Abstract
Understanding fish movement in river systems is essential in identifying habitats which are important for species to perpetuate. Water chemistry, paired with fish otoliths, can be used to trace movements among different habitats, although a detailed understanding of spatial variation in water chemistry is first required. To obtain this knowledge, we analysed surface water collected from 59 sites throughout the Clarence River Basin, a coastal river basin in northern New South Wales, Australia. The primary objective was to quantify the spatial variation of trace metals and strontium isotope ratios (87Sr:86Sr) for future and paired use with otolith microchemistry to track fish movement. Using ICP-OES, we identified that some trace metals, particularly strontium and barium as well as 87Sr:86Sr ratios, varied spatially when analysing all water samples and revealed three distinct regions centring around the estuary, interior and outer regions. These differences were driven by variation in the underlying surface geology between the Clarence–Moreton Basin and the New England Block. Tidal influence also affected trace metals and 87Sr:86Sr ratios throughout the estuarine area. We suggest that the level of differentiation identified in the current study has the potential to discriminate broad-scale fish movements among regions/habitats using otolith analysis, with fine scale movements best elucidated using complementary techniques.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Freshwater Fish move over a range of spatial scales, from small, localised movements to large oceanic migrations, allowing individuals to access habitats that optimize growth, survival and reproduction (Kahler et al. 2001; Lucas and Baras 2001; Cooke et al. 2022). As fish move through different habitats, dissolved minerals in the water column are incorporated into the individual’s bony structures, including the otoliths (Fowler et al. 1995; Gallahar & Kingsford 1996). Otoliths, a group of inner ear bones of fish, are made up primarily of calcium carbonate and aid in balance and sound reception (Degens et al. 1969; Campana 1999). As a fish grows, otoliths also develop via the deposition of concentric layers, sometimes referred to as “rings”, of calcium carbonate on their external surfaces (Campana 1999). In aragonite, the main form of calcium carbonate that makes up otoliths, trace elements such as Sr and Ba substitute for Ca in the calcium carbonate structure (Campana 1999). Therefore, as a fish grows, surface water environmental signatures (i.e. trace elements and isotopes) are incorporated into the otolith, effectively establishing a temporally continuous record of environmental signatures experienced by the fish. Analysis of the otolith microchemistry can, therefore, be used to retrace spatial movement patterns based on differences in the dissolved minerals incorporated into the calcium carbonate matrix (Reis-Santos et al. 2023). In riverscapes, where surface water environmental signatures are spatially heterogeneous, reconstructing a fish’s environmental history can then be used to reconstruct their movement history (Brennan & Schindler 2017). The spatial resolution of a fish’s movement history is tightly linked to the spatial heterogeneity present in surface water environmental signatures.
Elemental composition and concentration in otoliths are determined by several factors including environmental availability, concentration and ambient water temperature (Kennedy et al. 1997; Elsdon & Gillanders 2002). Barium, bromine, iron, potassium, magnesium, manganese, lead and strontium all provide useful indicators of fish movement and are typically reported directly, or as ratios, among elements such as calcium, barium or strontium (Campana 1999). Strontium and barium are the most common elements used to understand fish movement due to their relative stability across most environmental variables and physiological processes an individual may encounter; particularly for discriminating movements between freshwater and saltwater (Walther & Limburg 2012).
Understanding the geographical distribution of trace metals and isotopes throughout a region can determine origins and movement in both terrestrial and aquatic environments. The use of isotopes and isotope ratios, such as 87Sr:86Sr, have also been used to recreate the environmental histories of fish, most notably within varying freshwater environments (Reis-Santos et al. 2023). This is largely because isotopic ratios directly reflect the underlying geology of the water source (Ingram & Weber 1999; Kennedy et al. 2000). Strontium consists of several stable isotopes, and whilst 88Sr is the most abundant of the three stable Sr isotopes, the 87Sr:86Sr ratio is most often used in geological and environmental studies due to the isotope ratio fluctuating over spatial scales due to the decay of rubidium (Rb87) to 87Sr (Dickin 1995). As 87Rb has a half-life of almost 50 billion years, it is relatively unaffected by temperature or salinity, therefore making 87Sr:86Sr temporally stable (Kennedy et al. 2000; Faure & Mensing 2005). Variations in 87Sr concentrations due to differences in the underlying geology at a river’s source or along specific tributaries enable the use of Sr isotopes to interpret fish movement patterns in most instances (Holt et al. 2021). As a result, distinct isotope patterns observed in the fish otolith can provide insight into not only a fish’s movement between freshwater and marine habitats, but also movement between different freshwater habitats and the duration and frequency of these movements. However, otolith isotope ratio analyses can only be interpreted after the analysis of the water chemistry, coupled with an understanding of the associated underlying geology, is undertaken. Interpreted in the context of the timing, frequency and spatial scale of movement, this provides essential information for understanding the ecology of fish (Reis-Santos et al. 2023).
Coastal rivers provide important movement corridors for diadromous fish (McDowall 1988). Understanding these movements is paramount in managing the environments they occupy, but for many species, there is little or no information. The Clarence River and its tributaries make up the largest free-flowing coastal river system in Australia, rising to a maximum altitude of 800 m and draining 22,400 km2, and have an estuarine zone that extends approximately 110 km. Various igneous and sedimentary surface rocks occur throughout the Basin, with small regions of metamorphic and mixed rock types also present (Haworth and Ollier 1992). Diversity in the underlying geology throughout the Clarence River Basin suggests that surface water chemistry analysis using trace elements and strontium isotopes has high potential to illustrate the movement history of fish. To date, there has been no analysis of the distribution of trace elements and 87Sr:86Sr ratios in the Clarence River Basin, with the only research being limited elemental ratio analysis (Sr:Ca and Ba:Ca) (Miles et al. 2018) and modelling of 87Sr:86Sr ratios based on limited data (Bataille et al. 2020).
The primary objective of this study was to understand the spatial variation of six trace metals (barium, calcium, iron, manganese, magnesium and strontium) and strontium isotopes (87Sr:86Sr) throughout the Clarence River Basin. We measured trace metal and isotope ratios in water samples collected from various rivers throughout the Clarence River Basin to better understand the spatial composition and variation. The development of an isoscape for the Clarence River Basin represents a valuable tool to identify life-time movements in current and historic fish samples, including recreational and commercially important species such as Australian bass Percalates novemaculeata (Steindachner, 1866), freshwater mullet Trachystoma petardi (Castelnau, 1875) and sea mullet Mugil cephalus Linnaeus, 1758 as well as the locally abundant blue salmon catfish Neoarius graeffei (Kner & Steindachner, 1867). Quantifying the habitats in which these species exist and transition through is important for the design of effective species management and conservation plans. This becomes especially important when anthropogenic influences such as the regulation of natural flows via the construction of dams and are proposed. Here, we provide a basin-wide surface water chemistry assessment of the Clarence River and associated tributaries which can be used as a benchmark to elucidate fish movement using otoliths as well as providing baseline trace element information for future provenance studies.
Materials and methods
Water sample collection
Water samples were collected from 59 unique sites within the Clarence River Basin, New South Wales, throughout 2020 and 2021. Samples were collected within the main channel in both freshwater and estuary environments as well as from 23 of the systems main tributaries (Fig. 1). Due to a large flood in the Clarence River Basin in December 2020, sampling had to be completed over two separate periods: 9–14 December 2020 and 25 October–1 November 2021.
Water samples were collected at each site from an area with discernible current using acid-washed 600 mL plastic bottles attached to an extendable pole. Water quality parameters (temperature, pH, dissolved oxygen, conductivity and turbidity; see S2 in Supplementary materials) were also recorded at each site (Horiba U52, Horiba Corporation, Japan). All samples were kept cool and filtered within 48 h of collection. Samples were filtered using a 0.22 µm filter (Minisart, Sartorius Stedim) into new bottles containing 2 mL of Baseline Grade nitric acid (HNO3; CHOICE Analytical Pty Ltd,), resulting in a final volume of 100 mL. A “blank” was prepared using instrument grade water (18.20 MΩ.cm) instead of river water to account for possible field contamination. A subset of samples were stabilised with nitric acid prior to determine if filtering affected metal concentrations.
Trace metal analysis by ICP-OES
We analysed samples for a total of six elements (Ba, Ca, Fe, Mg, Mn and Sr) which are commonly used to reconstruct fish movement (Campana 1999). Four additional elements, Cu, K, Na and Se which are also common in reconstructing fish movement, were initially analysed but were unable to be measured due to low levels of these elements within the basin. We also analysed Fe spatial distribution throughout the Clarence River basin as despite its poor uptake in otoliths, initial inspection of the data revealed interesting patterns within the basin, and we endeavour for this baseline study to extend beyond fisheries ecology and include other disciplines that utilise provenance studies.
Samples were processed at the ICP-MS Research Facility in the Research School of Earth Sciences (RSES), Australia National University (ANU), Canberra. Water samples were analysed using Inductively Coupled Plasma Optical Emission Spectroscopy (ICP-OES; 5110 SVDV ICP-OES, Agilent Technologies, Santa Clara, CA, USA). Calibration was undertaken using 7-point calibration curve diluted from a 5% HNO3 matrix multi-element standard (IntelliQuant Multi-element Standard #1, Agilent Technologies) containing 48 elements and was performed over the range of 10 µg/L–100 mg/L. Independently prepared quality assurance analyses were undertaken at regular intervals during sample runs to detect if there were any changes to operating conditions. Samples were diluted 1 in 100 and 1 in 1000 using instrument grade water containing 2% (v/v) triple-distilled nitric acid as required.
87Sr:86Sr isotope analysis
Strontium isotopes were measured using multi-collector inductively coupled plasma mass spectrometry (MC-ICP-MS; Thermo Scientific, High Resolution NEPTUNE, Bremen, Germany). Purification of strontium for 87Sr:86Sr ratio analyses was completed using custom columns packed with Eichrom Sr-spec resin (~ 300 µL column with ~ 1 mL reservoir). Samples were acidified to 2 M with triple-distilled HNO3, and all solutions (samples and elution acids) were loaded on to the column in 0.3 mL aliquots to prevent disturbing the resin bed and to keep the resin wetted throughout chromatography. HNO3 (3 mL 2 M) was passed through the column to elute rubidium to waste. Barium and the bulk matrix were eluted with 4 mL 2 M, then 2 mL 7 M, then 0.3 mL 2 M HNO3 to waste. Finally, a clean 5.5 mL vial was used to collect strontium using 5.3 mL 0.02 M HNO3 for analysis directly by MC-ICP-MS. All elution acids were prepared using instrument grade water and triple-distilled HNO3. SRM987 (NIST; www.nist.gov) was used as a quality control for 87Sr:86Sr ratio analyses during extraction batches, with an absolute abundance ratio of 0.71034 ± 0.00026. The NIST SRM987 was also analysed during the sample analyses sessions and had a resulting average 87Sr:86Sr of 0.710289 ± 0.000067 (2 s.d., n = 59). Samples were run through columns in batches of 32, with each batch including a blank and SRM987 standard, whilst a subset of samples were selected at random to act as replicates. Additionally, samples were sample-standard bracketed using fresh SRM987 as the bracketing standard. Although isobaric interference was assumed to be nil due to the column chromatography method employed, 85Rb was corrected and internally normalized to the 88Sr:86Sr ratio of 8.375209.
Data analysis
Uptake of trace elements into bony structures within freshwater environments is often impacted by calcium concentrations of the aqueous environment, with trace element:Ca ratios being used to accurately represent the environmental availability of trace element concentrations (Campana 1999). As this study aims to use surface water microchemistry to build movement patterns in freshwater fish, trace element:Ca ratios were used for statistical analysis instead of direct trace element concentrations.
To identify sampling sites with similar water chemistry, a cluster analysis was undertaken using k-means clustering and the silhouette method (package "factoextra"; Kassambara & Mundt 2017). This method distinguishes how cohesive an observation is within its cluster comparative to other clusters. Despite its advantages, cluster analysis has its limitations which include difficulty interpreting ill-defined clusters and the success of the analysis relying on the choice of clustering algorithm (Fraley & Raftery 1998). Prior to clustering, all trace metal and strontium isotope ratios were scaled to account for variation in measures among trace metals and 87Sr:86Sr ratios. Trace metal ratios and 87Sr:86Sr concentrations were compared spatially between the groups using individual Kruskal–Wallis tests to determine if there was a significant difference between clusters. For any statistical differences, a post-hoc Dunn’s test was applied to determine which combinations were significantly different. All statistical analyses were conducted in R (R Core Team 2022).
Results
Analytical analysis of trace metal and 87Sr:86Sr isotope ratios
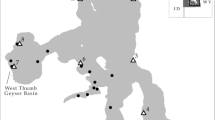
Cluster analysis revealed the optimal number of clusters was three, with the between sum of squares / total sum of squares value of 51.1% (Fig. 2a; Table 1). Most evident was cluster 2 which was located throughout the estuarine reaches of the basin (Fig. 2). The remaining sample sites were grouped into two clusters, with cluster 1 distributed throughout the east, south-eastern and at two sites in the northern edge of the basin. Cluster 3 was dominant throughout the interior of the basin.
a Spatial representation of the cluster analysis results using k-means producing three distinct clusters from the trace metal and 87Sr:86Sr ratio in the Clarence River Basin, and b Kmeans cluster plot highlighting three distinct clusters for trace metal and 87Sr:86Sr ratios recorded from throughout the Clarence River Basin
Mean trace element:Ca ratios (mmol/mol) highlighted the variation both between and within clusters in the Clarence River Basin. The Ba:Ca was highest in cluster 1 (3.0239 ± 1.6069), followed by cluster 3 (0.6747 ± 0.4696) and cluster 2 (0.2758 ± 0.3857; Fig. 3). Cluster 1 recorded very high Fe:Ca mean (188.6072 ± 101.6757) followed by cluster 3 (27.5032 ± 37.0630) and cluster 2 (0.9508 ± 2.6386; Fig. 3). The Mn:Ca varied between all three clusters with cluster 1 recording the highest mean (20.2320 ± 20.8836) followed by cluster 3 (4.6616 ± 6.6873) and cluster 2 (1.9903 ± 2.8371; Fig. 3). The Mn:Ca was highest in cluster 2 (5461.001 ± 1600.687), compared to cluster 1 (1139.743 ± 338.1844) and cluster 3 (1031.168 ± 466.3932; Fig. 3). Similarly, Sr:Ca values were also higher in cluster 2 (10.2709 ± 2.8440) followed by cluster 1 (5.0843 ± 1.8799) and cluster 3 (4.4601 ± 1.3261; Fig. 3). Throughout the Clarence River Basin, ratios of chemical tracers varied, but differences were almost entirely observed between cluster 2 and all other clusters (Table 1). Ba:Ca was significantly different between all clusters (H(2) = 35.81, p < 0.05), with pairwise comparisons indicating that cluster 1 was significantly different to cluster 2 (z = 5.686, p < 0.05) and cluster 3 (z = 4.604, p < 0.05), and clusters 2 and 3 were also different (z = − 2.217, p < 0.05; Table 2). Overall, Fe:Ca was significantly different (H(2) = 39.73, p < 0.05), with pairwise comparisons confirming that cluster 1 was significantly different from those of cluster 2 (z = 6.257, p < 0.05) and cluster 3 (z = 3.970, p < 0.05), and clusters 2 and 3 were also different (z = − 3.434, p < 0.05; Table 2). Mg:Ca was significantly different between clusters (H(2) = 29.64, p < 0.05), with pair wise comparisons confirming cluster 1 was significantly different compared to cluster 2 (z = − 3.859, p < 0.05) and clusters 2 significantly different from cluster 3 (z = 5.422, p < 0.05; Table 2). The comparison between clusters 1 and 3 was not significant for Mg:Ca (z = 1.201, p = 0.230; Table 2). Mn:Ca was significantly different between clusters (H(2) = 27.706, p < 0.05), with pairwise comparisons confirming that there are differences between clusters 1 and cluster 2 (z = 4.933, p < 0.05) and cluster 3 (z = 4.178, p < 0.05) as well as between clusters 2 and 3 (z = − 1.758, p < 0.05; Table 2). Sr:Ca was significantly different between clusters (H(2) = 27.260, p < 0.05), with cluster 1 significantly different compared to cluster 2 (z = − 3.618, p < 0.05): however there was no difference between cluster 1 and cluster 3 (z = 1.265, p = 0.206). The comparison between clusters 2 and 3 was also significant for Sr:Ca (z = 5.209, p < 0.05; Table 2).
87Sr:86Sr ratios varied throughout the Clarence River Basin and ranged from 0.70306 to 0.71506 (see supplementary materials S1). Both the lowest and highest values occurred in cluster 1, with the lowest 87Sr:86Sr ratio recorded in the Bielsdown River located in the south-eastern edge of the Basin, whilst the highest 87Sr:86Sr ratio recorded was in the Boonoo Boonoo River located at the north-western edge of the Basin. Cluster 2 had the highest mean 87Sr:86Sr values (0.70916 ± 0.00009), followed by cluster 1 (0.70839 ± 0.00304) and cluster 3 (0.70744 ± 0.00200; Fig. 3). Overall, 87Sr:86Sr was significantly different between clusters (H(2) = 9.756, p < 0.05), with pairwise comparisons indicating that only the comparison between clusters 2 and 3 was significant (z = 3.072, p < 0.05).
Spatial patterns of trace metal and 87Sr:86Sr ratios throughout the Clarence River Basin
Ba:Ca and Fe:Ca both followed a similar spatial pattern, with sites throughout the estuarine area exhibiting lower ratios compared to sites on the outer region of the Clarence River Basin particularly the northern, western and southern extents (Fig. 4). Conversely, Mg:Ca ratios were up to three times higher throughout estuarine areas compared to sample sites throughout the rest of the Basin (Fig. 4). Mn:Ca ratios were relatively low throughout the entirety of the Basin, with a few sites displaying elevated ratios in the outer region in the south-east and north (Fig. 4). Sr:Ca ratios varied geographically, with sites within the estuarine and eastern regions exhibiting elevated ratios compared to the western side of the basin. In general, 87Sr:86Sr ratios were lowest in the outer regions, particularly in the northern, southern and south-western regions, and increased throughout the interior, following the natural drainage structure of the Clarence River (Fig. 4). Despite this trend, a few sites located in the northern, western and south-eastern region of the Basin recorded higher 87Sr:86Sr ratios compared to sample sites around them. Sample sites located in the estuarine area displayed similar values to that of sea water (i.e. 0.70918 (Faure & Mensing 2005)).
Discussion
Trends in trace metals
The results of our study demonstrate that trace metal concentrations and 87Sr:86Sr ratio differences exist within the Clarence River Basin. Trace metal concentrations significantly differed throughout the Clarence River Basin, particularly between the estuarine, interior and outer edges of the Basin. As both magnesium and strontium are common ions found in sea water (Bruland 1983), the results are not unexpected, with -samples collected closer to the river mouth-anticipated-to have higher concentrations of these trace metals. Differences in trace metal and 87Sr:86Sr ratios were observed along the outer regions of the Clarence River Basin, particularly the northern, southern and south-western areas, compared to the inner basin and estuarine area. In addition to variation between clusters, intra-cluster variation was evident for both trace element ratios and 87Sr:86Sr ratios.
Concentration ratios of Ba:Ca and Fe:Ca had similar patterns throughout the Clarence River Basin, likely as a result-of similar geological parent sources. The Ba:Ca and Fe:Ca ratios were highest throughout the northern and southern edges of the basin, with ratios decreasing throughout the interior and towards the river mouth. This pattern suggests that elevated upstream concentrations of barium and iron were diluted as distance to the river mouth decreased, likely due to dilution by tributaries possessing lower barium and iron concentrations. Barium, a trace metal frequently used to understand fish movement patterns, is known to be more elevated in freshwater systems compared to sites influenced by marine water (Gaillardet et al. 2003; Walther & Limburg 2012).
As a trace metal, iron is present throughout both igneous and sedimentary rock (El Halim et al. 2022). Elevated iron concentrations in our study were evident around the southern, western and northern edges of the basin, particularly around areas of igneous rock. Although igneous basalts can be rich in iron, high concentrations of iron can also be related to the land use in these regions. The surface water iron concentrations in elevated headwater streams-throughout Finland were linked to increased concentrations of organic matter and rainfall (Sarkkola et al. 2013). Agricultural activities, particularly the grazing of livestock and the use of fertilizers, are common throughout the southern, western and northern edges of the Clarence River Basin (Butler et al. 2012) which may in part also explain the elevated iron concentrations in our water samples.
Concentration ratios of Mg:Ca displayed the inverse pattern of Fe:Ca and Ba:Ca, with elevated ratios recorded throughout the estuarine reaches (cluster 1), whilst those recorded throughout the interior and edges were lower. Elevated ratios of Mg:Ca throughout cluster 1 were strongly linked to changes in salinity, broadened due to the lengthy tidal limit which extends > 100 km upstream into the Clarence River, with both trace metals commonly present in sea water (Bruland 1983). Studies in the Mekong Delta have reported similar correlations between elevated Mg:Ca concentrations throughout estuarine areas highlighting the relationship between elevated Mg:Ca and salinity (Vu et al. 2021).
Conversely, Mn:Ca ratios exhibited a unique pattern, with relatively low concentrations recorded throughout much of the basin and only the odd elevated concentrations in the south-east, south-west and northern edges. Whilst manganese occurs naturally, elevated concentrations are often a result of human activity related to industrial discharge or agricultural activities. Elevated concentrations centred around these areas most likely relates to agricultural practices, with manganese sulphate (MnSO4) commonly used in fertilizers and livestock supplements (Howe et al. 2004). Similarly, Vu et al. (2021) found elevated concentrations of manganese at sites adjacent to large cities throughout the Mekong Basin and related these to effluent and organic pollution.
Barium and strontium concentrations are commonly used to understand fish movement patterns (Walther & Limburg 2012). Sr:Ca ratios were elevated throughout the estuarine area (cluster 2) and at some sites in the northern and southern edges. Outside of the estuarine area, higher Sr:Ca and Mg:Ca ratios, is possibly linked to the underlying geology. The Clarence–Moreton Basin in the east, which is a continent-derived sedimentary rock of Late Triassic to Late Jurassic age, overlaps the older New England Block in the west which originated in the late Devonian–late Carboniferous and late Permian–Late Triassic (Adams et al. 2022). Furthermore, both the northern and southern edges of the Clarence River Basin contain remnants of geologically young igneous volcanic rocks, such as basalts which are rich in trace metals including magnesium, strontium and iron.
Spatial variation in both Sr:Ca and Ba:Ca concentration ratios in the Clarence River Basin shows promise for resolving the movement of individual fish between fresh and marine waters. Strontium and barium are commonly used trace elements for this purpose because of their stability across various environmental conditions and physiological processes (Walther & Limburg 2012). However, the use of some trace metal ratios to reconstruct fish movement is not always recommended due to the influence of physiological processes on incorporation of ions into otoliths (Sturrock et al. 2015). For example, incorporation of magnesium and manganese in otoliths is influenced by fish growth and metabolism (Limburg et al. 2018; Hüssy et al. 2020), whilst temperature can influence the uptake of magnesium, iron and manganese (Fowler et al. 1995). As such, the spatial variation in the specific trace element ratios, such as iron, magnesium and manganese, may have little utility in discerning fish movement throughout the Clarence River Basin.
Trends in 87Sr:86Sr ratios
This current study is the first to complete a basin wide baseline assessment of the spatial variation of 87Sr:86Sr in the Clarence River Basin. However, there has been a number of basin wide assessments of the baseline 87Sr:86Sr ratios using surface water undertaken in Australia (Raiber et al. 2009; Crook et al. 2017). The range of values recorded throughout the Clarence River Basin (0.70306 to 0.71506) is similar to those modelled by Bataille et al. (2020), who modelled globally 87Sr:86Sr isotopes (0.705 to 0.706) using existing data to predict areas lacking records. Studies from throughout the Great Artesian Basin located across much of Australia’s eastern landmass, have noted similar 87Sr:86Sr values ranging spatially from 0.7045–0.7118 (Collerson et al. 1988). The importance of understanding 87Sr:86Sr spatial variation extends beyond fisheries science, with 87Sr:86Sr isoscapes also useful across a multitude of other fields of research. For example, Adams et al. (2019) created an 87Sr:86Sr isoscape of Cape York, in northeast Australia using a combination of soil, plant, water and faunal materials to understand provenance of human remains.
Strontium isotope ratios (87Sr:86Sr) in the Clarence River Basin were significantly different between clusters 2 and 3. In general, 87Sr:86Sr ratios were lower in the head waters/edges of the basin, before increasing as the distance to the river mouth decreased. Similar to trace metal concentrations, spatial patterns in 87Sr:86Sr ratios in the Clarence River Basin are likely due to differences in underlying geology, which along with atmospheric deposition, is the most common source of strontium in surface water (Bain & Bacon 1994; Capo et al. 1998). The geology in the southern and south–west edges of the basin where low ratios of 87Sr:86Sr were recorded, is part of the New England Block and is made up of tertiary volcanic rocks from the Cenozoic (e.g. basalt and pyroclastics) whilst the remainder is encompassed by the Ebor volcano (Haworth & Ollier 1992; McQueen 2016). Similarly, some sites at the north-east edges of the Clarence River Basin, near the Queensland border also exhibited low 87Sr:86Sr ratios. The Clarence–Moreton Basin extends throughout much of the northern and north-east side of the Clarence River Basin. The region also contains patches of tertiary volcanic rocks from the Cenozoic as well as those eroded from the Tweed volcano (Haworth & Ollier 1992; McQueen 2016). These volcanic rocks are relatively young in geological time (e.g. 19–24 million years (McQueen 2016)) and rocks younger in age exhibit lower Rb content equating lower 87Sr:86Sr ratios (Kennedy et al. 2000).
The interior of the basin is mostly covered by the New England Orogen and some of the Clarence–Moreton Basin in the east and most sites across this region displayed similar 87Sr:86Sr ratios. The change from the Clarence Moreton Basin to the New England Block did not result in any major differences in 87Sr:86Sr ratios. Two sites within the interior (both located on the Clarence River) recorded 87Sr:86Sr ratios lower than the rest of the interior. 87Sr:86Sr ratios from the adjacent upstream and downstream sites were similar to the rest of the interior sites suggesting that the origin of these lower 87Sr:86Sr ratios is local. Neighbouring geology, including pockets of sedimentary rock, could be responsible for the lower ratios, as lower 87Sr:86Sr ratios are often seen in young areas dominated by weathered calcium carbonate rocks (Palmer & Edmond 1992). Similar to trace metal concentrations, 87Sr:86Sr ratios throughout the estuarine area (cluster 2) are most likely influenced by changes in salinity and values recorded from these sites were similar to the commonly accepted value of sea water (i.e. 0.70918; Faure & Mensing 2005; McArthur & Howarth 2005).
Two sample sites in the current study recorded elevated 87Sr:86Sr ratios compared to rest of the region in which they were located, which included a site in the Boonoo Boonoo River (cluster 1) and a site in the Sara River (cluster 1). The previous studies have demonstrated that strontium isotopes can be influenced by environmental factors such as shell-based sediments, transportation of sediment through erosion, airborne inputs and non-local strontium carried into aquatic systems (Holt et al. 2021). Sites located in both these areas are surrounded by land subject to various agricultural pursuits such as cattle farming and are also impacted by various other anthropogenic pollutants as well. The elevated 87Sr:86Sr ratios found in these areas could be the result of contamination from external strontium inputs.
Using water microchemistry to understand fish movement in coastal rivers
Trace metals from the Clarence River Basin showed coarse geographic variation, although this was mostly apparent between the estuarine, interior and edges, particularly in the south-eastern and south-western regions as well as at some sites in the north. The salinity of surface water can also influence which method is best suited for understanding fish movement, which is particularly relevant when understanding fish movement in coastal river systems. 87Sr:86Sr ratios can be used to distinguish fish movement in environments where salinity is lower than 5%, whereas strontium concentration (Sr:Ca) is more useful to understand fish movement in environments with salinities > 5% (Milton & Chenery 2003). Furthermore, pairing trace elements such as strontium and barium with calcium to create ratios provide normalisation to account for variations in river discharge that would otherwise exhibit large fluctuations. Our results demonstrate that recreating movement patterns of fish solely within freshwater using 87Sr:86Sr ratios is possible, although the subtle variation in these values exhibited within our study limits the applicability of the technique to primarily resolving movements over coarse spatial scales. The use of strontium isotopes to understand freshwater movement patterns is common in larger systems and/or those that have greater differences in underlying geology and subsequently greater variation within 87Sr:86Sr (Crook et al. 2017; Adams et al. 2019). Using water microchemistry in coastal river systems to understand fish movement will be most suited to species that exhibit large spatial shifts across the basin, specifically movements from freshwater into the estuary and out into the open ocean (i.e, diadromy).
While the application of otolith microchemistry has numerous benefits to understanding how fish utilise their environment, there are limitations when compared to other commonly used techniques. For example, Miles et al. (2018) examined the movements of freshwater mullet in two river basins in NSW using both otolith microchemistry and acoustic telemetry (Miles et al. 2018). Acoustic telemetry was able to detect several brief movements between saline and freshwater environments which were not detected using otolith microchemistry (Miles et al. 2018). As a result, individuals that make brief/rapid movements between different chemical environments may not incorporate trace metals into their otoliths at detectable levels thus limiting the utility of the technique.
Conclusions
Understanding the trends in trace metal concentrations in river systems is essential for the interpretation of fish migration patterns. The current study illustrates the geographic variation of 87Sr:86Sr and some trace metals in surface waters of the coastal Clarence River Basin. The results have the potential for improving our understanding of the life-history and movement ecology of a range of fish species in the Clarence River Basin. The use of microchemical analysis using otoliths is frequently used to understand fish movements over time and space using both trace metal concentrations (Casselman 1980; Secor et al. 1992; Tzeng et al. 1997; Jessop et al. 2002; Wells et al. 2003; Strohm et al. 2017; Vu et al. 2021) and 87Sr:86Sr ratios (Hobbs et al. 2010; Stewart et al. 2021). Our results demonstrate spatial variation in some trace metal concentrations such as Ba:Ca, Sr:Ca as well as 87Sr:86Sr ratios, which could potentially be used to elucidate fish movements throughout the Clarence River Basin. While both Ba:Ca and Sr:Ca can be used to determine diadromy, 87Sr:86Sr ratios increased from the outer edges, throughout the interior and into the estuary, further highlighting their potential for understanding movement patterns in species that transition between the outer edges and into the interior of the basin or to the estuary. Outside of helping to understand fish movement patterns, this current study also provides baseline data for future water chemistry, paleontological and food biosecurity research. Serving as a baseline study for further research, we believe that future studies could utilise this study as a proof of concept and further dive into the spatial analysis using a tiered approach (e.g. Ciepiela & Walters, 2018). Furthering the understanding of the temporal variation of trace metals (e.g. Fowler et al. 1995), particularly seasonally would be of interest as well as further examination of differences within the saline and freshwater sections of the Clarence River Basin. Nevertheless, this study serves a proof of concept to provide baseline information for researchers to utilise and delve deeper into understanding trace elements in surface water in the Clarence River Basin.
Data availability
The datasets generated during the current study are available online at https://github.com/lstoot/Clarence-River-Water-Chemistry.
References
Adams, S., R. Grün, D. McGahan, J. X. Zhao, Y. Feng, A. Nguyen, M. Willmes, M. Quaresimin, B. Lobsey & M. Collard, 2019. A strontium isoscape of north-east Australia for human provenance and repatriation. Geoarchaeology 34(3): 231–251.
Adams, C., H. Campbell, R. Korsch & W. Griffin, 2022. Detrital zircons in Triassic-Cretaceous sandstones, Clarence-Moreton Basin, eastern Australia: speculations upon Australia and Zealandia provenances. Australian Journal of Earth Sciences 69(7): 909–928.
Bain, D. C. & J. Bacon, 1994. Strontium isotopes as indicators of mineral weathering in catchments. Catena 22(3): 201–214.
Bataille, C. P., B. E. Crowley, M. J. Wooller & G. J. Bowen, 2020. Advances in global bioavailable strontium isoscapes. Palaeogeography, Palaeoclimatology, Palaeoecology 555: 109849.
Brennan, S. R. & D. E. Schindler, 2017. Linking otolith microchemistry and dendritic isoscapes to map heterogeneous production of fish across river basins. Ecological Applications. 27: 363–377.
Bruland, K. W., 1983. Trace Elements in Seawater. Chemical Oceanography 8: 157–200.
Butler, G., B. Mackay, D. Gilligan & T. Broderick, 2012. Relative condition of the freshwater fish community in the Clarence Basin: North Coast New South Wales Ecohealth Program. A report to the Northern Rivers Catchment Management Authority.
Campana, S. E., 1999. Chemistry and composition of fish otoliths: pathways, mechanisms and applications. Marine Ecology Progress Series 188: 263–297.
Campana, S. E., D. H. Secor & J. M. Dean, 1995. Recent developments in fish otolith research, University of South Carolina Press:
Campana, S. E. & S. R. Thorrold, 2001. Otoliths, increments, and elements: keys to a comprehensive understanding of fish populations? Canadian Journal of Fisheries and Aquatic Sciences 58(1): 30–38.
Capo, R. C., B. W. Stewart & O. A. Chadwick, 1998. Strontium isotopes as tracers of ecosystem processes: theory and methods. Geoderma 82(1–3): 197–225.
Casselman, J., Chemical analysis of the optically different zones in ell otolith. In: Proceedings of the 1980 North American Eel Conference, 1982. p ages74–82.
Ciepiela, L. & A. Walters, 2018. Quantifying 87Sr/86Sr temporal stability and spatial heterogeneity for use in tracking fish movement. Canadian Journal of Fisheries and Aquatic Sciences 76 https://doi.org/10.1139/cjfas-2018-0124.
Collerson, K. D., W. J. Ullman & T. Torgersen, 1988. Ground waters with unradiogenic 87Sr/86Sr ratios in the Great Artesian Basin. Australia. Geology 16(1): 59–63.
Cooke, S. J., J. N. Bergman, W. M. Twardek, M. L. Piczak, G. A. Casselberry, K. Lutek, L. S. Dahlmo, K. Birnie-Gauvin, L. P. Griffin, J. W. Brownscombe, G. D. Raby, E. M. Standen, A. Z. Horodysky, S. Johnsen, A. J. Danylchuk, N. B. Furey, A. J. Gallagher, E. J. I. Lédée, J. D. Midwood, L. F. G. Gutowsky, D. M. P. Jacoby, J. K. Matley & R. J. Lennox, 2022. The movement ecology of fishes. Journal of Fish Biology n/a(n/a) https://doi.org/10.1111/jfb.15153.
Crook, D. A., K. Lacksen, A. J. King, D. J. Buckle, S. J. Tickell, J. D. Woodhead, R. Maas, S. A. Townsend & M. M. Douglas, 2017. Temporal and spatial variation in strontium in a tropical river: implications for otolith chemistry analyses of fish migration. Canadian Journal of Fisheries and Aquatic Sciences 74(4): 533–545. https://doi.org/10.1139/cjfas-2016-0153.
Degens, E. T., W. G. Deuser & R. L. Haedrich, 1969. Molecular structure and composition of fish otoliths. Marine Biology 2(2): 105–113.
Dickin, A., 1995. Radiogenic isotope geology.
El Halim, M., L. Daoudi, A. El Alaoui El & Fels, 2022. How elemental composition influences the color of igneous and sedimentary rocks: Case of the High Atlas rocks of Morocco. Color Research & Application 47(2): 475–485.
Elsdon, T. S., B. K. Wells, S. E. Campana, B. M. Gillanders, C. M. Jones, K. E. Limburg, D. H. Secor, S. R. Thorrold & B. D. Walther, 2008. Otolith chemistry to describe movements and life-history parameters of fishes: hypotheses, assumptions, limitations and inferences Oceanography and marine biology. CRC Press, 303–336.
Elsdon, T. S. & B. M. Gillanders, 2002. Interactive effects of temperature and salinity on otolith chemistry: challenges for determining environmental histories of fish. Canadian Journal of Fisheries and Aquatic Sciences 59(11): 1796–1808. https://doi.org/10.1139/f02-154.
Faure, G. & T. M. Mensing, 2005. Principles and applications, John Wiley & Sons Inc:
Fowler, A. J., S. E. Campana, S. R. Thorrold & C. M. Jones, 1995. Experimental assessment of the effect of temperature and salinity on elemental composition of otoliths using laser ablation ICPMS. Canadian Journal of Fisheries and Aquatic Sciences 52(7): 1431–1441.
Fraley, C. & A. E. Raftery, 1998. How Many Clusters? Which Clustering Method? Answers via Model-Based Cluster Analysis, the Computer Journal 41(8): 578–588.
Gaillardet, J., J. Viers & B. Dupré, 2003. Trace elements in river waters. Treatise on Geochemistry 5: 605.
Gallahar, N. & M. Kingsford, 1996. Factors influencing Sr/Ca ratios in otoliths of Girella elevata: an experimental investigation. Journal of Fish Biology 48(2): 174–186.
Gillanders, B. M., 2005. Otolith chemistry to determine movements of diadromous and freshwater fish. Aquatic Living Resources 18(3): 291–300.
Haworth, R. & C. Ollier, 1992. Continental rifting and drainage reversal: the Clarence River of eastern Australia. Earth Surface Processes and Landforms 17(4): 387–397.
Hobbs, J. A., L. S. Lewis, N. Ikemiyagi, T. Sommer & R. D. Baxter, 2010. The use of otolith strontium isotopes (87Sr/86Sr) to identify nursery habitat for a threatened estuarine fish. Environmental Biology of Fishes 89(3): 557–569. https://doi.org/10.1007/s10641-010-9672-3.
Holt, E., J. A. Evans & R. Madgwick, 2021. Strontium (87Sr/86Sr) mapping: a critical review of methods and approaches. Earth-Science Reviews:103593.
Howe, P., H. Malcolm & S. Dobson, 2004. Manganese and its compounds: environmental aspects. World Health Organization.
Hüssy, K., K. E. Limburg, H. de Pontual, O. R. Thomas, P. K. Cook, Y. Heimbrand, M. Blass & A. M. Sturrock, 2020. Trace Element Patterns in Otoliths: The Role of Biomineralization. Reviews in Fisheries Science & Aquaculture 29(4): 445–477.
Ingram, B. L. & P. K. Weber, 1999. Salmon origin in California’s Sacramento-San Joaquin river system as determined by otolith strontium isotopic composition. Geology 27(9): 851–854.
Jessop, B., J.-C. Shiao, Y. Iizuka & W.-N. Tzeng, 2002. Migratory behaviour and habitat use by American eels Anguilla rostrata as revealed by otolith microchemistry. Marine Ecology Progress Series 233: 217–229.
Kahler, T. H., P. Roni & T. P. Quinn, 2001. Summer movement and growth of juvenile anadromous salmonids in small western Washington streams. Canadian Journal of Fisheries and Aquatic Sciences 58(10): 1947–1956.
Kalish, J., 1990. Use of otolith microchemistry to distinguish the progeny of sympatric anadromous and nonanadromous salmonids. Fish Bull US 88: 657–666.
Kassambara, A. & F. Mundt, 2017. Package ‘factoextra’. Extract and visualize the results of multivariate data analyses 76(2).
Kennedy, B. P., C. L. Folt, J. D. Blum & C. P. Chamberlain, 1997. Natural isotope markers in salmon. Nature 387(6635): 766–767.
Kennedy, B. P., J. D. Blum, C. L. Folt & K. H. Nislow, 2000. Using natural strontium isotopic signatures as fish markers: methodology and application. Canadian Journal of Fisheries and Aquatic Sciences 57(11): 2280–2292.
Limburg, K. E., M. J. Wuenschel, K. Hüssy, Y. Heimbrand & M. Samson, 2018. Making the otolith magnesium chemical calendar-clock tick: plausible mechanism and empirical evidence. Reviews in Fisheries Science & Aquaculture 26(4): 479–493.
Lucas, M. & E. Baras, 2001. Migration of Freshwater Fishes Blackwell Science Ltd. London.
McArthur, J. M. & R. J. Howarth, 2005. Strontium isotope stratigraphy, Cambridge University Press:
McDowall, R. M., 1988. Diadromy in fishes: migrations between freshwater and marine environments, Croom Helm:
McQueen, K., 2016. Landscape evolution of the Clarence River catchment: Weird rivers and wild ideas.
Miles, N. G., G. L. Butler, S. L. Diamond, D. P. Bishop, D. E. van der Meulen, I. Reinfelds & C. T. Walsh, 2018. Combining otolith chemistry and telemetry to assess diadromous migration in pinkeye mullet, Trachystoma petardi (Actinopterygii, Mugiliformes). Hydrobiologia 808(1): 265–281.
Milton, D. A. & S. R. Chenery, 2003. Movement patterns of the tropical shad hilsa (Tenualosa ilisha) inferred from transects of 87Sr/86Sr isotope ratios in their otoliths. Canadian Journal of Fisheries and Aquatic Sciences 60(11): 1376–1385.
Palmer, M. & J. Edmond, 1992. Controls over the strontium isotope composition of river water. Geochimica Et Cosmochimica Acta 56(5): 2099–2111.
R Core Team, 2022. R: A language and environment for statistical computing vol R Foundation for Statistical Computing.
Raiber, M., J. A. Webb & D. A. Bennetts, 2009. Strontium isotopes as tracers to delineate aquifer interactions and the influence of rainfall in the basalt plains of southeastern Australia. Journal of Hydrology 367(3–4): 188–199.
Reis-Santos, P., B. M. Gillanders, A. M. Sturrock, C. Izzo, D. S. Oxman, J. A. Lueders-Dumont, K. Hüssy, S. E. Tanner, T. Rogers, Z. A. Doubleday, A. H. Andrews, C. Trueman, D. Brophy, J. D. Thiem, L. J. Baumgartner, M. Willmes, M.-T. Chung, P. Charapata, R. C. Johnson, S. Trumble, Y. Heimbrand, K. E. Limburg & B. D. Walther, 2023. Reading the biomineralized book of life: expanding otolith biogeochemical research and applications for fisheries and ecosystem-based management. Reviews in Fish Biology and Fisheries 33(2): 411–449. https://doi.org/10.1007/s11160-022-09720-z.
Sarkkola, S., M. Nieminen, H. Koivusalo, A. Laurén, P. Kortelainen, T. Mattsson, M. Palviainen, S. Piirainen, M. Starr & L. Finér, 2013. Iron concentrations are increasing in surface waters from forested headwater catchments in eastern Finland. Science of the Total Environment 463–464: 683–689. https://doi.org/10.1016/j.scitotenv.2013.06.072.
Secor, D. H., J. M. Dean & E. H. Laban, 1992. Otolith removal and preparation for microstructural examination. Otolith Microstructure Examination and Analysis Canadian Special Publication of Fisheries and Aquatic Sciences 117: 19–57.
Stewart, K. P., T. E. McMahon, T. M. Koel & R. Humston, 2021. Use of otolith microchemistry to identify subbasin natal origin and use by invasive Lake Trout in Yellowstone Lake. Hydrobiologia 848(10): 2473–2481.
Strohm, D. D., P. Budy & T. A. Crowl, 2017. Matching watershed and otolith chemistry to establish natal origin of an endangered desert lake sucker. Transactions of the American Fisheries Society 146(4): 732–743.
Sturrock, A. M., E. Hunter, J. A. Milton, F. Eim, R. C. Johnson, C. P. Waring & C. N. Trueman, 2015. Quantifying physiological influences on otolith microchemistry. Methods in Ecology and Evolution 6(7): 806–816.
Tzeng, W.-N., K. Severin & H. Wickström, 1997. Use of otolith microchemistry to investigate the environmental history of European eel Anguilla anguilla. Marine Ecology Progress Series 149: 73–81.
Vu, A. V., L. J. Baumgartner, G. S. Doran, M. Mallen-Cooper, J. D. Thiem, J. A. Howitt, K. E. Limburg, B. M. Gillanders & I. G. Cowx, 2021. Variability in water chemistry in the Lower Mekong Basin: Considerations for fish life history reconstruction. Estuarine, Coastal and Shelf Science 255: 107355. https://doi.org/10.1016/j.ecss.2021.107355.
Walther, B. & K. Limburg, 2012. The use of otolith chemistry to characterize diadromous migrations. Journal of Fish Biology 81(2): 796–825.
Wells, B. K., B. E. Rieman, J. L. Clayton, D. L. Horan & C. M. Jones, 2003. Relationships between water, otolith, and scale chemistries of westslope cutthroat trout from the Coeur d’Alene River, Idaho: the potential application of hard-part chemistry to describe movements in freshwater. Transactions of the American Fisheries Society 132(3): 409–424.
Acknowledgements
We thank K. Doyle and J. Bretzel for assistance with water sample collection and J. Howitt for developing the water sampling methodology. M. Misztela from ANU provided guidance and assistance during water microchemistry analysis. We thank comments from two anonymous reviewers which helped strengthen this manuscript and B.Hilna for aiding with analysis.
Funding
Open Access funding enabled and organized by CAUL and its Member Institutions. The funding for this project was provided by DPI Fisheries NSW, Gulbali Institute and Charles Sturt University.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by L. Stoot. The first draft of the manuscript was written by L. Stoot, and all authors contributed to subsequent versions of the manuscript. All authors read and approved the final manuscript.
Additional information
Editorial Responsibility: Michael Power.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Stoot, L.J., Baumgartner, L.J., Butler, G.L. et al. Spatial variability in water strontium isotopes and trace metals from the Clarence River Basin, New South Wales, Australia. Hydrobiologia (2024). https://doi.org/10.1007/s10750-024-05590-7
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10750-024-05590-7