells, and IgE in experimental allergic asthma. Journal of Immunology 187 (11): 5756–5763.
Sreenivas, P. 2017. Veeranki. Metformin use and asthma: further investigations. Respirology 22: 203–204.
Li, C.Y., S.R. Erickson, and C.H. Wu. 2016. Metformin use and asthma outcomes among patients with concurrent asthma and diabetes. Respirology 21: 1210–1218.
Park, C.S., B.R. Bang, H.S. Kwon, K.A. Moon, T.B. Kim, K.Y. Lee, H.B. Moon, and Y.S. Cho. 2012. Metformin reduces airway inflammation and remodeling via activation of AMP-activated protein kinase. Biochemical Pharmacology 84: 1660–1670.
Li, Chun-Yi, Steven R. Erickson, and Chung-Hsuen Wu. 2016. Metformin use and asthma outcomes among patients with concurrent asthma and diabetes. Respirology 21: 1210–1218.
Gaoa, Junling, Juntao Yuana, Qiao'e Wangb, Tong Leia, Xiyue Shena, Bingqing Cuia, Fang Zhanga, Wenjun Dinga, and Zhongbing Lu. 2020. Metformin protects against PM2.5-induced lung injury and cardiac dysfunction independent of AMP-activated protein kinase α2. Redox Biology 28: 101345.
Kent Denmark, T., Heather A. Crane, and Lance Brown. 2006. Ketamine to avoid mechanical ventilation in severe pediatric asthma. The Journal of Emergency Medicine. 30 (2): 163–166.
Elkoundi, Abdelghafour, Aziza Bentalha, Alae El Koraichi, and Salma Ech-Cherif El Kettani. 2018. Nebulized ketamine to avoid mechanical ventilation in a pediatric patient with severe asthma exacerbation. American Journal of Emergency Medicine 36 (4): 734.e3–734.e4.
Shlamovitz, Gil Z., and Tracy Hawthorne. 2011. Intravenous ketamine in a dissociating dose as a temporizing measure to avoid mechanical ventilation in adult patient with severe asthma exacerbation. The Journal of Emergency Medicine 41 (5): 492–494.
Hea, Zu-hong, Sheng-yu Zoua, Ming Lia, Fu-ling Liaoa, F. Xia Wua, Hai-ying Suna, Xue-yan Zhaoa, Yu-juan Hua, Dan Lia, Xiao-xiang Xug, Sen Chena, Yu Suna, Ren-jie Chaib, and Wei-jia Kong. 2020. The nuclear transcription factor FoxG1 affects the sensitivity of mimetic aging hair cells to inflammation by regulating autophagy pathways. Redox Biology 28: 101364.
Ivanov, Ivaylo I., Brent S. McKenzie, Zhou Liang, Carlos E. Tadokoro, Alice Lepelley, Juan J. Lafaille, Daniel J. Cua, and Dan R. Littman. 2006. The orphan nuclear receptor RORgt directs the differentiation program of proinflammatory il-17+ T Helper Cells. Cell 126: 1121–1133.
Lee, Gap Ryol. 2018. The balance of Th17 versus Treg cells in autoimmunity. International Journal of Molecular Sciences 19: 730.
Qiua, Yu-ying, Yan Wub, Min-jie Lina, Tao Bianb, Yong-long Xiaoa, and Chu Qin. 2019. LncRNA-MEG3 functions as a competing endogenous RNA to regulate Treg/Th17 balance in patients with asthma by targeting microRNA-17/ RORγt. Biomedicine & Pharmacotherapy 111: 386–394.
Athari, Seyyed Shamsadin, Seyyede Masoume Athari, Fateme Beyzay, Masoud Movassaghi, Esmaeil Mortaz, and Mehdi Taghavi. 2017. Critical role of Toll-like receptors in pathophysiology of allergic asthma. European Journal of Pharmacology 808: 21–27.
Helala, Manar G., Nermeen A. Megahedb, and Ahmed G. Abd Elhameeda. 2019. Saxagliptin mitigates airway inflammation in a mouse model of acute asthma via modulation of NF-kB and TLR4. Life Sciences 239: 117017.
Shang, Liqun, Li Wang, Xiaolan Shi, Ning Wang, Long Zhao, Jing Wang, and Cuicui Liu. 2020. HMGB1 was negatively regulated by HSF1 and mediated the TLR4/MyD88/NF-κB signal pathway in asthma. Life Sciences 241: 117120.
Yun, Chenxia, Ming Chang, Guanghan Houd, Taijin Lana, Hebao Yuane, Zhiheng Suf, Dan Zhuf, Weiping Liangb, Qiaofeng Lib, Hongyan Zhug, Jian Zhangc, Yi Luc, Jiagang Dengh, and Hongwei Guo. 2019. Mangiferin suppresses allergic asthma symptoms by decreased Th9 and Th17 responses and increased Treg response. Molecular Immunology 114: 233–242.
Hajimohammadi, B., S.M. Athari, M. Abdollahi, G. Vahedi, and S.S. Athari. 2020. Oral administration of acrylamide worsens the inflammatory responses in the airways of asthmatic mice through agitation of oxidative stress in the lungs. Frontiers in Immunology 11: 1940.
Author information
Authors and Affiliations
Contributions
BM, SSA, EMN, and LZ participated in the design, lab testing, analysis, and drafting of the manuscript. LZ and SSA supervised the study.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Animal investigations and studied methods have been approved by the ethical committee of animal house of ix.med.vet.dep, 2021 (No. IX.MED.VET.DEP.REC.2021.290099.0).
Consent for publication
Not Applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ma, B., Athari, S.S., Mehrabi Nasab, E. et al. PI3K/AKT/mTOR and TLR4/MyD88/NF-κB Signaling Inhibitors Attenuate Pathological Mechanisms of Allergic Asthma. Inflammation 44, 1895–1907 (2021). https://doi.org/10.1007/s10753-021-01466-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10753-021-01466-3