Abstract
Stygobionts, namely animals with strong adaptations to subterranean environments that are unable to complete their life cycles outside groundwater, can be observed in spring ecotones, but their occurrence is generally considered accidental. The aim of this paper is to assess if stygobiont occurrence in springs is linked to specific environmental conditions or if it is random, irrespective of their features. For three years, we surveyed 59 spring sites recording the occurrence of vertebrate and invertebrate stygobiont species and assessing if spring features were related to their distribution. Moreover, we recorded the escape reactions of two easily identifiable stygobiont species. We detected six taxa usually considered as strictly stygobiont based on their troglomorphic features. Two of them were quite widespread: the salamander Proteus anguinus and the shrimp Troglocaris planinensis. Environmental characteristics were significantly related to the distribution of stygobionts. Hydroperiod and occurrence of flooding were the factors that played the strongest role in affecting occurrence. Our study suggests that the occurrence of stygobionts in springs is linked to specific habitat features rather than being a random mechanism and that the exploitation of ecotones can be important for the lifecycle of some species usually assumed to be strictly associated to caves.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Despite their key role in hydrological systems and river networks, springs are often neglected in ecological and zoological studies. Springs are ecotones that represent the boundary between groundwaters and surface waters. Groundwaters are considered among the most fragile natural environments in the world (Kurwadkar et al., 2020). These environments hold the biggest reservoir of unfrozen freshwater, thus they represent a fundamental resource for life on Earth (Gibert and Deharveng, 2002). Lack of light strongly limits the abundance of primary producers and consequently makes groundwater usually depleted of trophic resources (Barzaghi et al., 2017). At the same time, groundwaters are more stable and safer environments when compared to surface waters. Across the year, they usually experience limited changes in terms of temperature, water level, and chemical features (Riedel, 2019). Furthermore, they are sheltered by the stressing conditions posed by sun radiations or highly oxidative environments and have simplified food webs, thus some groundwater organisms suffer limited predation risk (Lunghi et al., 2016; Mammola et al., 2019; Manenti et al., 2023; Pipan and Culver, 2013). Most subterranean species inhabiting groundwaters are supposed to have fine-tuned their behavioural, physiological, and metabolic responses to the relatively narrow and stable characteristics of their habitats (Culver and Pipan, 2019; Moldovan and Nastase-Bucur, 2019); for this reason, they are also supposed to have lower adaptive potential against environmental changes, compared to species inhabiting surface freshwaters (Friedrich, 2013; Mammola et al., 2019; Manenti and Ficetola, 2013). Animals that live their entire life cycles in groundwaters are called stygobionts (the suffix ‘stygo’ derives from the river Styx, the river flowing in the mythological Greek underworld). They show the highest degree of adaptation to subterranean freshwater environments and are often characterised by a set of traits including blindness, depigmentation, and elongation of appendages (Romero, 2011). Conversely, surface freshwaters are characterised by broader variation of physical features and are generally richer in trophic content, because of both the primary productivity of autotrophic organisms and the subsidisation from the surrounding epigean environments (Barzaghi et al., 2017). Surface habitats thus host a large variety of organisms, some of which may have more or less pronounced adaptations to thrive in spring ecotones and even to colonise groundwaters if conditions are favourable.
The features of both groundwater and surface freshwater interplay in characterising the selective pressures acting on animals exploiting springs (Alfaro and Wallace, 1994; Cantonati et al., 2006). This interplay between subterranean and surface habitats may lead to the formation of heterogeneous environments hosting relatively high species richness (Di Lorenzo et al., 2005; Cantonati et al., 2022). At the same time, ongoing climate change, with its effects like droughts and intense precipitation events, poses major constraints for both surface and subterranean organisms (Di Lorenzo and Galassi, 2017; Wynne et al., 2021). Ecological analyses of springs often focus on epigean organisms, neglecting the roles played by stygobionts and the pressures they face in these environments. The fact that the occurrence of stygobionts in springs could affect both the dynamics of these ecotones and the intrinsic traits of the populations occurring in them, is poorly considered in experimental and field studies. For instance, stygobionts of the genus Niphargus were often reported in springs (Vandel, 1920; Ginet, 1960; Kureck, 1967; Fišer et al., 2007; Manenti and Pezzoli, 2019). However, in the past century, the occurrence of these crustaceans in springs was considered only accidental through drift, with the water flow flushing them out of their primary habitat (Müller et al., 1963; Kureck, 1967; Mathieu et al., 1999; Malard et al., 2002). This even if some author already noticed variation of drifting rate between night and day (Kureck, 1967). More recently, cases of active spring exploitation of these crustaceans have been reported, suggesting that they are likely to affect the dynamics of the ecotones (Manenti and Barzaghi, 2021; Manenti et al., 2023). Similarly, recent observations reported that the olm Proteus anguinus, a highly specialised stygobiont, can regularly occur in surface environments (Manenti et al., 2024), but analyses on the whole communities of springs are extremely scarce.
In this study we assess, at the community level, whether stygobiont occurrence in springs is random or is linked to specific environmental conditions. Community assembly theory affirms that environmental filtering, dispersal limitation, and biotic interactions are the main processes shaping the non-random species assembly (Rosati et al., 2014; Kraft et al., 2015). Lack of clear associations between stygobionts and specific environmental conditions of springs (i.e. random occurrence) would support the idea that stygobionts are occasional inhabitants of surface environments, and their occurrence is not linked to clear ecological processes. Conversely, the identification of factors favouring the occurrence of stygobionts in springs (e.g. water flow, hydroperiod) would point out a role of ecological filters affecting their exploitation of surface habitats, as occurs with surface-dwelling animals. Moreover, if stygobionts actively exploit springs, we predict that in the presence of disturbance, they are able to shelter or move back to groundwater. Conversely, if their occurrence is linked to random or passive mechanisms, we expect a limited ability to re-enter groundwater, if needed. To test our hypotheses, we performed an extensive assessment of communities of springs in a karst area of Eastern Italy where strong interconnections between groundwater and surface freshwaters exist. Teasing apart these hypotheses can have important implications for the comprehension of the adaptive consequences of the environmental filtering acting at the interface between groundwaters and surface waters.
Material and methods
Study area
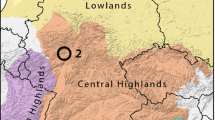
The study area (Fig. 1) is situated between the municipalities of Doberdò del Lago and Monfalcone (NE Italy) in the north-western part of the Classical Karst, which is the northernmost part of the Eastern Adriatic karst territory, including areas made up of limestone and dolomite (Jurkovšek et al., 2016). Here, we sampled sites extending in different poljes and belonging to a system of karst lakes comprising Lake Doberdò, Lake Pietrarossa, Lake Sablici, and Lake Mucille, which were strongly altered by human activities, especially around the 1950s. Karst lakes are typical temporary lakes characterised by strong variations of the hydroperiod and of the water level, which can vary up to several metres and also become completely absent (Knez and Slabe, 2005; Kovačič, 2014). Karst lakes are the expression on the surface of the variations of the karst aquifer close to areas of groundwater discharge (Culver and Pipan, 2014). The hydro-ecological features of the study area allow the occurrence of a wide variety of species and habitats (Castello et al., 2021) including springs, sinkholes, and estavelles (i.e. openings which, depending on aquifer level, can serve either as a spring or as a sink of freshwater). For this study, we sampled both classic springs and estavelles.
Study area. In yellow are underlined the three polje/karst lakes around which we investigated springs. Precise locations of the springs are omitted because of conservation reasons (Lunghi et al., 2019)
Field surveys
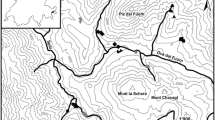
For three years, from June 2020 to June 2023, we surveyed multiple times (minimum: 10, maximum: 24) 59 spring sites (Fig. 2, Supplementary Table 1). Surveys were carried out both during day and night. During each survey, we visually recorded the occurrence of vertebrate and invertebrate aquatic animals, distinguishing those considered strictly stygobionts from those known to live in surface freshwaters. We did not use dip-netting or other sampling methods to avoid disturbance on spring sites in order to not influence stygobionts occurrence in subsequent surveys. Stygobiont species, thanks to their whitish appearance, are easy to spot when active. Their identification was based on existing reports for the groundwater fauna of the study area (Stoch, 2017). Visual surveys lasted 20 min for each spring. During surveys, we also recorded spring site features, such as the area of the spring, the water presence and depth, and the occurrence of macrophytes. In limnocrene springs, we surveyed the spring mouths and estavelles openings. In case of fissures and other openings feeding rheocrene springs, we sampled the habitats for a maximum length downstream of 1 m. During surveys, we also used visual encounter surveys to assess the occurrence of predatory fish species like pike (Esox lucius) gobies (genus Padogobius), and bullheads (genus Cottus).
Examples of springs surveyed. A estavella spring with visible openings/connections with the groundwater level where stygobionts were observed to be able to actively escape when disturbed. B dry estavella spring. C rheocrene stable spring. D, E: example of water level variation in an estavella spring, picture E provides example of a flooded spring where borders are visible but underwater. F shallow rheocrene spring, typically inhabited by Monolistra racovitzai stygobiont crustaceans
Moreover, from October 2022 to March 2023, during night surveys (minimum 1, maximum 5), we recorded in 22 springs the reaction to our presence of two easily identifiable stygobiont species: Proteus anguinus Laurenti, 1768 and Troglocaris planinensis Birštein, 1948 (Supplementary Table 3). Many stygobiont species are known to be often photophobic even when totally eyeless (Vandel, 1964; Galbiati et al., 2023). If their occurrence was not caused by drift alone, we expected that they would avoid our lights during nighttime surveys and re-enter into spring mouth. During each survey, we counted the number of visible individuals and how many of them escaped from the spring into the groundwater during the first five minutes of our survey time (20 min). We avoided small stygobiont species like Monolistra spp. as it would have been difficult to distinguish between their sheltering under stones/vegetation from active re-entry into the spring mouth.
Statistical analyses
We used constrained redundancy analysis (RDA) to evaluate the relative role of spring features on the stygobiont species composition. RDAs are canonical analyses, combining the properties of regression and ordination techniques, that allow evaluating how much of the variation of the structure of one dataset (e.g. community composition in a spring; endogenous dataset) is explained by independent variables (e.g. spring habitat features; exogenous datasets) (Borcard et al., 2011). We built a matrix of environmental features considering the whole study period and composed by: (1) the maximum depth of each spring; (2) if the spring dried or not; (3) the occurrence of aquatic macrophytes; (4) if the springs were subject at least once to flooding (i.e. water level overcame spring mouth/border by at least 10 cm, overcoming spring delimitation); and (5) if the springs hosted predatory fish at least once during the three years of survey. We used the matrix of species composition as endogenous. A species was considered as present if it occurred at least once in a spring during day/night surveys of the whole study period. The RDA analysis was performed for the five commonest stygobiont species found during surveys (see results). Variance partitioning was used to calculate the independent and joint effects of spring features, while the significance of explained variance was calculated by 10,000 permutation tests. RDA analyses were performed using ‘rda’ and ‘permutation’ functions of R package ‘vegan’.
To address factors that could affect the ability of stygobionts to actively escape springs and re-enter groundwater, we used a binomial generalised linear mixed model (GLMM). The dependent variable was the number of escaped/remaining individuals at each survey (Venables and Ripley, 2002). As a fixed factor, we included species identity and water level of the springs during the survey, distinguishing between flooded, stable but not flooded, increasing, and decreasing. Site identity was included as a random factor. We assessed the significance of the fixed factors using likelihood ratio tests (Bolker et al., 2008). We assessed differences between the two species and the three levels with a post hoc Tukey test using the package emmeans.
Results
In springs, we detected six taxa usually considered as strictly stygobiont on the basis of their troglomorphic features and/or of previous studies in the Karst (Fig. 3; Supplementary Table 1). The most common species were the olm, Proteus anguinus (23.3% of the site) and Troglocaris planinensis (43.3%). The crustacean isopod species Monolistra racovitzai and M. schottlanderi were more localised in springs (they occurred in 13.3% and in 5% of the sites, respectively), still they were often observed with high abundances of active individuals. Finally, we reported the occurrence of two troglomorphic blind and depigmented taxa of planarians, one belonging to the genus Dendrocoelum, and one to family Planaridae with size and external morphology resembling the genus Atrioplanaria. The latter was found only on three occasions in two sites, with a record of observing more than 30 individuals simultaneously.
Stygobiont taxa were significantly related to spring features (F = 3.01; P < 0.001). Recorded environmental features explained the 22.15% of the variation of stygobiont communities. The first RDA axis was related to springs not subjected to flooding, that are perennial, with predators and macrophytes, while the second RDA axis was associated with temporary springs without predators (Fig. 4; Supplementary Table 2). Proteus anguinus was related to temporary springs without predators and subjected to flooding. Troglocaris planinensis was associated with slow flowing springs subjected to flooding, but perennial. Both Monolistra isopod species were positively associated with the first RDA axis. No strong relationships were evidenced for Dendrocoelum sp. planarians.
Stygobionts were able to escape the spring mouth, sheltering towards groundwater. On average (± SE) 99 ± 1% of P. anguinus individuals and 69.4 ± 4.7% of Troglocaris planinensis individuals escaped to underground shelters within five minutes after we approached springs (Supplementary Table 3). The rate of escape was significantly different between species (\(\chi \)2 = 9.17, P < 0.01) and was related to the trend of spring water level (\(\chi \)2 = 15.08, P < 0.01). Post hoc Tukey test showed that P. anguinus had higher escape rates than T. planinensis. For both species, escape rates were higher when the springs were flooded or stable than when the water level was decreasing (Table 1).
Discussion
This is one of the few studies assessing the occurrence of stygobiont species in springs at the community level. The presence in springs of groundwater-dwelling animals with pronounced adaptations to live underground is well reported (Lescher-Moutoué, 1973; Rouch, 1986; Malard et al., 2002; ; Cantonati et al., 2011; Aljančič, 2019 ). However, these reports occur often in the grey literature or as uncritical associations with passive drift (Manenti and Piazza, 2021). Drift and passive mechanisms, especially after heavy rains, can explain stygobiont observations in surfaces where they occur without the possibility of returning to the underground environment (Husmann, 1976; Mori et al., 2015). However, in our study area, the distribution of stygobiont species was significantly related to specific environmental features, rather than being a passive random mechanism, and was characterised by the ability to quickly return to groundwater under disturbance.
Occurrence of stygobionts in springs was quite widespread, with both iconic subterranean vertebrates like the olm, Proteus anguinus and invertebrates not studied in surface environments like the crustaceans Troglocaris planinensis and Monolistra racovitzai. Moreover, we observed unidentified species of planarians, possibly not yet described.
The main drivers of stygobionts occurrence were related to spring hydroperiod (temporary or perennial), spring flooding, and predator occurrence. Both P. anguinus and T. planinensis were associated with springs subject to flooding. Since stygobionts in spring habitats are often reported during floodings (Bressi et al., 1999; Malard et al., 2002; Aljančič, 2019; Manenti and Piazza, 2021), the association of stygobionts with springs subject to flooding could suggest that they are the result of passive drift from groundwater, pointing out a role for passive process. However, in our surveys the occurrence of stygobionts in springs was frequent across the study area and did not occur only during floodings. Most of these ecotones are estavelles, i.e. depressions that, when the water level of the polje raises, are very slowly flooded, generally without strong water flow. Both P. anguinus and T. planinensis preferred slow flowing springs that form a small pond-like habitat when water level rises. In this perspective, our results provide a possible explanation for the fact that most stygobiont observations in surface habitats are reported during floodings. Stygobionts may exploit resources that become available during such events, but this increases the possibility of drift or being spotted by researchers. The active exploitation of surface environments by stygobionts is also supported by the observation that, even during floods, stygobionts are able to shelter in groundwater once disturbed, and behavioural responses were stronger when water level was declining. This is supported by studies on stygobionts in springs, which showed that Niphargus crustaceans, once drifted, are able to actively migrate back to groundwater (Müller et al., 1963; Kureck, 1967), and by short-term observations in Slovenia suggesting that P. anguinus observed in surface habitats are able to re-enter groundwater (Premate et al., 2022). Moreover, the troglomorphic amphipod Niphargus valachicus is known to enter the surface waters, likely to exploit the available food resources (Copilaş-Ciocianu et al., 2018; Markovic et al., 2018). Our results thus underline that, at the interface between groundwater and surface, at least some species usually considered as a strict stygobiont taxon may find specific resources and even extend their ecological niche towards the surface, with implications for spring communities. In spring ecotones, flooding events can allow stygobionts to exploit resources that are usually stored in terrestrial environments. When groundwater reaches soils that border temporary spring mouths, soil animals are forced to exit from soil as air-breathing becomes impossible, and thus can be easily captured by spring-dwelling predators. Moreover, terrestrial prey not exploited by surface-dwelling species may become available to subterranean predators, as suggested by the observations that P. anguinus prefers not only sites that are subject to flooding, but also temporary springs, where it can feed on earthworms (Manenti et al., 2024). Recent studies have highlighted that springs are complex ecotones involving more than just the groundwater runoff (Reiss and Chifflard, 2017; Cantonati et al., 2020), and that some subterranean animals exploiting springs can retrieve most of their food from organic matter of terrestrial origin (Nair et al., 2021). Our results call for further studies on the diet of P. anguinus both in springs and in subterranean environments with varying water level, and suggest that the inputs provided by the multiple surface habitats that border groundwater should be included when studying the biology of subterranean animals.
Our results reveal also that, at least in the case of P. anguinus, the use of temporary springs can also be linked to the necessity to avoid predator fish like the pike that usually occur in stable freshwater environments. These results suggest that further investigations could try to assess responses of stygobionts towards surface predators and the role played by the landscape of fear in affecting freshwater surface environments and exploitations by stygobiont species.
Contrary to P. anguinus, T. planinensis atyids were related to perennial springs, suggesting that also these environments can provide important trophic resources, and highlighting that different stygobionts can exploit different microhabitats. Other important stygobiont components of springs communities included Monolistra crustacean isopods, which seemed to avoid springs subjected to flooding and prefer shallow sites, but did not show particularly strong relationships with the features assessed in this study. Monolistra isopods are scrapers that can reach high abundances in subterranean habitats (Sket, 1997). Their biomass and role as scrapers can be important also in the dynamics of spring communities and should be investigated in the future.
The fact that stygobiont occurrence in springs is linked to specific habitat features implies that epigean habitat filtering plays a role, and their occurrence is not just determined by random or passive mechanisms. It might be argued that springs are simple extensions of the stygobionts’ primary habitat, especially during nighttime. However, our study highlights a role of multiple environmental features suggesting complex patterns of occurrence of stygobionts at the boundary between groundwaters and the surface. Thus, studying groundwater environments without recognising the functional role of their ecotones could hamper a complete understanding of stygobionts’ biology and of the functioning of these critical ecosystems.
Data availability
All data on the measured springs variables indicating stygobiont occurrence/behaviour that support the findings of this study are included within this paper and its Supplementary Information files.
References
Alfaro, C. & M. Wallace, 1994. Origin and classification of springs and historical review with current applications. Environ Geol 24(2): 112–124.
Aljančič, G., 2019. History of research on Proteus anguinus Laurenti 1768 in Slovenia. Folia Biologica Et Geologica 60: 39–69.
Barzaghi, B., G. F. Ficetola, R. Pennati & R. Manenti, 2017. Biphasic predators provide biomass subsidies in small freshwater habitats: A case study of spring and cave pools. Freshw Biol 62(9): 1637–1644. https://doi.org/10.1111/fwb.12975.
Borcard, D., F. Gillet & P. Legendre, 2011. Numerical Ecology with R, Springer, New York:
Bressi, N., M. Aljancic & L. Lapini, 1999. Notes on presence and feeding of Proteus anguinus Laurenti, 1768 outside caves. Riv Idrobiol 38: 431–435.
Cantonati, M., R. Gerecke & E. Bertuzzi, 2006. Springs of the Alps – sensitive ecosystems to environmental change: from biodiversity assessments to long-term studies. Hydrobiologia 562: 59–96.
Cantonati, M., R. Gerecke, I. Juttner & E. J. Cox, 2011. Springs: neglected key habitats for biodiversity conservation Introduction to the Special Issue. J Limnol 70:1–1.
Cantonati, M., L. E. Stevens, S. Segadelli, A. E. Springer, N. Goldscheider, F. Celico, M. Filippini, K. Ogata & A. Gargini, 2020. Ecohydrogeology: The interdisciplinary convergence needed to improve the study and stewardship of springs and other groundwater-dependent habitats, biota, and ecosystems. Ecol Indic 110 https://doi.org/10.1016/j.ecolind.2019.105803.
Cantonati, M., K. Lichtenwöhrer, G. Leonhardt, L. Seifert, A. Mustoni, R. Hotzy, E. Schubert, L. Blattner, O. Bilous, A. Lotz, B. Poschlod & R. Gerecke, 2022. Using Springs as Sentinels of Climate Change in Nature Parks North and South of the Alps: A Critical Evaluation of Methodological Aspects and Recommendations for Long-Term Monitoring Water. vol 14.
Castello, M., L. Poldini & A. Altobelli, 2021. The aquatic and wetland vegetation of Lake Doberdò: an analysis for conservation value assessment of a disappearing lake of the Classical Karst (North East Italy). Plant Sociology 58: 75–106.
Copilaş-Ciocianu, D., C. Fišer, P. Borza & A. Petrusek, 2018. Is subterranean lifestyle reversible? Independent and recent large-scale dispersal into surface waters by two species of the groundwater amphipod genus Niphargus. Molecular Phylogenetics and Evolution 119: 37–49. https://doi.org/10.1016/j.ympev.2017.10.023.
Culver, D. C. & T. Pipan, 2014. Shallow Subterranean Habitats: Ecology, Evolution, and Conservation, Oxford University Press, New York, NY, USA:
Culver, D. C. & T. Pipan, 2019. Biology of Caves and Other Subterranean Habitats, 2nd Edition.
Di Lorenzo, T. & D. M. P. Galassi, 2017. Effect of Temperature Rising on the Stygobitic Crustacean Species Diacyclops belgicus: Does Global Warming Affect Groundwater Populations? Water 9(12).
Fišer, C., R. Keber, V. Kerezi, A. Moskric, A. Palandancic, V. Petkovska, H. Potocnik & B. Sket, 2007. Coexistence of species of two amphipod genera: Niphargus timavi (Niphargidae) and Gammarus fossarum (Gammaridae). J Nat Hist 41(41–44): 2641–2651.
Friedrich, M., 2013. Biological Clocks and Visual Systems in Cave-Adapted Animals at the Dawn of Speleogenomics. Integr Comp Biol 53(1): 50–67.
Galbiati, M., S. Lapadula, M. Forlani, B. Barzaghi & R. Manenti, 2023. Both Light Stimuli and Predation Risk Affect the Adult Behavior of a Stygobiont Crustacean Diversity. vol 15.
Gibert, J. & L. Deharveng, 2002. Subterranean ecosystems: A truncated functional biodiversity. Bioscience 52(6): 473–481. https://doi.org/10.1641/0006-3568(2002)052[0473:seatfb]2.0.co;2.
Ginet, R., 1960. Ecologie, éthologie et biologie de" Niphargus"(Amphipodes Gammaridés hypogés). Ann Speleol 15: 127–237.
Husmann, S., 1976. Studies on Subterranean Drift of Stygobiont Crustaceans (Niphargus, Crangonyx, GraeterielIa). Int J Speleol 8: 81–92.
Jurkovšek, B., S. Biolchi, S. Furlani, T. Kolar-Jurkovšek, L. Zini, J. Jež, G. Tunis, M. Bavec & F. Cucchi, 2016. Geology of the Classical Karst Region (SW Slovenia–NE Italy). Journal of Maps 12(sup1): 352–362. https://doi.org/10.1080/17445647.2016.1215941.
Knez, M. & T. Slabe, 2005. Lithostratigraphic characteristics of the intermittent Pivka lakes region and Matijeva jama cave estavelle. Acta Carsol 34(3): 582–598.
Kovačič, G., 2014. The Šembije Intermittent Lake. Geografski Obzornik 61(4): 13–21.
Kraft, N. J. B., P. B. Adler, O. Godoy, E. C. James, S. Fuller & J. M. Levine, 2015. Community assembly, coexistence and the environmental filtering metaphor. Funct Ecol 29(5): 592–599. https://doi.org/10.1111/1365-2435.12345.
Kureck, A., 1967. Über die tagesperiodische ausdrift von Niphargus aquilex schellenbergi Karaman aus Quellen. Zeitschrift Für Morphologie und Ökologie der Tiere 58: 247–262.
Kurwadkar, S., S. R. Kanel & A. Nakarmi, 2020. Groundwater pollution: Occurrence, detection, and remediation of organic and inorganic pollutants. Water Environ Res 92: 1659–1668. https://doi.org/10.1002/wer.1415.
Lescher-Moutoué, F., 1973. Sur la biologie et l’écologie des copépodes cyclopides hypogés. Ann Speleol 28(3): 429–502.
Di Lorenzo, T., D. Cipriani, P. Bono, L. Rossini, P. De Laurentiis, B. Fiasca, C. Pantani & D. M. P. Galassi, 2005. Dynamics of groundwater copepod assemblages from the Mazzoccolo karstic spring (central Italy). Meiofauna Marina 14: 97–103.
Lunghi, E., C. Corti, R. Manenti & G. F. Ficetola, 2019. Consider species specialism when publishing datasets. Nat Ecol Evol 3(3): 319. https://doi.org/10.1038/s41559-019-0803-8.
Lunghi, E., R. Manenti, G. Canciani, G. Scari, R. Pennati & G. F. Ficetola, 2016. Thermal equilibrium and temperature differences among body regions in European plethodontid salamanders. J Therm Biol 60: 79–85. https://doi.org/10.1016/j.jtherbio.2016.06.010.
Malard, F., M. J. Dole-Olivier, J. Mathieu & F. Stoch, 2002. Sampling Manual for the Assessment of Regional Groundwater Biodiversity. PASCALIS Project, Lyon.
Mammola, S., E. Piano, P. Cardoso, P. Vernon, D. Dominguez & M. Isaia, 2019. Climate change going deep: the effects of global climatic alterations on cave ecosystems. The Anthropocene Review 6: 98–116.
Manenti, R. & B. Barzaghi, 2021. Diel activity of Niphargus amphipods in spring habitats. Crustaceana 94(6): 705–721.
Manenti, R. & G. F. Ficetola, 2013. Salamanders breeding in subterranean habitats: local adaptations or behavioural plasticity? J Zool 289(3): 182–188. https://doi.org/10.1111/j.1469-7998.2012.00976.x.
Manenti, R. & E. Pezzoli, 2019. Think of what lies below, not only of what is visible above, or: a comprehensive zoological study of invertebrate communities of spring habitats. Eur Zool J 86(1): 272–279. https://doi.org/10.1080/24750263.2019.1634769.
Manenti, R. & B. Piazza, 2021. Between darkness and light: spring habitats provide new perspectives for modern researchers on groundwater biology. PeerJ 9: e11711. https://doi.org/10.7717/peerj.11711.
Manenti, R., M. Galbiati, S. Lapadula, M. Forlani, B. Barzaghi, A. Melotto & G. F. Ficetola, 2023. Behavioural drivers of ecotone exploitation: activity of groundwater animals in spring. Behav Ecol Sociobiol 77(2): 23. https://doi.org/10.1007/s00265-023-03297-8.
Manenti, R., M. R. Di Nicola, V. Zampieri, G. Grassi, T. Creanza, E. Mauri, G. F. Ficetola & B. Barzaghi, 2024. Wandering outside of the Styx: Surface activity of an iconic subterranean vertebrate, the olm (Proteus anguinus). Ecology 105(3): e4252. https://doi.org/10.1002/ecy.4252.
Markovic, V., B. Novakovic, M. Ilic & V. Nikolic, 2018. Epigean niphargids in Serbia: New Records of Niphargus valachicus Dobreanu & Manolache, 1933 (Amphipoda: Niphargidae), with Notes on its ecological preferences. Acta Zool Bulg 70(1): 45–50.
Mathieu, J., K. Essafi & H. Chergui, 1999. Spatial and temporal variations of stygobite Amphipod populations in interstitial aquatic habitats of karst floodplain interfaces in France and Morocco. Annales De Limnologie-International Journal of Limnology 35(2): 133–139. https://doi.org/10.1051/limn/1999018.
Moldovan, O. T. & R. Nastase-Bucur, 2019. Cave Biology. In Ponta, G. M. L. & B. P. Onac (eds) Cave and Karst Systems of Romania. Cave and Karst Systems of the World, 485–492.
Mori, N., T. Kanduč, M. Opalički Slabe & A. Brancelj, 2015. Groundwater Drift as a Tracer for Identifying Sources of Spring Discharge. Groundwater 53(S1): 123–132. https://doi.org/10.1111/gwat.12314.
Müller, K., A. Kureck & A. Müller-Haeckel, 1963. Zur Tagesperiodik von Niphargus aquilex schellenbergi Karaman (Gammaridae, Amphipoda). Naturwissenschaften 50: 579–580.
Nair, P., P. H. Diaz & W. H. Nowlin, 2021. Interactions at surface–subterranean ecotones: structure and function of food webs within spring orifices. Oecologia 196(1): 235–248. https://doi.org/10.1007/s00442-021-04912-z.
Pipan, T. & D. C. Culver, 2013. Forty years of epikarst: what biology have we learned? Int J Speleol 42: 215–223. https://doi.org/10.5038/1827-806X.42.3.5.
Premate, E., Ž Fišer, Ž Kuralt, A. Pekolj, T. Trajbarič, E. Milavc, Ž Hanc & R. Kostanjšek, 2022. Behavioral observations of the olm (Proteus anguinus) in a karst spring via direct observations and camera trapping. Subterranean Biology 44: 69–83.
Reiss, M. & P. Chifflard, 2017. An Opinion on Spring Habitats within the Earth’s Critical Zone in Headwater Regions. Water 9(9): 645.
Riedel, T., 2019. Temperature-associated changes in groundwater quality. Journal of Hydrology 572: 206–212. https://doi.org/10.1016/j.jhydrol.2019.02.059.
Romero, A., 2011. The Evolution of Cave Life. American Scientist 99(2): 144–151. https://doi.org/10.1511/2011.89.144.
Rosati, M., M. Cantonati, R. Primicerio & G. Rossetti, 2014. Biogeography and relevant ecological drivers in spring habitats: A review on ostracods of the western palearctic. Int Rev Hydrobiol 99(6): 409–424. https://doi.org/10.1002/iroh.201301726.
Rouch, R., 1986. Sur l’écologie des eaux souterraines dans le karst. Stygologia 2: 352–398.
Sket, B., 1997. Distribution of Proteus (Amphibia: Urodela: Proteidae) and its possible explanation. J Biogeogr 24(3): 263–280. https://doi.org/10.1046/j.1365-2699.1997.00103.x.
Stoch, F., 2017. Il Lacus Timavi: la fauna acquatica sotterranea, con particolare riguardo alle risorgive del Fiume Timavo. Atti e Memorie della Commissione Grotte “E Boegan” 47:173 - 203.
Vandel, A., 1920. Sur la faune des sources. Bull Soc Zool Fr 45: 177–183.
Vandel, A., 1964. Biospeleologie: la biologie des animaux cavernicoles, vol. XVIII. Gauthiers-Villars, Paris:
Venables, W. N. & B. D. Ripley, 2002. Modern Applied Statistics with S. Fourth edition. Springer.
Wynne, J. J., F. G. Howarth, S. Mammola, R. L. Ferreira, P. Cardoso, T. D. Lorenzo, D. M. P. Galassi, R. A. Medellin, B. W. Miller, D. Sánchez-Fernández, M. E. Bichuette, J. Biswas, C. W. BlackEagle, C. Boonyanusith, I. R. Amorim, P. A. V. Borges, P. J. Boston, R. N. Cal, N. Cheeptham, L. Deharveng, D. Eme, A. Faille, D. Fenolio, C. Fišer, Ž. Fišer, S. M. ʻOhukaniʻōhiʻa Gon, F. Goudarzi, C. Griebler, S. Halse, H. Hoch, E. Kale, A. D. Katz, Ľ. Kováč, T. M. Lilley, S. Manchi, R. Manenti, A. Martínez, M. B. Meierhofer, A. Z. Miller, O. T. Moldovan, M. L. Niemiller, S. B. Peck, T. G. Pellegrini, T. Pipan, C. M. Phillips-Lander, C. Poot, P. A. Racey, A. Sendra, W. A. Shear, M. S. Silva, S. Taiti, M. Tian, M. P. Venarsky, S. Y. Pakarati, M. Zagmajster & Y. Zhao, 2021. A conservation roadmap for the subterranean biome. Conservation Letters 14(5) https://doi.org/10.1111/conl.12834.
Acknowledgements
We are grateful to Edgardo Mauri of Speleoviviarium of Trieste, Marco Restaino of Società Adriatica di Speleologia and Gaia Fior, for help in locating sites and fruitful discussions. We thank Tiziano Fiorenza, Gerardo Sarno of PAVEES cooperative, Lucia Luciani of Club Alpino Italiano, section Monfalcone, and the direction of the stables Centro Ippico Pietra Rossa for logistical help during field surveys. We thank Clara Citterio for botanical investigations. Research on P. anguinus was authorised by the ‘Ministero della Transizione Ecologica’, permits number 6957 of 13 February 2020 and 3267 of 15 March 2022. We are grateful to two anonymous reviewers and G. M. Bucciarelli for comments on an early version of the manuscript.
Funding
Open access funding provided by Università degli Studi di Milano within the CRUI-CARE Agreement.
Author information
Authors and Affiliations
Contributions
Conceptualisation was done by RM, BB, and GFF. RM and MF were involved in data analysis. VZ, GP, FC, MG, SL, MG, VM, VB, RM, and BB helped in field surveys. Writing was done by RM (first draft), GFF, VZ, and SL; all the authors reviewed, commented, and edited drafts of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
Authors declare no conflict of interest.
Additional information
Handling editor: Gary Bucciarelli.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Manenti, R., Zampieri, V., Pacinotti, G. et al. Back from the underworld: the exploitation of spring habitats by stygobiont species. Hydrobiologia (2024). https://doi.org/10.1007/s10750-024-05638-8
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10750-024-05638-8