Abstract
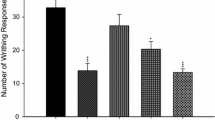
The possible protective and curative effects of paeonol on carrageenan-induced acute hind paw edema in rats and dextran sulfate sodium (DSS)-induced colitis in mice have been evaluated. After oral administration, paeonol (20 and 40 mg/kg) reduced the edema increase in paw volumes and also the development of DSS-induced murine colitis. Furthermore, anti-inflammatory and anti-oxidant activities of paeonol (1) together with its 10 metabolites (M2~M11) were investigated by using in vitro anti-inflammatory and anti-oxidant assays. M3 and M11 exhibited significant 2,2-diphenyl-1-picrylhydrazyl (DPPH) radical scavenging activities (with EC50 values of 93.44 and 23.24 μM, respectively). All the metabolites except M8 showed hydroxyl radical scavenging activities, and M3 and M11 were the most potent agents (with EC50 values of 336.02 and 124.05 μM, respectively). Inhibitory effects of paeonol, M2~M11 on the overproduction of nitric oxide (NO), and the release of TNF-α were also tested. M3 and M11 potently inhibited lipopolysaccharide (LPS)-induced overproduction of NO in macrophage RAW 264.7. Western blot results demonstrated that paeonol, M3, and M11 downregulated the high expression of inducible nitric oxide synthase (iNOS) and COX-2 proteins, and the effects of M3 and M11 were more potent when compared with paeonol. These findings indicated that paeonol may play anti-inflammatory and anti-oxidant roles by changing to its active metabolites after absorption. In addition, further investigations on the mechanism showed that paeonol, M3, and M11 blocked the phosphorylation of MAPK/ERK 1/2 and p38, whereas they showed no effect on the phosphorylation of JNK. The above results suggested that pre-treatment with paeonol might be an effective therapeutic intervention against inflammatory diseases including colitis.










Similar content being viewed by others
References
Chou, T.C. 2003. Anti-inflammatory and analgesic effects of paeonol in carrageenan-evoked thermal hyperalgesia. British Journal of Pharmacology 139: 1146–1152.
Hong, M.H., J.H. Kim, S.H. Na, H. Bae, Y.C. Shin, S.H. Kim, and S.G. Ko. 2010. Inhibitory effects of Paeonia suffruticosa on allergic reactions by inhibiting the NF-kappaB/I kappaB-alpha signaling pathway and phosphorylation of ERK in an animal model and human mast cells. Bioscience Biotechnology and Biochemistry 74: 1152–1156.
Zhang, L.H., P.G. Xiao, and Y. Huang. 1996. Recent progresses in pharmacological and clinical studies of paeonol. Zhongguo Zhong Xi Yi Jie He Za Zhi 16: 187–190.
Lin, H.C., H.Y. Ding, F.N. Ko, C.M. Teng, and Y.C. Wu. 1999. Aggregation inhibitory activity of minor acetophenones from Paeonia species. Planta Medica 65: 595–599.
Hirai, A., T. Terano, T. Hamazaki, J. Sajiki, H. Saito, K. Tahara, Y. Tamura, and A. Kumagai. 1983. Studies on the mechanism of antiaggregatory effect of Moutan Cortex. Thrombosis Research 31: 29–40.
Ding, H.Y., T.H. Chou, R.J. Lin, L.P. Chan, G.H. Wang, and C.H. Liang. 2011. Antioxidant and antimelanogenic behaviors of Paeonia suffruticosa. Plant Foods for Human Nutrition 66: 275–284.
Bao, M.H., Y.W. Zhang, and H.H. Zhou. 2013. Paeonol suppresses oxidized low-density lipoprotein induced endothelial cell apoptosis via activation of LOX-1/p38MAPK/NF-κB pathway. Journal of Ethnopharmacology 146: 543–551.
Li, H., M. Dai, and W. Jia. 2009. Paeonol attenuates high-fat-diet-induced atherosclerosis in rabbits by anti-inflammatory activity. Planta Medica 75: 7–11.
Tsai, H.Y., H.Y. Lin, Y.C. Fong, J.B. Wu, Y.F. Chen, M. Tsuzuki, and C.H. Tang. 2008. Paeonol inhibits RANKL-induced osteoclastogenesis by inhibiting ERK, p38 and NF-κB pathway. European Journal of Pharmacology 588: 124–133.
Xie, Y., H. Zhou, Y.F. Wong, H.X. Xu, Z.H. Jiang, and L. Liu. 2008. Study on the pharmacokinetics and metabolism of paeonol in rats treated with pure paeonol and an herbal preparation containing paeonol by using HPLC–DAD-MS method. Journal of Pharmaceutical and Biomedical Analysis 46: 748–756.
Ding, L., Z. Liu, F. Zhao, G. Bai, L. Chen, X. Yao, and F. Qiu. 2012. Isolation and identification of the metabolites of paeonol in human urine. Xenobiotica 42: 1206–1212.
Niu, X., Q. Mu, W. Li, H. Yao, H. Li, and H. Huang. 2014. Esculentic acid, a novel and selective COX-2 inhibitor with anti-inflammatory effect in vivo and in vitro. European Journal of Pharmacology 740: 532–538.
Ohkawara, T., H. Takeda, K. Kato, K. Miyashita, M. Kato, T. Iwanaga, and M. Asaka. 2005. Polaprezinc (N-(3-aminopropionyl)-L-histidinato zinc) ameliorates dextran sulfate sodium-induced colitis in mice, Scand. Journal of Gastroenterology 40: 1321–1327.
Denizot, F., and R. Lang. 1986. Rapid colorimetric assay for cell growth and survival. Modifications to the tetrazolium dye procedure giving improved sensitivity and reliability. Journal of Immunological Methods 89: 271–277.
Zhao, F., L. Wang, and K. Liu. 2009. In vitro anti-inflammatory effects of arctigenin, a lignan from Arctium lappa L., through inhibiton on iNOS pathway. Journal of Ethnopharmacology 122: 457–462.
Zhao, F., H. Xu, E.Q. He, Y.T. Jiang, and K. Liu. 2008. Inhibitory effects of sesquiterpenes from Saussurea lappa on the overproduction of nitric oxide and TNF-α release in LPS-activated macrophages. Journal of Asian Natural Products Research 10: 1045–1053.
Zhao, F., L. Chen, C. Bi, M. Zhang, W. Jiao, and X. Yao. 2013. In vitro anti-inflammatory effect of picrasmalignan A by the inhibition of iNOS and COX-2 expression in LPS-activated macrophage RAW 264.7 cells. Molecular Medicine Reports 8: 1575–1579.
Chan, E.D., and D.W. Riches. 1998. Potential role of the JNK/SAPK signal transduction pathway in the induction of iNOS by TNF-α. Biochemical and Biophysical Research Communications 253: 790–796.
Kim, S.H., J. Kim, and R.P. Sharma. 2004. Inhibition of p38 and ERK MAP kinases blocks endotoxin-induced nitric oxide production and differentially modulates cytokine expression. Pharmacological Research 49: 433–439.
Kaminska, B. 2005. MAPK signalling pathways as molecular targets for anti-inflammatory therapy—from molecular mechanisms to therapeutic benefits. Biochimica et Biophysica Acta 1754: 253–262.
Nakamura, M., and S.H. Ferreira. 1987. A peripheral sympathetic component in inflammatory hyperalgesia. European Journal of Pharmacology 135: 145–153.
Ferreira, S.H., B.B. Lorenzetti, and S. Poole. 1993. Bradykinin initiates cytokine mediated inflammatory hyperalgesia. British Journal of Pharmacology 110: 1227–1231.
Jung, H.W., C.H. Yoon, K.M. Park, H.S. Han, and Y.K. Park. 2009. Hexane fraction of zingiberis rhizoma crudus extract inhibits the production of nitric oxide and proinflammatory cytokines in LPS-stimulated BV2 microglial cells via the NF-kappaB pathway. Food and Chemical Toxicology 47: 1190–1197.
Kim, K.N., Y.J. Ko, M.C. Kang, H.M. Yang, S.W. Roh, T. Oda, Y.J. Jeon, W.K. Jung, S.J. Heo, W.J. Yoon, and D. Kim. 2013. Anti-inflammatory effects of trans-1, 3-diphenyl-2, 3-epoxypropane-1-one mediated by suppression of inflammatory mediators in LPS-stimulated RAW 264.7 macrophages. Food and Chemical Toxicology 53: 371–375.
Yoon, W.J., S.J. Heo, S.C. Han, H.J. Lee, G.J. Kang, H.K. Kang, J.W. Hyun, Y.S. Koh, and E.S. Yoo. 2012. Anti-inflammatory effect of sargachromanol G isolated from Sargassum siliquastrum in RAW 264.7 cells. Archives of Pharmacal Research 35: 1421–1430.
Acknowledgments
This work was supported by the State Key Laboratory of Mycology, Institute of Microbiology, Chinese Academy of Sciences, and the Taishan Scholar Project to Fenghua Fu, and the Scientific and Technological Project of Yantai City, as well as by the Undergraduate Scientific and Technological Innovation Project of Yantai University.
Conflict of interest
The authors report no conflicts of interest in this work.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jin, X., Wang, J., Xia, ZM. et al. Anti-inflammatory and Anti-oxidative Activities of Paeonol and Its Metabolites Through Blocking MAPK/ERK/p38 Signaling Pathway. Inflammation 39, 434–446 (2016). https://doi.org/10.1007/s10753-015-0265-3
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10753-015-0265-3