Abstract
A controlled local inflammatory response is essential for adequate fracture healing. However, the current literature suggests that local and systemic hyper-inflammatory conditions after major trauma induce increased influx of neutrophils into the fracture hematoma (FH) and impair bone regeneration. Inhibiting neutrophil chemotaxis towards the FH without compromising the hosts’ defense may therefore be a target of future therapies that prevent impairment of fracture healing after major trauma. We investigated whether chemotaxis of neutrophils towards the FH could be studied in vitro. Moreover, we determined whether chemotaxis of neutrophils towards the FH was mediated by the CXCR1, CXCR2, FPR, and C5aR receptors. Human FHs were isolated during an open reduction internal fixation (ORIF) procedure within 3 days after trauma and spun down to obtain the fracture hematoma serum. Neutrophil migration towards the FH was studied using Ibidi™ Chemotaxis3D μ-Slides and image analysis of individual neutrophil tracks was performed. Our study showed that the human FH induces significant neutrophil chemotaxis, which was not affected by blocking CXCR1 and CXCR2. In contrast, neutrophil chemotaxis towards the FH was significantly inhibited by chemotaxis inhibitory protein of Staphylococcus aureus (CHIPS), which blocks FPR and C5aR. Blocking only C5aR with CHIPSΔ1F also significantly inhibited neutrophil chemotaxis towards the FH. Our finding that neutrophil chemotaxis towards the human FH can be blocked in vitro using CHIPS may aid the development of therapies that prevent impairment of fracture healing after major trauma.
Similar content being viewed by others
INTRODUCTION
Fracture healing starts with an inflammatory phase during which leukocytes infiltrate the blood collection surrounding the fracture site [1, 2]. Animal studies suggest that this blood collection, which is generally referred to as fracture hematoma (FH), forms a reservoir of essential factors and cells that regulate downstream processes of bone repair. This is illustrated by the finding that transplantation of the FH into muscle tissue induced ectopic bone formation and angiogenesis in animal models [3, 4]. Moreover, removal or repetitive irrigation of the FH impaired fracture healing in rats [5, 6].
Although controlled local inflammation is essential for adequate fracture healing [7], several animal studies have also shown that both local and systemic “hyper-inflammatory” conditions impair bone regeneration. For instance, injection of beta-glucan into the fracture site induces local hyper-inflammation and impairs fracture healing in rats [8]. Moreover, intraperitoneal injection of lipopolysaccharides in rats induces systemic inflammation and negatively affects the outcome of bone repair [9]. In addition, blunt chest injury, which is a model of trauma-induced systemic inflammation, also impairs fracture healing in rats [10].
It is well known that severely injured patients have an increased risk of developing impaired fracture healing [11, 12]. This not only has a significant impact on quality of life, but also carries a substantial economical burden to society [13]. Based on the abovementioned animal studies, we hypothesized that the systemic immune response after major trauma contributes to the high incidence of impaired fracture healing in multitrauma patients [1, 11]. The underlying mechanism remains unclear. However, experimental studies suggest that major trauma pre-activates neutrophils and induces increased influx of neutrophils towards sites of inflammation, such as the fracture hematoma [10, 14, 15], and impairs bone healing.
Such a pathological role of neutrophils was supported by the finding that depletion of neutrophils improved the outcome of bone repair in rats [16, 17]. However, systemic depletion of neutrophils would significantly compromise the hosts’ defense against pathogens.
Therefore, we tried to identify neutrophil chemoattractants within the sterile FH that may be blocked in the future without affecting chemotaxis of neutrophils towards sites of infection. As a first step, we tested whether neutrophil chemotaxis towards the human FH could be studied in vitro. Furthermore, we explored whether neutrophil chemotaxis towards the FH is mediated by IL-8 receptors CXCR1 and CXCR2, formylated peptide receptors (FPR), and complement receptor C5aR.
MATERIALS AND METHODS
Isolation of Neutrophils
Blood from anonymous healthy donors was acquired from the blood bank “Mini Donor Dienst” of the University Medical Center Utrecht after written informed consent was obtained. Neutrophils were isolated from peripheral blood, as has been described previously [18] and is summarized here. Briefly, 9 ml of blood was drawn into a sterile vacuum container with sodium citrate as anti-coagulant. The blood was diluted 1:1 in phosphate-buffered saline (PBS) at room temperature. The diluted peripheral blood was pipetted onto 15 ml of Ficoll-Paque (Pharmacia, Uppsala, Sweden) and centrifuged for 20 min at 900g. After centrifugation, the plasma, Ficoll, and mononuclear fraction were removed. The remaining erythrocytes and granulocytes were resuspended in 50 ml isotonic ice-cold ammonium chloride solution (NH4Cl) containing 155 mM NH4Cl, 10 mM KHCO3, and 0.1 mM EDTA (pH 7.2) and incubated on ice for 20 min. Subsequently, the cell suspension was centrifuged, the supernatant was removed, and the cell pellet was resuspended in 20 ml of NH4Cl. After centrifugation, the cell pellet was resuspended in HEPES3+ (20 mM HEPES, 132 mM NaCl, 6.0 mM KCl, 1.0 mM MgSO4, 1.2 mM KH2PO4, supplemented with 5.0 mM glucose, 1.0 mM CaCl2, and 0.5% (w/v) human serum albumin) and centrifuged again. The supernatant was removed and the cell pellet was resuspended in HEPES3+. Cells were counted using the Cell-Dyn® 1800 (Abbott Laboratories, Abbot Park, IL, USA) and diluted in HEPES3+ to concentrations needed during the experimental conditions (3.2 × 106 cells/ml) and stored on ice until further use.
Isolation of Human Fracture Hematoma Serum
Human fracture hematomas (FHs) were isolated during open reduction internal fixation (ORIF) procedures within 3 days after trauma from patients with closed fractures and without relevant comorbidity and collected in sterile plastic containers. The blood clot was isolated from the fracture site, which is generally required during an ORIF procedure in order to allow adequate reduction of the fracture and placement of fixation materials. FH was deemed residual tissue and could therefore be collected without obtaining informed consent, unless the patient explicitly refused (opt-out method). This procedure is formalized in our hospital and therefore approval by our local ethics committee was not required. All samples were stripped of identifiers and fully anonymized. The serum of the FHs was obtained by centrifugation (5 min, 2300 rcf) of the FH within 1 h after isolation. The serum was aliquoted and stored at − 20 °C until further use. The FH sera of different donors were used for each experimental condition and these sera were not pooled. Thirty microliters of FH was used for each experiment. There was no significant difference in chemotaxis towards fresh or frozen FH. The variation in neutrophil response towards the FH of different donors is depicted in Fig. 2c.
Chemotaxis Assay
The Ibidi™ Chemotaxis3D μ-Slide was used to analyze neutrophil chemotaxis towards the FH serum in a three-dimensional, porous in vitro environment (IBIDI, Martinsried, Germany). Setup and data analysis of the Ibidi™ Chemotaxis3D μ-Slide have been described previously by other authors [19]. The Ibidi™ Chemotaxis3D μ-Slide is a chemotaxis chamber that enables the investigator to create time-lapse images and videos of cell migration.
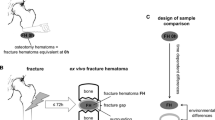
Three microliters of ultra-pure human fibrinogen (25 mg/ml FIB3 obtained from Kordia, Leiden, the Netherlands) and 3.75 μl of thrombin (20 U/ml in PBS, purchased from Sigma, St. Louis, MO, USA) were added to the 30 μl of neutrophil suspension (final concentrations: fibrinogen 2.04 mg/ml; thrombin 2.04 U/ml; neutrophils 2.45 × 106 cells/ml). Six microliters of this neutrophil/fibrinogen/thrombin suspension, containing approximately 1.5 × 104 neutrophils, was pipetted into each center channel of Ibidi™ Chemotaxis3D μ-Slide (observation area) using round tips according to the manufacturer’s protocol (Fig. 1a). This fibrin gel was allowed to solidify for 10 min at room temperature. HEPES3+ was pipetted into the right (C0) chamber and each experimental condition was pipetted into the left (C100) chamber (Fig. 1a).
a Analysis of neutrophil chemotaxis towards the fracture hematoma using the Ibidi™ Chemotaxis3D μ-Slide. A neutrophil/fibrinogen/thrombin suspension was injected into the observation area of the slide. After the fibrin gel solidified, HEPES3+ was injected into the C0 chamber. All experimental conditions were injected into the C100 chamber after which neutrophil chemotaxis was analyzed with time-lapse microscopy and cell tracking software. b Representative example of neutrophil migration towards the fracture hematoma and towards HEPES3+. The red lines are Euclidean distances, which are the shortest distances between each beginning and endpoint of all neutrophils that were analyzed. Vector speed was defined as the mean Euclidean distances of all neutrophils that were analyzed divided by imaging time.
A gradient of chemoattractants was rapidly established over the center channel (observation area). The slides were immediately placed in a pre-warmed microscopy chamber (37 °C, Heidolph Instruments inkubator 1000) onto an automated stage (Märzhäuser Wetzlar GmbH & Co., Wetzlar-Steindorf, Germany).
Time-lapse point revisiting microscopy (Quantimet 570C, DXMRE microscope, PL fluotar ×5 low power objective lens, Leica, Heidelberg, Germany) was used to track the movement of neutrophils through the fibrin gel. Sequences consisted of 100 images per spot with a maximum of 3 revisited spots. The time-lapse interval was typically 15–25 s. Consecutive images were converted into a movie using ImageJ (version 1.46r, Public Domain). OPTIMAS software (version 6.51, Media Cybernetics, Inc.) was used to derive trajectory plots and to quantify various parameters that describe chemotactic or chemokinetic responses which have been described previously [19]. Neutrophil chemotaxis was measured using mean vector speed, which is the Euclidean distance between starting point and end point of all neutrophils that were analyzed (Fig. 1b) divided by imaging time.
Experimental Conditions
N-Formyl-methionyl-leucyl-phenylalanine (fMLF) (Sigma-Aldrich, St. Louis, MO, USA), and recombinant human IL-8 (PeproTech EC Ltd., Rocky Hill, NJ, USA) were diluted in HEPES3+ (10−7 M and 50 ng/ml, respectively) and used as positive controls, since these factors are well-known chemoattractants for neutrophils [20, 21]. HEPES3+ was used as a negative control. CXCR1 and CXCR2 were simultaneously blocked on neutrophils using blocking antibodies αCXCR1 (Monoclonal Mouse IgG2A Clone # 42705, 500 μg/ml, R&D Systems®, Abingdon, UK) and αCXCR2 (Monoclonal Mouse IgG2A Clone # 48311 500 μg/ml, R&D Systems®, Abingdon, UK). Additionally, the C5aR and FPRs were simultaneously blocked using chemotaxis inhibitory protein of Staphylococcus aureus (CHIPS) which was donated and manufactured by the Department of Medical Microbiology, University Medical Center Utrecht, the Netherlands, as described by de Haas et al. [22, 23]. In addition, a CHIPS mutant lacking the first N-terminal amino acid was used (CHIPSΔ1F), which has impaired or absent FPR but still intact C5aR-blocking activity [24]. The isolated neutrophils were incubated with αCXCR1 and αCXCR2 or CHIPS and CHIPSΔ1F for 30 min on ice in 30 μl of solution (final concentrations: neutrophils 3.0 × 106 cells/ml; αCXCR1/2 20 μg/ml; CHIPS and CHIPSΔ1F 10 μg/ml). After blocking the CXCR1 and CXCR2 receptors, neutrophil chemotaxis towards IL-8 and the FH was studied. After blocking the C5aR and FPR receptors with CHIPS, neutrophil chemotaxis towards fMLF and the FH was studied. Subsequently, neutrophil chemotaxis towards the FH was studied after blocking C5aR with CHIPSΔ1F. We did not use technical duplicates or triplicates with the same FH/neutrophil donor combinations analyzed at the same time point. Figure 2 therefore depicts the pooled data of single experiments with different FH/neutrophil donor combinations analyzed at different time points.
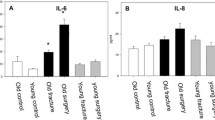
a Neutrophil chemotaxis towards IL-8 with and without blocking CXCR1 and CXCR2. IL-8 induced significant neutrophil chemotaxis compared to HEPES3+. Blocking CXCR1 and CXCR2 significantly inhibited migration towards IL-8. ***p < 0.001. b Migration of neutrophils through a 3D fibrin gel towards fMLF with and without blocking FPR with CHIPS. There was significant chemotaxis of neutrophils towards fMLF compared to the negative control HEPES3+. Blocking the FPR receptors with CHIPS significantly inhibited migration towards fMLF. ***p < 0.001. c Migration of neutrophils through a 3D fibrin gel towards the fracture hematoma (donor variation). Neutrophil migration towards the FH is depicted for each neutrophil/FH donor combination. Fourteen neutrophil donors were combined with 8 FH donors (14 neutrophil/FH donor combinations). A distinct icon is used to plot each FH donor. We were unable to find a significant difference in neutrophil migration towards the FH between different FH donors. d Migration of neutrophils through a 3D fibrin gel towards the fracture hematoma (dose response). Neutrophils significantly migrated towards the FH, even after diluting the FH 1:10, 1:20, and 1:50 in HEPES 3+. The 1:500 diluted FH did not induce significant neutrophil chemotaxis. ***p < 0.001 and *p < 0.05 compared to HEPES3+. e Neutrophil chemotaxis towards the fracture hematoma serum with and without blocking CXCR1 and CXCR2. Chemotaxis towards the FH was not significantly inhibited by blocking CXCR1 and CXCR2. ***p < 0.001. f Neutrophil chemotaxis towards the fracture hematoma with and without blocking C5aR and FPR with CHIPS and blocking C5aR with CHIPSΔ1F. CHIPS and CHIPSΔ1F significantly inhibited neutrophil chemotaxis towards the FH. ***p < 0.001, **p < 0.01.
Statistical Analysis
GraphPad Prism version 5.00 was used for all statistical analyses. All experimental conditions were compared using an ANOVA with a Bonferroni multiple comparison post hoc test. Multiple dilutions of the FH were compared to HEPES3+ using an ANOVA with Dunnett’s multiple comparison test. A p value < 0.05 was considered statistically significant. p values are described in Fig. 2 as * (< 0.05), ** (< 0.01), and *** (< 0.001).
RESULTS
Chemotaxis of Human Neutrophils Towards fMLF and IL-8
As a control study, we first determined whether neutrophil chemotaxis towards interleukin-8 (IL-8) and fMLF could be studied with the Ibidi™ Chemotaxis3D μ-Slides, since these two factors are well-known neutrophil chemoattractants. Neutrophil chemotaxis towards HEPES3+ (negative control), IL-8, and fMLF is depicted in Fig. 2a, b, respectively. When compared to HEPES3+, there was a significant increase in migration towards IL-8 (mean: 0.4 vs 6.1 μm/min, n = 21 vs 8, p < 0.001) and fMLF (mean: 0.4 vs 3.6 μm/min, n = 21 vs 14, p < 0.001).
Blocking Chemotaxis of Human Neutrophils Towards IL-8 and fMLF
Chemotaxis of neutrophils towards IL-8 is dependent on the CXCR1 and CXCR2 receptors. Blocking these two receptors induced a significant decrease in vector speed (mean: 6.1 vs 2.6 μm/min, n = 8 vs 4, p < 0.001 without and with blocking the CXCR1 and CXCR2 receptors) as depicted in Fig. 2a. CHIPS specifically binds to the formylated peptide receptors (FPRs) and C5a receptor (C5aR). Chemotaxis of neutrophils towards fMLF was significantly inhibited by CHIPS (mean: 3.6 vs 0.4 μm/min, n = 14 vs 12, p < 0.001, without and with CHIPS), as depicted in Fig. 2b.
Chemotaxis of Neutrophils Towards the Human Fracture Hematoma
Neutrophils exhibited very potent chemotaxis towards the human fracture hematoma (FH) serum in vitro. The variation in neutrophil migration towards the FH for each neutrophil/FH donor combination (n = 14) is depicted in Fig. 2c. A dose response is depicted in Fig. 2d. The vector speed of neutrophils towards the FH was significantly greater when compared to migration towards HEPES3+ (mean: 5.8 vs 0.4 μm/min, n = 14 vs 21, p < 0.001, respectively). Neutrophil chemotaxis towards the FH remained significant when the FH was diluted in HEPES3+ 1:10 (mean: 3.7 vs 0.4 μm/min, n = 4 vs n = 21, p < 0.001), 1:20 (mean: 2.7 vs 0.4 μm/min, n = 5 vs 21, p < 0.001), and 1:50 (mean: 1.6 vs 0.4 μm/min, n = 8 vs 21, p < 0.05). When the FH was diluted 1:500, no significant chemotaxis could be observed (mean: 0.4 vs 0.4 μm/min, n = 6 vs 21).
Chemotaxis of Neutrophils Towards the Fracture Hematoma After Blocking the CXCR1, CXCR2, FPR, and C5aR Receptors
Blocking the CXCR1 and CXCR2 receptors did not significantly affect neutrophil chemotaxis towards the FH (mean: 5.8 vs 6.1 μm/min, n = 14 vs 8, without and with blocking the CXCR1 and CXCR2 receptors). There was still significant chemotaxis towards the FH after blocking these receptors compared to HEPES3+ (mean: 6.1 vs 0.4 μm/min, p < 0.001) as depicted in Fig. 2e. CHIPS, which blocks FPR and C5aR, significantly inhibited neutrophil chemotaxis towards the FH (mean: 5.8 vs 3.3 μm/min, n = 14 vs 4, p < 0.001 without and with CHIPS, Fig. 2f). In addition, CHIPSΔ1F, which only blocks C5aR, also induced a significant decrease in neutrophil chemotaxis towards the FH (mean: 5.8 vs 4.0 μm/min, n = 14 vs 5, p < 0.01 without and with CHIPSΔ1F, Fig. 2f).
DISCUSSION
The current literature suggests that increased influx of neutrophils into the fracture hematoma (FH) during hyper-inflammatory conditions impairs fracture healing after major trauma [1, 25]. Future therapies that inhibit influx of neutrophils into the FH without compromising the hosts’ defense against pathogens may therefore prevent impairment of bone healing in multitrauma patients. Our study shows that chemotaxis of neutrophils towards the FH can be studied in vitro with Ibidi™ Chemotaxis3D μ-Slides. We found that serum from the human FH significantly induces neutrophil chemotaxis, which was not affected by blocking the CXCR1 and CXCR2 receptors (Fig. 2e). In contrast, CHIPS induced a significant decrease in neutrophil chemotaxis towards the human FH in vitro (Fig. 2f). CHIPS is an exoprotein produced by several strains of S. aureus and is a potent inhibitor of neutrophil and monocyte chemotaxis towards C5a and formylated peptides like fMLF [23]. It is known that tissue injury induces complement activation and release of C5a [15, 26], as well as release of formylated peptides from mitochondria into the circulation [27]. CHIPS exclusively binds directly to the C5aR and FPR1 and FPR2 receptors, thereby preventing their natural ligands from activating these receptors [23, 28]. We additionally used a CHIPS mutant lacking the first N-terminal amino acid (CHIPSΔ1F), which has impaired or absent FPR but still intact C5aR-blocking activity [24]. Our data shows that blocking C5aR with CHIPSΔ1F also significantly inhibits neutrophil chemotaxis towards the FH (Fig. 2f). Previous studies have shown that systemic antagonism of the C5aR improves fracture healing after major trauma in rats [15]. It is tempting to speculate that systemic C5aR antagonism prevents increased influx of neutrophils into the FH and thereby reduces the deleterious effect of major trauma on fracture healing.
In our in vitro experiments, we were unable to completely block neutrophil chemotaxis towards the FH using CHIPS or CHIPSΔ1F. One possible explanation for this effect is that the concentrations of blocking antibodies were insufficient to completely block all receptors. Also, several additional neutrophil chemoattractants may be present within the FH that do not exert their effect through CXCR1/2, FPR, or C5aR. Neutrophils possess several receptors that detect chemoattractants, such as chemokines, complement components, and several other chemotactic lipids and peptides [29]. Nineteen chemokine receptors have been identified so far, which include seven CXC receptors (CXCR1–7), ten CCR (CCR1–10), one CX3CR (CX3CR1), and one CR (XCR1) receptor [30]. Neutrophils are traditionally known to express only a very limited number of chemokine receptors and mainly express CXCR1 and CXCR2 in healthy individuals [31]. CXCR1 and CXCR2 are used by neutrophils to recognize N-terminal ELR (glutamic acid-leucine-arginine) motif-containing CXC chemokines. Human CXCR1 binds to CXCL8 (interleukin-8/IL-8) and CXCL6 (granulocyte chemotactic protein-2) [20, 29], as well as the ECM breakdown product N-acetyl PGP [32]. These three factors can also bind to CXCR2. However, CXCR2 is more promiscuous and binds different additional CXC chemokines, including CXCL1 (growth regulated oncogene-alpha/GRO-α), CXCL2 (GRO-β), CXCL3 (GRO-γ), CXCL5 (epithelial cell-derived neutrophil activating peptide-78/ ENA-78), and CXCL7 (neutrophil activating protein-2/GCP-2) [29]. Our study implies that these CXCR1 and CXCR2 ligands are not relevant in migration of neutrophils towards the FH in vitro. However, although neutrophils in healthy individuals mainly express CXCR1 and CXCR2 [31], it has been shown that infiltrated neutrophils from patients with chronic inflammatory lung diseases and rheumatoid arthritis express additional chemokine receptors on their surface, i.e., CCR1, CCR2, CCR3, CCR5, CXCR3, and CXCR4 [31]. Moreover, major trauma induces the release of several neutrophil subsets into the peripheral circulation, including young banded neutrophils and hyper-segmented neutrophils, which exhibit different properties and receptor expressions compared to mature neutrophils from healthy individuals [33]. Future studies may focus on the role of these neutrophil subsets in fracture healing and determine whether neutrophils within the FH express other chemokine receptors compared to neutrophils isolated from peripheral blood of healthy donors.
Another chemotactic factor for neutrophils is leukotriene B4 (LTB4), which is recognized by a high-affinity receptor (BLT1) and a low-affinity receptor (BLT2) [34]. Animal studies have shown that LTB4 mediates neutrophil influx after experimental spinal cord injury [35]. It is tempting to speculate that LTB4 also mediates neutrophil influx into other types of sterile tissue injury, such as bone injury. An additional chemoattractant for neutrophils is platelet-activating factor (PAF), which is a phospholipid that is bound by the PAF receptor (PAFR) [36]. Little is known about the role of PAF in tissue injury although animal studies did show that inactivation of PAF by PAF acetylhydrolase significantly decreased neutrophil influx in a rabbit model of myocardial ischemia/reperfusion injury [37]. Future studies should investigate to which extent the abovementioned factors are also relevant in chemotaxis of neutrophils towards the FH.
In summary, our study shows that chemotaxis of neutrophils towards the FH can be studied in vitro with Ibidi™ Chemotaxis3D μ-Slides. We found that serum from the human FH significantly induces chemotaxis, which was not affected by blocking CXCR1 and CXCR2. In contrast, CHIPS and CHIPSΔ1F, which blocks C5aR, induced a significant decrease in chemotaxis of neutrophils towards the FH. These findings may aid the development of therapies that prevent impairment of fracture healing after major trauma.
References
Bastian, Okan, Janesh Pillay, Jacqueline Alblas, Luke Leenen, Leo Koenderman, and Taco Blokhuis. 2011. Systemic inflammation and fracture healing. Journal of Leukocyte Biology 89: 669–673.
Giannoudis, Peter V., Thomas A. Einhorn, and David Marsh. 2007. Fracture healing: the diamond concept. Injury 38 (Suppl 4): S3–S6.
Mizuno, K., K. Mineo, T. Tachibana, M. Sumi, T. Matsubara, and K. Hirohata. 1990. The osteogenetic potential of fracture haematoma. Subperiosteal and intramuscular transplantation of the haematoma. Journal of Bone and Joint Surgery-British 72 (JBJS (Br)): 822.
Street, J, D Winter, J H Wang, A Wakai, A McGuinness, and H P Redmond. 2000. Is human fracture hematoma inherently angiogenic? Clin Orthop.Relat Res. Department of Academic Surgery, Cork University Hospital, Ireland: 224–237.
Grundnes, O, and O Reikeras. 1993. The importance of the hematoma for fracture healing in rats. Acta Orthop.Scand 64. Department of Orthopedics, University Hospital, Tromso, Norway: 340–342.
Park, Sang-Hyun, Mauricio Silva, Won-Jong Bahk, Harry McKellop, and Jay R. Lieberman. 2002. Effect of repeated irrigation and debridement on fracture healing in an animal model. Journal of Orthopaedic Research : Official Publication of the Orthopaedic Research Society 20: 1197–1204. https://doi.org/10.1016/S0736-0266(02)00072-4.
Bastian, Okan W., Leo Koenderman, Jacqueline Alblas, Luke P.H. Leenen, and Taco J. Blokhuis. 2016. Neutrophils contribute to fracture healing by synthesizing fibronectin + extracellular matrix rapidly after injury. Clinical Immunology 164: 78–84. https://doi.org/10.1016/j.clim.2016.02.001.
Grundnes, O, and O Reikeraas 2000. Effects of macrophage activation on bone healing. J Orthop.Sci 5. Departments of Orthopedics, Institute of Clinical Medicine, University of Tromso and National Hospital, University of Oslo, Norway: 243–247.
Reikerås, Olav, Hamid Shegarfi, Jacob E. Wang, and Stein E. Utvåg. 2005. Lipopolysaccharide impairs fracture healing: an experimental study in rats. Acta Orthopaedica 76: 749–753. https://doi.org/10.1080/17453670510045327.
Recknagel, Stefan, Ronny Bindl, Julian Kurz, Tim Wehner, Christian Ehrnthaller, Markus Werner Knöferl, Florian Gebhard, Markus Huber-Lang, Lutz Claes, and Anita Ignatius. 2011. Experimental blunt chest trauma impairs fracture healing in rats. Journal of Orthopaedic Research : Official Publication of the Orthopaedic Research Society 29: 734–739.
Bastian, Okan, Anne Kuijer, Leo Koenderman, Rebecca K. Stellato, Wouter W. van Solinge, Luke P.H. Leenen, and Taco J. Blokhuis. 2016. Impaired bone healing in multitrauma patients is associated with altered leukocyte kinetics after major trauma. Journal of Inflammation Research 9: 69. https://doi.org/10.2147/JIR.S101064.
Karladani, A H, H Granhed, J Kärrholm, J Styf, and J Karrholm. 2001. The influence of fracture etiology and type on fracture healing: a review of 104 consecutive tibial shaft fractures. Arch Orthop.Trauma Surg 121. Department of Orthopedics Surgery, Sahlgrenska University Hospital, Goteborg University, Sweden. abbas.karladani@orthop.gu.se: 325–328.
Kanakaris, N.K., and Peter V. Giannoudis. 2007. The health economics of the treatment of long-bone non-unions. Injury 38 (Elsevier): S77–S84.
Recknagel, Stefan, Ronny Bindl, Christoph Brochhausen, Melanie Göckelmann, Tim Wehner, Philipp Schoengraf, Markus Huber-Lang, Lutz Claes, and Anita Ignatius. 2013. Systemic inflammation induced by a thoracic trauma alters the cellular composition of the early fracture callus. The Journal of Trauma and Acute Care Surgery 74: 531–537. https://doi.org/10.1097/TA.0b013e318278956d.
Recknagel, Stefan, Ronny Bindl, Julian Kurz, Tim Wehner, Philipp Schoengraf, Christian Ehrnthaller, Qu Hongchang, et al. 2012. C5aR-antagonist significantly reduces the deleterious effect of a blunt chest trauma on fracture healing. Journal of orthopaedic research : official publication of the Orthopaedic Research Society 30: 581–586. https://doi.org/10.1002/jor.21561.
Grogaard, B, B Gerdin, and O Reikeras. 1990. The polymorphonuclear leukocyte: has it a role in fracture healing? Arch Orthop.Trauma Surg 109. Department of Surgery, Ulleval University Hospital, Oslo, Norway: 268–271.
Chung, Rosa, Johanna C. Cool, Michaela a Scherer, Bruce K. Foster, and Cory J. Xian. 2006. Roles of neutrophil-mediated inflammatory response in the bony repair of injured growth plate cartilage in young rats. Journal of Leukocyte Biology 80: 1272–1280. https://doi.org/10.1189/jlb.0606365.
Koenderman, Leo, Jan A.M. Van Der Linden, Henk Honing, and Laurien H. Ulfman. 2010. Integrins on neutrophils are dispensable for migration into three-dimensional fibrin gels. Thrombosis and Haemostasis 104: 599–608.
Pepperell, Emma E., and Suzanne M. Watt. 2013. A novel application for a 3-dimensional timelapse assay that distinguishes chemotactic from chemokinetic responses of hematopoietic CD133(+) stem/progenitor cells. Stem Cell Research 11: 707–720. https://doi.org/10.1016/j.scr.2013.04.006.
Wolf, M, M B Delgado, S A Jones, B Dewald, I Clark-Lewis, and M Baggiolini. 1998. Granulocyte chemotactic protein 2 acts via both IL-8 receptors, CXCR1 and CXCR2. European Journal of Immunology 28: 164–70. doi: https://doi.org/10.1002/(SICI)1521-4141(199801)28:01<164::AID-IMMU164>3.0.CO;2-S.
Ye, Richard D., François Boulay, Ji Ming Wang, Claes Dahlgren, Craig Gerard, Marc Parmentier, Charles N. Serhan, and Philip M. Murphy. 2009. International Union of Basic and Clinical Pharmacology. LXXIII. Nomenclature for the formyl peptide receptor (FPR) family. Pharmacological Reviews 61: 119–161. https://doi.org/10.1124/pr.109.001578.
de Haas, Carla J C, Karin Ellen Veldkamp, Andreas Peschel, Floor Weerkamp, Willem J B Van Wamel, Erik C J M Heezius, Miriam J J G Poppelier, Kok P M Van Kessel, and Jos A G van Strijp. 2004. Chemotaxis inhibitory protein of Staphylococcus aureus, a bacterial antiinflammatory agent. The Journal of experimental medicine 199. The Rockefeller University Press: 687–95. https://doi.org/10.1084/jem.20031636.
Postma, Bent, Miriam J. Poppelier, Joost C. van Galen, Eric R. Prossnitz, Jos A.G. van Strijp, Carla J.C. de Haas, and Kok P.M. van Kessel. 2004. Chemotaxis inhibitory protein of Staphylococcus aureus binds specifically to the C5a and formylated peptide receptor. Journal of immunology (Baltimore, Md. : 1950) 172: 6994–7001.
Haas, Pieter-Jan, Carla J.C. de Haas, Wendy Kleibeuker, Miriam J.J.G. Poppelier, Kok P.M. van Kessel, John A.W. Kruijtzer, Rob M.J. Liskamp, and Jos A.G. van Strijp. 2004. N-Terminal residues of the chemotaxis inhibitory protein of Staphylococcus aureus are essential for blocking formylated peptide receptor but not C5a receptor. The Journal of Immunology 173.
Claes, Lutz, Stefan Recknagel, and Anita Ignatius. 2012. Fracture healing under healthy and inflammatory conditions. Nature reviews. Rheumatology 8: 133–143. https://doi.org/10.1038/nrrheum.2012.1.
Flierl, Michael A., Mario Perl, Daniel Rittirsch, Christoph Bartl, Heike Schreiber, Vera Fleig, Gerald Schlaf, et al. 2007. The role of C5A in the innate immune response after experimental blunt chest trauma. Shock PAP: 25–31. https://doi.org/10.1097/shk.0b013e3180556a0b.
Zhang, Q, M Raoof, Y Chen, Y Sumi, T Sursal, W Junger, K Brohi, K Itagaki, and C J Hauser. 2010. Circulating mitochondrial DAMPs cause inflammatory responses to injury. Nature 464. Department of Surgery, Division of Trauma, Beth Israel Deaconess Medical Center and Harvard Medical School, Boston, Massachusetts 02215, USA: 104–107.
Thammavongsa, Vilasack, Hwan Keun Kim, Dominique Missiakas, and Olaf Schneewind. 2015. Staphylococcal manipulation of host immune responses. Nature Reviews Microbiology 13: 529–543. https://doi.org/10.1038/nrmicro3521.
Stillie, RoseMarie, Shukkur Muhammed Farooq, John R. Gordon, and Andrew W. Stadnyk. 2009. The functional significance behind expressing two IL-8 receptor types on PMN. Journal of Leukocyte Biology 86: 529–543. https://doi.org/10.1189/jlb.0208125.
Rossi, D., and A. Zlotnik. 2000. The biology of chemokines and their receptors. Annual Review of Immunology 18: 217–242. https://doi.org/10.1146/annurev.immunol.18.1.217.
Hartl, D., S. Krauss-Etschmann, B. Koller, P.L. Hordijk, T.W. Kuijpers, F. Hoffmann, A. Hector, et al. 2008. Infiltrated neutrophils acquire novel chemokine receptor expression and chemokine responsiveness in chronic inflammatory lung diseases. The Journal of Immunology 181. American Association of Immunologists: 8053–8067. https://doi.org/10.4049/jimmunol.181.11.8053.
Weathington, Nathaniel M., Anneke H. van Houwelingen, Brett D. Noerager, Patricia L. Jackson, Aletta D. Kraneveld, F. Shawn Galin, Gert Folkerts, Frans P. Nijkamp, and J. Edwin Blalock. 2006. A novel peptide CXCR ligand derived from extracellular matrix degradation during airway inflammation. Nature Medicine 12: 317–323. https://doi.org/10.1038/nm1361.
Pillay, J., F. Hietbrink, L. Koenderman, and L.P.H. Leenen. 2007. The systemic inflammatory response induced by trauma is reflected by multiple phenotypes of blood neutrophils. Injury 38: 1365–1372. https://doi.org/10.1016/j.injury.2007.09.016.
Yokomizo, T. 2014. Two distinct leukotriene B4 receptors, BLT1 and BLT2. Journal of Biochemistry 157: 65–71. https://doi.org/10.1093/jb/mvu078.
Saiwai, Hirokazu, Yasuyuki Ohkawa, Hisakata Yamada, Hiromi Kumamaru, Akihito Harada, Hideyuki Okano, Takehiko Yokomizo, Yukihide Iwamoto, and Seiji Okada. 2010. The LTB4-BLT1 axis mediates neutrophil infiltration and secondary injury in experimental spinal cord injury. The American Journal of Pathology 176: 2352–2366. https://doi.org/10.2353/ajpath.2010.090839.
Wardlaw, A.J., R. Moqbel, O. Cromwell, and A.B. Kay. 1986. Platelet-activating factor. A potent chemotactic and chemokinetic factor for human eosinophils. The Journal of Clinical Investigation 78: 1701–1706. https://doi.org/10.1172/JCI112765.
Morgan, E.N., E.M. Boyle, W. Yun, J.C. Kovacich, T.G. Canty, E. Chi, T.H. Pohlman, and E.D. Verrier. 1999. Platelet-activating factor acetylhydrolase prevents myocardial ischemia-reperfusion injury. Circulation 100: II365–II368.
Acknowledgements
The authors would like to thank Jan van der Linden for his help with analyzing chemotaxis of neutrophils towards the fracture hematoma. Moreover, we would like to thank Kok van Kessel for providing CHIPS and CHIPSΔ1F.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors would kindly like to acknowledge the financial support provided by the Osteosynthesis and Trauma Care Foundation (grant number 2010-TBLL, http://www.otcfoundation.org/about-otc/) and the Alexandre Suerman MD/PhD grant provided by the University Medical Center of Utrecht (http://www.umcutrecht.nl/). The study sponsors were not involved in the study design, collection, analysis, interpretation of data, writing of the manuscript or the decision to submit the manuscript for publication.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Bastian, O.W., Mrozek, M.H., Raaben, M. et al. Serum from the Human Fracture Hematoma Contains a Potent Inducer of Neutrophil Chemotaxis. Inflammation 41, 1084–1092 (2018). https://doi.org/10.1007/s10753-018-0760-4
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10753-018-0760-4