Abstract
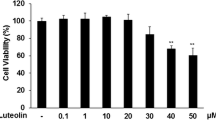
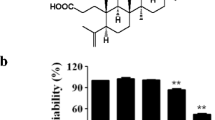
The effect of CO on the expression of iNOS and COX-2 was investigated by using a CO-releasing molecule (CORM)-2 in LPS-activated RAW 264.7 cells in vitro. Interestingly, CORM-2 significantly inhibited iNOS (NO) but not COX-2 (PGE2) expression. PPAR-γ activators such as troglitazone, GW1929, and 15-deoxy-Δ12, 14- prostaglandin J2 showed preferential inhibitory effect on iNOS over COX-2 expression in LPS-activated macrophages. The same effect was shown in lung tissues (iNOS, COX-2) and serum (NO, PGE2) when administered of CORM-2 in LPS-induced septic mice, indicating that CO derived from CORM-2 differentially regulates iNOS and COX-2 through PPAR-γ activation under inflammation state.




Similar content being viewed by others
References
Xie, Q. W., Y. Kashiwabara, and C. Nathan. 1994. Role of transcription factor NF-κB/Rel in induction of nitric oxide synthase. J. Biol. Chem. 269:4705–4708.
Nakao, S., Y. Ogata, E. Shimizu-Sasaki, M. Yamazaki, S. Furuyama, and H. Sugiya. 2000. Activation of NF-kappaB is necessary for IL-1beta-induced cyclooxygenase-2 (COX-2) expression in human gingival fibroblasts. Mol. Cell Biochem. 209:113–118.
Wang, X. M., H. P. Kim, K. Nakahira, S. W. Ryter, and A. M. Choi. 2009. The heme oxygenase-1/carbon monoxide pathway suppresses TLR4 signaling by regulating the interaction of TLR4 with caveolin-1. J. Immunol. 182:3809–3818.
Alcaraz, M. J., M. I. Guillen, M. L. Ferrandiz, J. Megias, and R. Motterlini. 2008. Carbon monoxide-releasing molecules: a pharmacological expedient to counteract inflammation. Curr. Pharm. Des. 14:465–472.
Sun, J. J., H. J. Kim, H. G. Seo, J. H. Lee, H. S. Yun-Choi, and K. C. Chang. 2008. YS 49, 1-(alpha-naphtylmethyl)-6, 7-dihydroxy-1, 2, 3, 4-tetrahydroisoquinoline, regulates angiotensin II-stimulated ROS production, JNK phosphorylation and vascular smooth muscle cell proliferation via the induction of heme oxygenase-1. Life Sci. 82:600–607.
Chung, S. W., X. Liu, A. A. Macias, R. M. Baron, and M. A. Perrella. 2008. Heme oxygenase-1-derived carbon monoxide enhances the host defense response to microbial sepsis in mice. J. Clin. Invest. 118:239–247.
Ryter, S. W., J. Alam, and A. M. Choi. 2006. Heme oxygenase/carbon monoxide: from basic science to therapeutic application. Physiol. Rev. 86:583–650.
Vane, J. R., J. A. Mitchell, I. Appelton, A. Tomlinson, D. Bishop-Bailey, J. Croxtall, and D. A. Willoughby. 1994. Inducible isoforms of cyclooxygenase and nitric-oxide synthase in inflammation. Proc. Natl. Acad. Sci. U. S. A. 91:2046–2050.
Welch, J. S., M. Ricote, T. E. Akiyama, F. J. Gonzales, and C. K. Glass. 2003. PPARgamma and PPARdelta negatively regulate specific subsets of lipoposaccharide and IFN-gamma target genes in macrophage. Proc. Natl. Acad. Sci. U. S. A. 100:6712–6717.
Kang, Y. J., E. B. Koo, Y. S. Lee, H. S. Yun-Choi, and K. C. Chang. 1999. Prevention of the expression of inducible nitric oxide synthase by novel positive inotropic agent, YS 49, in rat vascular smooth muscle and RAW 264.7 macrophages. Br. J. Pharmacol. 128:357–364.
Otterbein, L. E., F. H. Bach, J. Alam, M. Soares, H. Tao Lu, M. Wysk, R. J. Davis, R. A. Flavell, and A. M. Choi. 2000. Carbon monoxide has anti-inflammatory effects involving the mitogen-activated protein kinase pathway. Nat. Med. 6:422–428.
Motterlini, R., J. E. Clark, R. Foresti, P. Sarathchandra, B. E. Mann, and C. J. Green. 2002. Carbon monoxide-releasing molecules: characterization of biochemical and vascular activities. Circ. Res. 90:E17–E24.
Nobre, L. S., F. Al-Shahrouh, J. Dopazo, and L. M. Saraiva. 2009. Exploring the antimicrobial action of a carbon monoxide-releasing compound through whole-genome transcription profiling of Escherichia coli. Microbiology 155:813–824.
Davidge, K. S., G. Sanguinetti, C. H. Yee, A. G. Cox, C. W. McLeod, C. E. Monk, B. E. Mann, R. Motterlini, and R. K. Poole. 2009. Carbon monoxide-releasing antibacterial molecules target respiration and global transcriptional regulators. J. Biol. Chem. 284:4516–4524.
Guillen, M. I., J. Megias, V. Clerigues, F. Gomar, and M. J. Alcaraz. 2008. The CO-releasing molecule CORM-2 is a novel regulator of the inflammatory process in osteoarthritic chondrocytes. Rheumatology (Oxford) 47:1323–1328.
De Backer, O., E. Elinck, P. Sips, E. Buys, P. Brouckaert, and R. A. Lefebvre. 2008. Role of the soluble guanylyl cyclase alpha1/alpha2 subunits in the relaxant effect of CO and CORM-2 in murine gastric fundus. Naunyn Schmiedebergs. Arch. Pharmacol. 378:493–502.
Masini, E., A. Vannacci, P. Failli, R. Mastroianni, L. Giannini, M. C. Vinci, C. Uliva, R. Motterlini, and P. F. Mannaioni. 2008. A carbon monoxide-releasing molecule (CORM-3) abrogates polymorphonuclear granulocyte-induced activation of endothelial cells and mast cells. FASEB J. 22:3380–3388.
Davies, A. L., A. L. Kramer, and K. C. Hayes. 2008. Carbon monoxide-releasing molecule tricarbonyldichlororuthenium (II) dimmer induces concentration-dependent alterations in the electrophysiological properties of axons in mammalian spinal cord. Neuroscience 151:1104–1111.
Sawle, P., R. Foresti, B. E. Mann, T. R. Johnson, C. J. Green, and R. Motterlini. 2005. Carbon monoxide-releasing molecules (CO-RMs) attenuate the inflammatory response elicited by lipopolysaccharide in RAW2647 murine macrophages. Br. J. Pharmacol. 145:800–810.
Sarady, J. K., B. S. Zuckerbraun, M. Bilban, O. Wagner, A. Usheva, F. Liu, E. Ifedigbo, R. Zamora, A. M. Choi, and L. E. Otterbein. 2004. Carbon monoxide protection against endotoxic shock involves reciprocal effects on iNOS in the lung and liver. FASEB J. 18:854–856.
Bilban, M., F. H. Bach, S. L. Otterbein, E. Ifedigbo, J. C. d’Avila, H. Esterbauer, B. Y. Chin, A. Usheva, S. C. Robson, O. Wagner, and L. E. Otterbein. 2006. Carbon monoxide orchestrates a protective response through PPARγ. Immunity 24:601–610.
Hoetzel, A., T. Dolinay, S. Vallbracht, Y. Zhang, H. P. Kim, E. Ifedigbo, S. Alber, A. M. Kaynar, R. Schmidt, S. W. Ryter, and A. M. Choi. 2008. Carbon monoxide protects against ventilator-induced lung injury via PPAR-gamma and inhibition of Egr-1. Am. J. Respir. Crit. Care Med. 177:1223–1232.
Suh, G. Y., Y. Jin, A. K. Yi, X. M. Wang, and A. M. Choi. 2006. CCAAT/enhancer-binding protein mediates carbon monoxide-induced suppression of cyclooxygenase-2. Am. J. Respir. Mol. Cell. Biol. 35:220–226.
Cheng, C., Y. Qin, X. Shao, H. Wang, Y. Gao, M. Cheng, and A. Shen. 2007. Induction of TNF-alpha by LPS in Schwann cell is regulated by MAPK activation signals. Cell Mol. Neurobiol. 27:909–921.
Wang, H., C. Cheng, Y. Qin, S. Niu, S. Gao, X. Li, T. Tao, and A. Shen. 2009. Role of mitogen-activated protein kinase cascades in inducible nitric oxide synthase expression by lipopolysaccharide in a rat schwann cell line. Neurochem. Res. 34:430–437.
Nakao, A., D. J. Kaczorowski, B. S. Zuckerbraun, J. Lei, G. Faleo, K. Deguchi, K. R. McCurry, T. R. Billiar, and S. Kanno. 2008. Galantamine and carbon monoxide protect brain microvascular endothelial cells by heme oxygenase-1 induction. Biochem. Biophys. Res. Commun. 367:674–679.
Kim, K. M., H. O. Pae, M. Zhung, H. Y. Ha, Y. A. Ha, K. Y. Chai, Y. K. Cheong, J. M. Kim, and H. T. Chung. 2008. Involvement of anti-inflammatory heme oxygenase-1 in the inhibitory effect of curcumin on the expression of pro-inflammatory inducible nitric oxide synthase in RAW264.7 macrophages. Biomed. Pharmacother. 62:630–636.
Tsoyi, K., H. J. Kim, J. S. Shin, D. H. Kim, H. J. Cho, S. S. Lee, S. K. Ahn, H. S. Yun-Choi, J. H. Lee, H. G. Seo, and K. C. Chang. 2008. HO-1 and JAK-2/STAT-1 signals are involved in preferential inhibition of iNOS over COX-2 gene expression by newly synthesized tetrahydroisoquinoline alkaloid, CKD712, in cells activated with lipopolysaccharide. Cell. Signal. 20:1839–1847.
Acknowledgments
This study was supported by a grant of the Korea Healthcare technology R&D Project, Ministry of Health & Welfare, Republic of Korea. (A080506) and also supported by MRC program of Ministry of Science and Technology/Korea Science and Engineering Foundation (R13-2005-012-01003-0). Konstantin T and Y.M Ha were supported by BK21 program. We greatly thank Dr. Choi A. M. K. from Harvard Medical School for his commentaries.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tsoyi, K., Ha, Y.M., Kim, Y.M. et al. Activation of PPAR-γ by Carbon Monoxide from CORM-2 Leads to the Inhibition of iNOS but not COX-2 Expression in LPS-Stimulated Macrophages. Inflammation 32, 364–371 (2009). https://doi.org/10.1007/s10753-009-9144-0
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10753-009-9144-0