Abstract
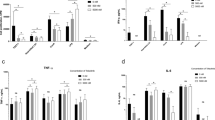
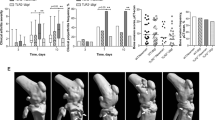
Chloramphenicol is mostly used against coagulase-negative Staphylococcus aureus, and its protective role against coagulase-positive S. aureus is not well studied. In our study, arthritis was induced in mice by S. aureus (Apollo Gleneagles 33 (AG-33) or American Type Culture Collection 25923 (ATCC-25923)) infection. Chloramphenicol was administered after 2 h of infection. Mice were killed at 1, 3, 5 days post-infection. Mice inoculated with pathogenic Staphylococci (AG-33) expressing coagulase and Toxic shock syndrome toxin-1 (TSST-1), displayed severe arthritis with enhanced bacterial burden in the spleen, cytokine production in serum and synovial tissue, neutrophil recruitment, and cyclooxegenase-2 expression in synovial tissue compared with ATCC-25923-infected groups. Severity of arthritis was regulated by chloramphenicol treatment. Our study suggests that alteration in the inflammatory cytokine levels and pronounced production of cyclooxygenase-2 play important roles in progression of arthritis which is regulated by application of chloramphenicol.







Similar content being viewed by others
References
Mark, E., J. Shirtliff, and T. Mader. 2002. Acute septic arthritis. Clinical Microbiology Reviews 15: 527–544.
Wysenbeek, A.J., J. Volchek, M. Amit, D. Robinson, I. Boldur, and Z. Nevo. 1998. Treatment of Staphylococcal septic arthritis in rabbits by systemic antibiotics and intra-articular corticosteroids. Annals of the Rheumatic Diseases 57: 687–690.
Das, D., and B. Bishayi. 2009. Staphylococcal catalase protects intracellularly survived bacteria by destroying H2O2 produced by the murine peritoneal macrophages. Microbiolial Pathogen 47: 57–67.
Christian, G., and L.K. William. 2009. Staphylococcus aureus: new evidence for intracellular persistence. Trends in Microbiology 17: 59–65.
Cunningham, R., A. Cockayne, and H. Humphrey. 1996. Clinical and molecular aspects of the pathogenesis of Staphylococcus aureus bone and joint infections. Journal of Medical Microbiology 44: 157–164.
Goldenberg, D.I., and J.I. Reed. 1985. Bacterial arthritis. The New England Journal of Medicine 312: 764–771.
Bremell, T., and A. Tarkowski. 1995. Preferential induction of septic arthritis and mortality by superantigen-producing staphylococci. Infection and Immunity 63: 4185–4187.
Tarkowski, A., L.V. Collins, I. Gjertsson, O.H. Hultgren, I.-M. Jonsson, E. Sakiniene, and M. Verdrengh. 2001. Model systems: Modeling human staphylococcal arthritis and sepsis in the mouse. Trends in Microbiology 9: 321–326.
Luciana, T., M. Pierfrancesco, M. Paolo, M. Lucio, C. Paola, and R. Emanuela. 1990. Experimental model of type IV Streptococcus agalactiae (Group B Streptococcus) infection in mice with early development of septic arthritis. Infection and Immunity 58: 3093–3100.
Goldenberg, D.L. 1998. A recent review of nongonococcal and gonococcal arthritis. Lancet 351: 197–202.
Bremell, T., S. Lange, L. Svensson, E. Jennische, K. Grondahl, and A. Tarkowski. 1990. Outbreak of spontaneous staphylococcal arthritis and osteitis in mice. Arthritis and Rheumatism 33: 1739–1744.
Bremell, T., A. Abdelnour, and A. Tarkowski. 1992. Histopathological and serological progression of experimental Staphylococcus aureus arthritis. Infection and Immunity 60: 2976–2985.
Victor, J., D. Torres, L. Stauff, P. Gleb, S.B. Jelena, and E. Laura. 2007. A Staphylococcus aureus regulatory system that responds to host heme and modulates virulence. Cell Host Microb 19: 109–119.
Cohen, M.L. 2000. Changing patterns of infectious disease. Nature 406: 762–767.
Goldenberg, D.L., and J.J. Reed. 1989. Bacterial arthritis. In Text book of Rheumatology, 3rd ed, ed. W.O. Kelly, E.D. Harris, S. Ruddy, and C.B. Sledge, 1567–1585. Philadelphia: The W B Saunders Co.
Brennan, F.M. 1994. Role of cytokines in experimental arthritis. Clinical and Experimental Immunology 97: 1–3.
Nakane, A., M. Okamoto, M. Asano, M. Kohanawa, and T. Minagawa. 1995. Endogenous gamma interferon, tumor necrosis factor, and interleukin-6 in Staphylococcus aureus infection in mice. Infection and Immunity 63: 1165–1172.
Strindhall, J., P. Lindgren, S. Löfgren, and E. Kihlström. 2005. Clinical isolates of Staphylococcus aureus vary in ability to stimulate cytokine expression in human endothelial cells. Scandinavian Journal of Immunology 61: 57–60.
Abdelnour, A., S. Arvidson, T. Bremell, C. Ryden, and A. Tarkowski. 1993. The accessory gene regulator (agr) controls Staphylococcus aureus virulence in a murine arthritis model. Infection and Immunity 61: 3879–3885.
Raheem, B., and K.S. Kherani. 2007. Septic arthritis in patients with pre-existing inflammatory arthritis. Canadian Medical Association Journal 176: 1605–1608.
Nelson, J.D. 1972. The bacterial etiology and antibiotic management of septic arthritis in infants and children. Pediatrics 50: 437–440.
Benedicte, F., and J.P. Dana. 2005. Recognition of Staphylococcus aureus by the innate immune system. Clinical Microbiology Reviews 18: 521–540.
Abraham, E. 2002. Anti cytokine therapy. In The sepsis text, ed. J.L. Vincent, J. Carlet, and S.M. Opal, 719–728. Norwell: Kluwer Academic Publisher.
Tarkowski, A. 2006. Infectious arthritis. Best Practice & Research. Clinical Rheumatology 20: 1029–1044.
Gemmell, J. 1995. Antibiotics and the expression of staphylococcal virulence. Antimicrob Chemothr 36: 283–291.
Van den Broek, P.J. 1989. Antimicrobial drugs, microorganisms, and phagocytes. Reviews of Infectious Diseases 11: 213–245.
Tarkowski, A., M. Bokarewa, L.V. Collins, I. Gjertsson, O.H. Hultgren, T. Jin, I.M. Jonsson, E. Josefsson, E. Sakiniene, and M. Verdrengh. 2002. Current status of pathogenetic mechanisms in staphylococcal arthritis. FEMS Microbiology Letters 217: 125–132.
Sen, R., D. Das, and B. Bishayi. 2009. Staphylococcal catalase regulates its virulence and induces arthritis in catalase deficient mice. Indian Journal of Physiology and Pharmacology 53: 307–317.
Yao, L., J.W. Berman, M.F. Stephen, and D.L. Franklin. 1997. Correlation of histopathologic and bacteriologic changes with cytokine expression in an experimental murine model of bacteremic Staphylococcus aureus infection. Infection and Immunity 65: 3889–3895.
Bremell, T., S. Lange, A. Yacoub, C. Ryden, and A. Tarkowski. 1991. Experimental Staphylococcus aureus arthritis in mice. Infection and Immunity 59: 2615–2623.
Hultgren, O., M. Stensson, and A. Tarkowski. 2001. Role of IL-12 in Staphylococcus aureus-triggered arthritis and sepsis. Arthritis Research 3: 41–47.
Mcclain, J.B., B. Joshi, and R. Rice. 1988. Chloramphenicol, Gentamicin, and Ciprofloxacin against Murine Scrub Typhus. Antimicrobial Agents and Chemotherapy 32: 285–286.
Hultgren, O., H.P. Eugster, J.D. Sedgwick, H. Korner, and A. Tarkowski. 1998. TNF/lymphotoxin-alpha double-mutant mice resist septic arthritis but display increased mortality in response to Staphylococcus aureus. Journal of Immunology 161: 5937–5942.
Nilsson, I.-M., O. Hartford, T. Foster, and A. Tarkowski. 1999. Alpha-toxin and gamma-toxin jointly promote Staphylococcus aureus virulence in murine septic arthritis. Infection and Immunity 67: 1045–1049.
Lefkowitz, D.L., M.P. Gelderman, S.R. Fuhrmann, J.D. Grahams Starnes, S.S. Lefkowitz, A. Bollen, and N. Moguilevsky. 1999. Neutrophil myeloperoxidase–macrophage interactions perpetuate chronic inflammation associated with experimental arthritis. Clinical Immunology 91: 145–155.
Ghosal, A., S. Das, S. Ghosh, M.K. Mishra, V. Sharma, P. Koli, E. Sen, and A. Basu. 2007. Proinflammatory mediators released by activated microglia induces neuronal death in Japanese Encephalitis. Glia 55: 483–496.
Oser, B.L. 1976. In Hawk’s Physiological Chemistry, ed. Oser B.L., 1044–1048. McGraw-Hill, New York.
Mishra, M.K., K. Dutta, S.K. Saheb, and A. Basu. 2009. Understanding the molecular mechanism of blood–brain barrier damage in an experimental model of Japanese encephalitis: Correlation with minocycline administration as a therapeutic agent. Neurochemistry International 55: 717–723.
Johnson, W.M., S.D. Tyler, E.P. Ewan, F.E. Ashton, D.R. Pollard, and K.R. Rozee. 1991. Detection of genes for enterotoxins, exfoliative toxins, and toxic shock syndrome toxin 1 in Staphylococcus aureus by the polymerase chain reaction. Journal of Clinical Microbiology 29: 426–430.
Hookey, J.V., J.F. Richardson, and B.D. Cookson. 1998. Molecular typing of S. aureus based on PCR RFLP and DNA sequence analysis of coa gene. Journal of Clinical Microbiology 36: 1083–1089.
Becker, K., R. Rotn, and G. Peters. 1998. Rapid and specific detection of toxicogenic S. aureus. Use of two multiplex PCR enz. Immunoassay for amplification and hybridization of staphylococcal enterotoxin gene, exfoliative toxin gene, and toxic shock syndrome toxin 1 gene. Journal of Clinical Microbiology 36: 2548–2553.
Das, D., and A. Das. 2005. Analysis of variance. In Statistics in Biology and Psychology, ed. D. Das, 280–293. Calcutta: Academic Publisher.
Das, D., S.S. Saha, and B. Bishayi. 2008. Intracellular survival of Staphylococcus aureus: Correlating production of catalase and superoxide dismutase with levels of inflammatory cytokines. Inflammation Research 57: 340–349.
Zhao, Y.-X., I.-M. Nilsson, and A. Tarkowski. 1998. The dual role of interferon-γ in experimental Staphylococcus aureus septicaemia versus arthritis. Immunology 93: 80–85.
Zhao, Y.-X., A. Abdelnour, R. Holmdahl, and A. Tarkowski. 1995. Mice with the xid B cell defect are less susceptible to developing Staphylococcus aureus-induced arthritis. Journal of Immunology 155: 2067–2076.
Gjertsson, I., O.H. Hultgren, and A. Tarkowski. 2002. Interleukin-10 ameliorates the outcome of Staphylococcus aureus arthritis by promoting bacterial clearance. Clinical and Experimental Immunology 130: 409–414.
Hyon, K.C., D.B. Mount, and A.M. Reginato. 2005. Pathogenesis of gout. Annals of Internal Medicine 143: 499–516.
Acknowledgements
The author (Biswadev Bishayi) thanks the University of Calcutta for providing the fellowship to Ms. Sayantani Majumdar (Sanction No UGC/1000/Fellow (Univ) date 13 Oct 2009) under the scheme of University Research Fellowship (URF), University of Calcutta from University Grants Commission, New Delhi, India. The corresponding author also thanks Professor Dhrubojyoti Chattopadhyay, Department of Biochemistry, University of Calcutta for providing facilities for PCR.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Majumdar, S., Dutta, K., Manna, S.K. et al. Possible Protective Role of Chloramphenicol in TSST-1 and Coagulase-Positive Staphylococcus aureus-Induced Septic Arthritis with Altered Levels of Inflammatory Mediators. Inflammation 34, 269–282 (2011). https://doi.org/10.1007/s10753-010-9233-0
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10753-010-9233-0