Abstract
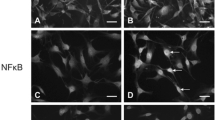
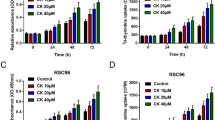
β4 Galactosylation of glycoproteins is one of the most important post-translational modifications. Recent studies have demonstrated that aberrant galactosylation associates with some inflammation diseases. β-1,4-galactosyltransferase-I (β-1,4-GalT-I), which transfers galactose to the terminal N-acetylglucosamine of N- and O-linked glycans in a β-1,4- linkage, considered to be the major galactosyltransferse among the seven members of the subfamily responsible for β4 galactosylation. In the present study, we investigated the expression of β-1,4-GalT-I in Schwann cells under Lipopolysaccharide (LPS) treatment. RT-PCR revealed that the β-1,4-GalT-I mRNA was significant increased as early as 2 h after LPS stimulation. Immunofluorescence showed that β-1,4-GalT-I was located in Golgi apparatus and membrane of Schwann cells. With the 1 μg/ml LPS treatment, expression levels of β-1,4-GalT-I was much higher compared with control group. In addition, lectin blot indicated that the β4 galactosylation of glycoproteins such as integrin α5 was enhanced, which may due to the induced β-1,4-GalT-I expression. These results suggested that β-1,4-GalT-I may play an important role in adhesion and migration of Schwann cells during inflammation.




Similar content being viewed by others
References
Powell, L. D., K. Paneerselvam, R. Vij, S. Diaz, A. Manzi, N. Buist, H. Freeze, and A. Varki. 1994. Carbohydrate-deficient glycoprotein syndrome: not an N-linked oligosaccharide processing defect, but an abnormality in lipid-linked oligosaccharide biosynthesis? J. Clin. Invest. 94:1901–1909. doi:10.1172/JCI117540.
Fukuda, M. N., K. A. Masri, A. Dell, L. Luzzatto, and K. W. Moremen. 1990. Incomplete synthesis of N-glycans in congenital dyserythropoietic anemia type ii caused by a defect in the gene encoding alpha-mannosidase ii. Proc. Natl. Acad. Sci. U. S. A. 87:7443–7447.
Watanabe, H., K. Nakashima, H. Saito, and M. Slaytor. 2002. New endo-beta-1,4-glucanases from the parabasalian symbionts, pseudotrichonympha grassii and holomastigotoides mirabile of coptotermes termites. Cell. Mol. Life Sci. 59:1983–1992.
Apweiler, R., H. Hermjakob, and N. Sharon. 1999. On the frequency of protein glycosylation, as deduced from analysis of the swiss-prot database. Biochim. Biophys. Acta. 1473:4–8. doi:S0304-4165(99)00165-8[pii].
Shi, X., S. Amindari, K. Paruchuru, D. Skalla, H. Burkin, B. D. Shur, and D. J. Miller. 2001. Cell surface beta-1,4-galactosyltransferase-I activates g protein-dependent exocytotic signaling. Development. 128:645–654.
Hathaway, H. J., and B. D. Shur. 1992. Cell surface beta 1,4-galactosyltransferase functions during neural crest cell migration and neurulation in vivo. J. Cell Biol. 117:369–382.
Huang, Q., B. D. Shur, and P. C. Begovac. 1995. Overexpressing cell surface beta 1.4-galactosyltransferase in PC12 cells increases neurite outgrowth on laminin. J. Cell Sci. 108(Pt 2):839–847.
Eckstein, D. J., and B. D. Shur. 1992. Cell surface beta-1,4-galactosyltransferase is associated with the detergent-insoluble cytoskeleton on migrating mesenchymal cells. Exp. Cell Res. 201:83–90. doi:0014-4827(92)90350-H[pii].
Evans, S. C., L. C. Lopez, and B. D. Shur. 1993. Dominant negative mutation in cell surface beta 1,4-galactosyltransferase inhibits cell–cell and cell–matrix interactions. J. Cell Biol. 120:1045–1057.
Maillet, C. M., and B. D. Shur. 1994. Perturbing cell surface beta-(1,4)-galactosyltransferase on F9 embryonal carcinoma cells arrests cell growth and induces laminin synthesis. J. Cell Sci. 107(Pt 6):1713–1724.
Miller, D. J., M. B. Macek, and B. D. Shur. 1992. Complementarity between sperm surface beta-1,4-galactosyltransferase and egg-coat ZP3 mediates sperm–egg binding. Nature. 357:589–593. doi:10.1038/357589a0.
Asano, M., S. Nakae, N. Kotani, N. Shirafuji, A. Nambu, and N. Hashimoto. 2003. Impaired selectin-ligand biosynthesis and reduced inflammatory responses in beta-1,4-galactosyltransferase-I-deficient mice. Blood. 102:1678–1685. doi:10.1182/619blood-2003-03-0836.
Mori, R., T. Kondo, T. Nishima, and M. Asano. 2004. Impairment of skin wound healing in β-1,4-galactosyltransferase-deficient mice with reduced leukocyte recruitment. Am. J. Pathol. 164:1303–1314.
Shen, A., H. Wang, Y. Zhang, J. Yan, D. Zhu, and J. Gu. 2002. Expression of beta-1,4-galactosyltransferase ii and v in rat injured sciatic nerves. Neurosci. Lett. 327:45–48. doi:S0304394002003816[pii].
Shen, A., J. Chen, J. Qian, J. Zhu, L. Hu, M. Yan, D. Zhou, Y. Gao, J. Yang, F. Ding, and C. Cheng. 2009. Elevated beta1,4-galactosyltransferase-i induced by the intraspinal injection of lipopolysaccharide. Glycoconj. J. 26:19–31. doi:10.1007/s10719-008-9158-0.
Liu, J., C. H. Chau, H. Liu, B. R. Jang, X. Li, Y. S. Chan, and D. K. Shum. 2006. Upregulation of chondroitin 6-sulphotransferase-1 facilitates schwann cell migration during axonal growth. J. Cell Sci. 119:933–942. doi:119/5/933[pii]10.1242/jcs.02796.
Gold, R., K. V. Toyka, and H. P. Hartung. 1995. Synergistic effect of IFN-gamma and TNF-alpha on expression of immune molecules and antigen presentation by schwann cells. Cell. Immunol. 165:65–70. doi:S0008-8749(85)71187-2[pii]10.1006/cimm.1995.1187.
Hathaway, H. J., S. C. Evans, D. H. Dubois, C. I. Foote, B. H. Elder, and B. D. Shur. 2003. Mutational analysis of the cytoplasmic domain of beta1,4-galactosyltransferase i: Influence of phosphorylation on cell surface expression. J. Cell Sci. 116:4319–4330. doi:10.1242/jcs.00720 jcs.00720[pii].
Glasgow, L. R., J. C. Paulson, and R. L. Hill. 1977. Systematic purification of five glycosidases from streptococcus (diplococcus) pneumoniae. J. Biol. Chem. 252:8615–8623.
Yan, M., C. Cheng, X. Shao, J. Qian, A. Shen, and C. Xia. 2008. Expression change of beta-1,4 galactosyltransferase I, V mRNAs and Galbeta1,4GlcNAc group in rat sciatic nerve after crush. J. Mol. Histol. 39:317–328.
Begovac, P. C., D. E. Hall, and B. D. Shur. 1991. Laminin fragment E8 mediates PC12 cell neurite outgrowth by binding to cell surface beta 1,4 galactosyltransferase. J. Cell Biol. 113:637–644.
Begovac, P. C., and B. D. Shur. 1990. Cell surface galactosyltransferase mediates the initiation of neurite outgrowth from PC12 cells on laminin. J. Cell Biol. 110:461–470.
Perlin, J. R., and W. S. Talbot. 2007. Putting the glue in glia: Necls mediate schwann cell axon adhesion. J. Cell Biol. 178:721–723. doi:jcb.200708019[pii]10.1083/jcb.200708019.
Papastefanaki, F., J. Chen, A. A. Lavdas, D. Thomaidou, M. Schachner, and R. Matsas. 2007. Grafts of schwann cells engineered to express PSA-NCAM promote functional recovery after spinal cord injury. Brain. 130:2159–2174. doi:awm155[pii]10.1093/brain/awm155.
Constantin, G., L. Piccio, S. Bussini, A. Pizzuti, E. Scarpini, P. Baron, G. Conti, S. Pizzul, and G. Scarlato. 1999. Induction of adhesion molecules on human schwann cells by proinflammatory cytokines, an immunofluorescence study. J. Neurol. Sci. 170:124–130. doi:S0022-510X(99)00202-6[pii].
Jimenez, D., P. Roda-Navarro, T. A. Springer, and J. M. Casasnovas. 2005. Contribution of N-linked glycans to the conformation and function of intercellular adhesion molecules (ICAMs). J. Biol. Chem. 280:5854–5861. doi:M412104200[pii]10.1074/jbc.M412104200.
Furukawa, K., and T. Sato. 1999. Beta-1,4-galactosylation of N-glycans is a complex process. Biochim. Biophys. Acta. 1473:54–66. doi:S0304-4165(99)00169-5[pii].
Zhao, Y., Y. Sato, T. Isaji, T. Fukuda, A. Matsumoto, E. Miyoshi, J. Gu, and N. Taniguchi. 2008. Branched N-glycans regulate the biological functions of integrins and cadherins. FEBS J. 275:1939–1948. doi:EJB6346[pii]10.1111/j.1742-4658.2008.06346.x.
Kido, M., and M. Shibuya. 1998. Isolation and characterization of mouse ovarian surface epithelial cell lines. Pathol. Res. Pract. 194:725–730.
Semel, A. C., E. C. Seales, A. Singhal, E. A. Eklund, K. J. Colley, and S. L. Bellis. 2002. Hyposialylation of integrins stimulates the activity of myeloid fibronectin receptors. J. Biol. Chem. 277:32830–32836. doi:10.1074/jbc.M202493200M202493200[pii].
Isaji, T., Y. Sato, Y. Zhao, E. Miyoshi, Y. Wada, N. Taniguchi, and J. Gu. 2006. N-glycosylation of the beta-propeller domain of the integrin alpha5 subunit is essential for alpha5beta1 heterodimerization, expression on the cell surface, and its biological function. J. Biol. Chem. 281:33258–33267. doi:M607771200[pii]10.1074/jbc.M607771200.
Moss, L., A. Prakobphol, T. W. Wiedmann, S. J. Fisher, and C. H. Damsky. 1994. Glycosylation of human trophoblast integrins is stage and cell-type specific. Glycobiology. 4:567–575.
Wadsworth, S., M. J. Halvorson, A. C. Chang, and J. E. Coligan. 1993. Multiple changes in vla protein glycosylation, expression, and function occur during mouse t cell ontogeny. J. Immunol. 150:847–857.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Huiguang Yang and Ling Hu contributed equally to this work.
Grant sponsor: National Natural Science Foundation of China (no.30770488 and no. 30870320); Natural Science Foundation of Jiangsu province (no.BK2006547); Health Project of Jiangsu Province (H200632).
Rights and permissions
About this article
Cite this article
Yang, H., Hu, L., Chen, J. et al. Lipopolysaccharide Induced Upregulation of β-1,4-Galactosyltransferase-I in Schwann cell. Inflammation 32, 279–286 (2009). https://doi.org/10.1007/s10753-009-9131-5
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10753-009-9131-5