Abstract
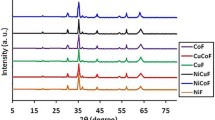
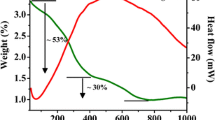
Tetragonal CuFe2O4 is prepared using solid-state techniques assisted by a low-energy ball milling procedure, starting from cuprous oxide (CuO) and hematite (α-Fe2O3). The crystalline material was studied by Powder XRD analysis, ATR-FTIR, and room temperature Mössbauer spectroscopy showed that tetragonal CuFe2O4 is an inverse spinel. The formation of CuFe2O4 was favoured by milling. FESEM indicates the occurrence a distribution of the particles of irregular grain agglomeration. In the EDS scanning of the tetragonal CuFe2O4 pure samples, the Cu and Fe are homogeneously dispersed, and the Fe/Cu proportion is close to 2. Cyclic voltammetry studies were conducted using the voltammetry of immobilised microparticles technique, with platinum as a counter electrode, Ag/AgCl as a reference, and CuFe2O4 NPs over carbon paste as the working electrode. Pure copper ferrites exhibit two anodic peaks and two cathodic peaks in an acidic solution. A capacitor's behaviour is exhibited in alkaline electrolytes.









Similar content being viewed by others
Data availability
No datasets were generated or analysed during the current study.
References
Balagurov, A.M., Bobrikov, I.A.M., Pomjakushin, VYu., Sheptyakov, D.V., Yushankhai, VYu.: Interplay between structural and magnetic phase transitions in copper ferrite studied with high-resolution neutron diffraction. J. Magn. Magn. Mater. 374, 591–599 (2015)
Rocha, A.K.S., Magnago, L.B., Pegoretti, V.C.B., de Freitas, M.B.J.G., Lelis, Fabris, J. D., Porto, A.O.: Copper local structure in spinel ferrites determined by x-ray absorption and Mössbauer spectroscopy and their catalytic performance. Mater. Res. Bull. 109, 117–123 (2019)
Rosa, J.C., Rubí, M.S.: Influence of the synthesis route in obtaining the cubic or tetragonal copper ferrite phases. Inorg. Chem. 59(13), 8775–8788 (2020)
Tasca, J.E., Quincoces, C.E., Lavat, A., Alvarez, A.M., Gloria González, M.: Preparation and characterization of CuFe2O4 bulk catalysts. Ceram. Internat. 37, 803–812 (2011)
Rocha, A.K.S., Magnago, L.B., Santos, J.J., Leal, V.M., Marins, A.A.L., Pegoretti, V.C.B., de Ferreira, S.A.D., Lelis, M.F.F., Freitas, M.B.J.G.: Copper ferrite synthesis from spent Li-ion batteries for multifunctional application as catalyst in photo Fenton process and as electrochemical pseudocapacitor. Mater. Res. Bull. 113, 231–240 (2019)
Covaliu, I.C., Neamtu, J., Georgescu, G., Malaeru, T., Cristea, C., Jitaru, I.: Synthesis and characterization of ferrites (Fe3O4/CuFe2O4)-calcium alginate hybrids for magnetic resonance imaging. Dig. J. Nanomater. Biostruct. 6, 245–252 (2015)
Yang, H., Yan, J., Lu, Z., Cheng, X., Tang, Y.: Photocatalytic activity evaluation of tetragonal CuFe2O4 nanoparticles for the H2 evolution under visible light irradiation. J. Alloys Compd. 476, 715–719 (2009)
Fu, Y., Chen, Q., He, M., Wan, Y., Sun, X., Xia, H., Wang, X.: Copper ferrite-graphene hybrid: a multifunctional hetero-architecture for photocatalysis and energy storage. Ind. Eng. Chem. Res. 51, 11700–11709 (2012)
Kumari, S., Manglam, M.K., Pradhan, L.K., Kumar, L., Borah, J.P., Kar, M.: Modification in crystal structure of copper ferrite fiber by annealing and its hyperthermia application. Appl. Phys. A 127, 273 (2021). https://doi.org/10.1007/s00339-021-04429-5
Roy, S., Ghose, J.: Superparamagnetic nanocrystalline CuFe2O4. J. Appl. Phys. 87(9), 6226 (2000)
Stewart, S.J., Tueros, M.J., Cernicchiaro, G., Scorzelli, R.B.: Magnetic size growth in nanocrystalline copper ferrite. Solid State Commun. 129, 347–351 (2004)
Stewart, S.J., Mercader, R.C., Punte, G., Desimoni, J., Cernicchiaro, G., Scorzelli, R.B.: Shifting the superparamagnetic limit of nanosized copper iron spinel. Hyperfine Interact. 156, 89–95 (2004)
Vittorio Berbenni, V., Marini, A., Milanese, C., Bruni, G.: Solid state synthesis of CuFe2O4 from Cu(OH)2 ∙CuCO3-4FeC2O4 ∙2H2O mixtures: mechanism of reaction and thermal characterization of CuFe2O4. J. Therm. Anal. Calorim. 99, 437–442 (2010)
Goya, G.F., Rechenberg, H.R., Jiang, J.Z.: Structural and magnetic properties of ball milled copper ferrite. J. Appl. Phys. 84, 1101–1108 (1998)
Marinca, T.F., Chicinas, I., Isnard, O.: Structural and magnetic properties of the copper ferrite obtained by reactive milling and heat treatment. Ceram. Int. 39(4), 4179–4186 (2013)
Salavati-Niasari, M., Mahmoudi, T., Sabet, M., Hosseinpour-Mashkani, S.M., Soofivand, F., Tavakoli, F.: Synthesis and characterization of copper ferrite nanocrystals via coprecipitation. J. Cluster Sci. 23, 1003–1010 (2012)
Kanagaraj, M., Sathishkumar, P., Selvan, G.K., Kokila, P., Arumugam, S.: Structural and magnetic properties of CuFe2O4 as-Prepared and thermally treated spinel nanoferrites. Indian J. Pure Appl. Phys. 52, 124–130 (2014)
Lv, W., Liu, B., Luo, Z., Ren, X., Zhang, P.: XRD studies on the nanosized copper ferrite powders synthesized by sonochemical method. J. Alloys Compd. 2008(465), 261–264 (2008)
Mir, N., Salavati-Niasari, M., Davar, F.: Preparation of ZnO nanoflowers and zn glycerolate nanoplates using inorganic precursors via a convenient rout and application in dye sensitized solar cells. Chem. Eng. J. 181–182, 779–789 (2012)
Kalam, A., Al-Sehemi, A.G., Assiri, M., Du, G., Ahmad, T., Ahmad, I., Pannipara, M.: Modified solvothermal synthesis of cobalt ferrite (CoFe2O4) magnetic nanoparticles photocatalysts for degradation of methylene blue with H2O2 /visible light. Results Phys. 8, 1046–1053 (2018)
Kurian, J., Mathew, M.J.: Structural, optical and magnetic studies of CuFe2O4, MgFe2O4 and ZnFe2O4 nanoparticles prepared by hydrothermal/solvothermal method. J. Magn. Magn. Mater. 451, 121–130 (2018)
Zakiyah, L.B., Saion, E., Al-Hada, N.M., Gharibshahi, E., Salem, A., Soltani, N., Gene, S.: Up-scalable synthesis of size-controlled copper ferrite nanocrystals by thermal treatment method. Mater. Sci. Semicond. Process. 40, 564–569 (2015)
López-Ramón, M.V., Álvarez, M.A., Moreno-Castilla, C., Fontecha-Cámara, M.A., Yebra-Rodríguez, Á., Bailón-García, E.: Effect of calcination temperature of a copper ferrite synthesized by a sol-gel method on its structural characteristics and performance as Fenton catalyst to remove gallic acid from water. J. Colloid Interface Sci. 511, 193–202 (2018)
Zhuravlev, V.A., Minin, R.V., Itin, V.I., Lilenko, I.Y.: Structural parameters and magnetic properties of copper ferrite nanopowders obtained by the sol-gel combustion. J. Alloys Compd. 692, 705–712 (2017)
Satheeshkumar, M.K., Ranjith Kumar, E., Srinivas, C., Prasad, G., Meena, S.S., Pradeep, I., Suriyanarayanan, N., Sastry, D.L.: Structural and magnetic properties of CuFe2O4 ferrite nanoparticles synthesized by cow urine assisted combustion method. J. Magn. Magn. Mater. 484, 120–125 (2017)
Satheeshkumar, M.K., Ranjith Kumar, E., Srinivas, C., Suriyanarayanan, N., Deepty, M., Prajapat, C.L., Rao, T.V.C., Sastry, D.L.: Study of structural, morphological and magnetic properties of ag substituted cobalt ferrite nanoparticles prepared by honey assisted combustion method and evaluation of their antibacterial activity. J. Magn. Magn. Mater. 469, 691–697 (2019)
Zinatloo-Ajabshir, S., Morassaei, M.S., Salavati-Niasari, M.: Facile fabrication of Dy2Sn2O7-SnO2 nanocomposites as an effective photocatalyst for degradation and removal of organic contaminants. J. Colloid Interface Sci. 497, 298–308 (2017)
Masunga, N., Mmelesi, O.K., Kefeni, K.K., Mamba, B.B.: Recent advances in copper ferrite nanoparticles and nanocomposites synthesis, magnetic properties and application in water treatment: review. J. Environ. Chem. Eng. 7, 103179 (2019). https://doi.org/10.1016/j.jece.2019.103179
Calvo-de la Rosa, J., Segarra, M.: Optimization of the synthesis of copper ferrite nanoparticles by a polymer-assisted sol−gel method. ACS Omega 4, 18289–18298 (2019). https://doi.org/10.1021/acsomega.9b02295
Prabhu, D., Narayanasamy, A., Shinoda, K., Jeyadeven, B., Greneche, J.M., Chattopadhyay, K.: Grain size effect on the phase transformation temperature of nanostructured CuFe2O4. J. Appl. Phys. 109, 013532 (2011). https://doi.org/10.1063/1.3493244
Palacio Gómez, C.A., Barrero Meneses, C.A., Matute, A.: Structural parameters and cation distributions in solid state synthesized Ni-Zn ferrites. Mater. Sci. Eng. B 236–237(2018), 48–55 (2018)
Palacio Gómez, C.A., Barrero Meneses, C.A., Jaén, J.A.: Raman, infrared and mössbauer spectroscopic studies of solid-state synthesized ni-zn ferrites. J. Magn. Magn. Mater. 505, 166710 (2020). https://doi.org/10.1016/j.jmmm.2020.166710
Larson, A.C., Von Dreele, R.B.: General Structure Analysis System (GSAS), Los Alamos National Laboratory Report LAUR 86–748 (2004)
Naseri, M.G., Saion, E.B., Ahangar, H.A., Shaari, A.H.: Fabrication, characterization, and magnetic properties of copper ferrite nanoparticles prepared by a simple, thermal-treatment method. Mater. Res. Bull. 48(4), 1439–1446 (2013)
El-Masry, M.M., Ramadan, R.: The effect of CoFe2O4, CuFe2O4 and Cu/CoFe2O4 nanoparticles on the optical properties and piezoelectric response of the PVDF polymer. Appl. Phys. A 128(2), 1–13 (2022)
Al-Kadhi, N.S., Al-Senani, G.M., Almufarij, R.S., Abd-Elkader, O.H., Deraz, N.M.: Green synthesis of nanomagnetic copper and cobalt ferrites using corchorus olitorius. Crystals 13(5), 758 (2023). https://doi.org/10.3390/cryst13050758
Greenwood, N.N., Gibb, T.C.: Mössbauer spectroscopy. Chapman and Hall, London (1971). pp 241
Kohout, J., Kmječ, T., Kučera, M., Závěta, K.: CuFe2O4 thin films: hyperfine interactions of 57Fe nuclei and distribution cations. Conf. Int. Conf. Appl. Phys. Condens. Matter 22, 279–283 (2016)
Alonso, A., Tascón, M., Vázquez, M., Sánchez, P.: Electrochemical study of copper and iron compounds in the solid state by using voltammetry of immobilized microparticles: application to copper ferrite characterization. J. Electroanal. Chem. 566(2), 433–441 (2004)
Mouhandess, M.T., Chassagneux, F., Vittori, O., Accary, A., Reeves, R.: Some theoretical aspects of electrodissolution of iron oxide (α-Fe2O3) in carbon paste electrodes with acidic binder. J. Electroanal. Chem. Interf. Electrochem. 181(1–2), 93–105 (1984)
Barrado, E., Prieto, F., Vega, M., Pardo, R., Medina, J.: Characterization and electrochemical behavior of a copper ferrite obtained by in situ precipitation from aqueous solutions. Electroanalysis 12(5), 383–389 (2000)
Amulya, M.S., Nagaswarupa, H., Kumar, M., Ravikumar, C., Kusuma, K., Prashantha, S.: Evaluation of bifunctional applications of CuFe2O4 nanoparticles synthesized by a sonochemical method. J. Phys. Chem. Solids 148, 109756 (2021). https://doi.org/10.1016/j.jpcs.2020.109756
Ng, E.: Caracterización electroquímica, morfológica y estructural de ferritas de cobre sintetizadas por método sol-gel. Thesis. Universidad de Panamá (2021)
Saikova, S., Pavlikov, A., Karpov, D., Samoilo, A., Kirik, S., Volochaev, M., Trofimova, T., Velikanov, D., Kuklin, A.: Copper ferrite nanoparticles synthesized using anion-exchange resin: influence of synthesis parameters on the cubic phase stability. Materials 16(6), 2318 (2023). https://doi.org/10.3390/ma16062318
Zhang, W., Quan, B., Lee, C., Kang, Y.C., Li, X., Choi, E., Diao, G., Piao, Y.: One-step facile solvothermal synthesis of copper ferrite-graphene composite as a high-performance supercapacitor material. ACS Appl. Mater. 7(4), 2404–2414 (2015)
Encinas, P., Lorenzo, L.N.P., Tascón, M., Vázquez, M., Sánchez-Batanero, P.: Electrochemical study of iron(II) and iron(III) compound mixtures in the solid state. Application to magnetite characterization. J. Electroanal. Chem. 371(1–2), 161–166 (1994)
Sahu, V., Shekhar, S., Ahuja, P., Gupta, G., Singh, S.K., Sharma, R.K., Singh, G.: Synthesis of hydrophilic carbon black; role of hydrophilicity in maintaining the hydration level and protonic conduction. RSC Adv. 3(12), 3917–3924 (2013)
Wu, Q., He, T., Zhang, Y., Zhang, J., Wang, Z., Liu, Y., Zhao, L., Wu, Y., Ran, F.: Cyclic stability of supercapacitors: materials, energy storage mechanism, test methods, and device. J. Mater. Chem. A 9(43), 24094–24147 (2021)
Liang, W., Yang, W., Sakib, S., Zhitomirsky, I.: Magnetic CuFe2O4 nanoparticles with pseudocapacitive properties for electrical energy storage. Molecules 27(16), 5313 (2022). https://doi.org/10.3390/molecules27165313
Acknowledgements
The authors would like to thank Dr. Rolando A Gittens of INDICASAT for the ATR-FTIR and SEM measurements. This work has been partly supported by SENACYT (Grant 050-2021-4-PFID-INF2020-13).
Funding
None.
Author information
Authors and Affiliations
Contributions
JAJ: Conceptualization, Formal analysis, Investigation, Resources, Writing - original draft, Writing - review & editing, Supervision. MC: Methodology, Investigation, Data curation. EC: Formal analysis, Investigation, Resources, Data curation, Writing - review & editing. AM: Formal analysis, Investigation. MD: Methodology, Investigation, Data curation. GC: Formal analysis, Investigation, Resources, Data curation, Writing - review & editing.
Corresponding author
Ethics declarations
Ethical approval
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Jaén, J.A., Coronado, M., Chung, E. et al. Structural and electrochemical characterization of tetragonal copper ferrite nanoparticles. Hyperfine Interact 245, 4 (2024). https://doi.org/10.1007/s10751-024-01848-7
Accepted:
Published:
DOI: https://doi.org/10.1007/s10751-024-01848-7