Abstract
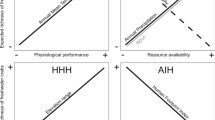
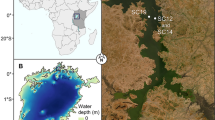
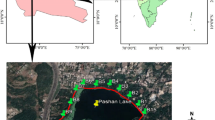
Invasion of non-native fishes threatens freshwater biodiversity worldwide. Yet, detailed estimates of population demography for invasive species, such as population size and body size, are rarely integrated in evaluating aquatic community responses. Our study capitalised on detailed brook trout population demographic data collected for a replicated whole lake ecosystem experiment involving experimental harvesting of exotic brook trout in nine mountain lakes. We applied environmental DNA (eDNA) metabarcoding and morphological taxonomy to examine the response of crustacean zooplankton and macroinvertebrate communities to gradients in brook trout effective density and lake elevation. Density-dependent effects of brook trout on crustacean zooplankton and macroinvertebrate communities were detected even decades after their first introductions (between 1926 & 1980). However, they were moderated by environmental factors such as elevation, lake maximum depth and dissolved organic carbon. Elevation was important in structuring crustacean zooplankton and macroinvertebrate community composition. While there were differences in explanatory variables when describing communities characterized by eDNA metabarcoding and morphological taxonomy, the principal environmental factors that structured the communities were similar. Our paper highlights persisting density-dependent impacts of exotic trout on invertebrate communities even decades after first introduction, and it considers the conservation implications for lake restoration.







Similar content being viewed by others
Data availability
The datasets generated during and/or analysed during the current study are available in the Environmental Data Initiative repository (Trépanier-Leroux et al., 2023): https://doi.org/10.6073/pasta/9fae641495fea570c60f0ad2777344b8
References
Albert, J. S., G. Destouni, S. M. Duke-Sylvester, A. E. Magurran, T. Oberdorff, R. E. Reis, K. O. Winemiller & W. J. Ripple, 2021. Scientists’ warning to humanity on the freshwater biodiversity crisis. Ambio 50: 85–94. https://doi.org/10.1007/s13280-020-01318-8.
Anderson, R. S., 1970. Effects of rotenone on zooplankton communities and a study of their recovery patterns in two mountain lakes in Alberta. Journal of the Fisheries Research Board of Canada. 27(8): 1335–1356. https://doi.org/10.1139/f70-159.
Anderson, R. S., 1974. Crustacean plankton communities of 340 lakes and ponds in and near the National Parks of the Canadian Rocky Mountains. Journal of the Fisheries Research Board Canada 31: 855–869. https://doi.org/10.1139/f74-105.
Antil, S., J. S. Abraham, S. Sripoorna, S. Maurya, J. Dagar, S. Makhija, P. Bhagat, R. Gupta, U. Sood, R. Lal & R. Toteja, 2023. DNA barcoding, an effective tool for species identification: a review. Molecular Biology Reports 50: 761–775. https://doi.org/10.1007/s11033-022-08015-7.
Banting, A. L., M. K. Taylor, R. D. Vinebrooke, C. M. Carli & M. S. Poesch, 2021. Assisted colonization of a regionally native predator impacts benthic invertebrates in fishless mountain lakes. Conservation Science and Practice 3(2): e344. https://doi.org/10.1111/csp2.344.
Barnes, M. A. & C. R. Turner, 2016. The ecology of environmental DNA and implications for conservation genetics. Conservation Genetics 17(1): 1–17. https://doi.org/10.1007/s10592-015-0775-4.
Barnes, M. A., C. R. Turner, C. L. Jerde, M. A. Renshaw, W. L. Chadderton & D. M. Lodge, 2014. Environmental conditions influence eDNA persistence in aquatic systems. Environmental Science & Technology 48(3): 1819–1827. https://doi.org/10.1021/es404734p.
Barton, K. 2022. MuMIn: Multi-Model Inference. R package version 1.46.0. https://CRAN.R-project.org/package=MuMIn
Beaulieu, J., D. Trépanier-Leroux, J. M. Fischer, M. H. Olson, S. Thibodeau, S. Humphries, D. J. Fraser & A. M. Derry, 2021. Rotenone for exotic trout eradication: nontarget impacts on aquatic communities in a mountain lake. Lake & Reservoir Management 37(3): 323–338. https://doi.org/10.1080/10402381.2021.1912864.
Beaulieu, J., Yates, M.C., Astorg, L., Trépanier-Leroux, D., Jeon, H., Rudko, S.P., Humphries, S., Fraser, D.J., Cristescu, M.E. & Derry, A.M. (in review). Environmental DNA for assessing impact and recovery of aquatic communities in an invaded mountain lake.
Bechara, J. A., D. Planas & S. Paquet, 2007. Indirect effects of brook trout (Salvelinus fontinalis) on the structure of epilithic algal communities in an oligotrophic boreal forest stream. Fundamental & Applied Limnology 169(2): 89–99. https://doi.org/10.1127/1863-9135/2007/0169-0089.
Benjamin, J. R., K. D. Fausch & C. V. Baxter, 2011. Species replacement by a non-native salmonid alters ecosystem function by reducing prey subsidies that support riparian spiders. Oecologia 167: 503–512. https://doi.org/10.1007/s00442-011-2000-6.
Bernery, C., C. Bellard, F. Courchamp, S. Brosse, R. E. Gozlan, I. Jarić, F. Teletchea & B. Leroy, 2022. Freshwater fish invasions: A comprehensive review. Annual Reviews in Ecology, Evolution, & Systematics 53(1): 427–456. https://doi.org/10.1146/annurev-ecolsys-03522-015551.
Brendonck, L. & L. De Meester, 2003. Egg banks in freshwater zooplankton: evolutionary and ecological archives in the sediment. Hydrobiologia 491: 65–84. https://doi.org/10.1023/A:1024454905119.
Brookes, B., H. B. Jeon, A. M. Derry, J. R. Post, S. M. Rogers, S. Humphries & D. J. Fraser, 2022. Neutral and adaptive drivers of genomic change in introduced brook trout (Salvelinus fontinalis) populations revealed by pooled sequencing. Ecology & Evolution 12(2): 1–14. https://doi.org/10.1002/ece3.8584.
Carlisle, D. & C. Hawkins, 1988. Relationships between invertebrate assemblage structure, two trout species, and habitat structure in Utah mountain lakes. Journal of North American Benthological Society 17(3): 286–300. https://doi.org/10.2307/1468332.
Carpenter, S. R., J. F. Kitchell & J. R. Hodgson, 1985. Cascading trophic interactions and lake productivity. BioScience 35(10): 634–639. https://doi.org/10.2307/1309989.
Collins, R. A., J. Bakker, O. S. Wangensteen, A. Z. Soto, L. Corrigan, D. W. Sims, M. J. Genner & S. Mariani, 2019. Non-specific amplification compromises environmental DNA metabarcoding with COI. Methods in Ecology & Evolution 10(11): 1985–2001. https://doi.org/10.1111/2041-210X.13276.
CoreTeam, R, 2021. R: A language and environment for statistical computing, R Foundation for Statistical Computing, Vienna:
Cristescu, M. E., 2014. From barcoding single individuals to metabarcoding biological communities: towards an integrative approach to the study of global biodiversity. Trends in Ecology & Evolution 29(10): 566–571. https://doi.org/10.1016/j.tree.2014.08.001.
Cristescu, M. E. & P. D. N. Hebert, 2018. Uses and misuses of environmental DNA in biodiversity science and conservation. Annual Reviews in Ecology, Evolution & Systematics 49(1): 209–230. https://doi.org/10.1146/annurev-ecolsys-110617-062306.
Deagle, B. E., S. N. Jarman, E. Coissac, F. Pompanon & P. Taberlet, 2014. DNA metabarcoding and the cytochrome c oxidase subunit I marker: not a perfect match. Biology Letters 10(9): 20140562. https://doi.org/10.1098/rsbl.2014.0562.
Deiner, K., H. M. Bik, E. Mächler, M. Seymour, A. Lacoursière-Roussel, F. Altermatt, S. Creer, I. Bista, D. M. de Lodge, N. Vere, M. Pfrender & L. Bernatchez, 2017. Environmental DNA metabarcoding: Transforming how we survey animal and plant communities. Molecular Ecology 26(21): 5872–5895. https://doi.org/10.1111/mec.14350.
Dejean, T., A. Valentini, A. Duparc, S. Pellier-Cuit, F. Pompanon, P. Taberlet & C. Miaud, 2011. Persistance of environmental DNA in freshwater ecosystems. PLoS ONE 6(8): e23398. https://doi.org/10.1371/journal.pone.0023398.
DeRoy, E. M., R. Scott, N. E. Hussey & H. J. MacIsaac, 2020. Density dependence mediates the ecological impact of an invasive fish. Diversity and Distributions 26(7): 867–880. https://doi.org/10.1111/ddi.13063.
Desforges, J. E., J. Clarke, E. J. Harmsen, A. M. Jardine, J. A. Robichaud, S. Serré, P. Chakrabarty, J. R. Bennett, D. E. L. Hanna, J. P. Smol, T. Rytwinski, J. J. Taylor, A. L. Martel, A. K. Winegardner, J. Marty, M. K. Taylor, C. O’Connor, S. A. Robinson, A. J. Reid, I. F. Creed, I. Gregory-Eaves, N. W. R. Lapointe & S. J. Cooke, 2022. The alarming state of freshwater biodiversity in Canada. Canadian Journal of Fisheries & Aquatic Sciences 79(2): 352–365. https://doi.org/10.1139/cjfas-2021-0073.
Donald, D. B., 1987. Assessment of the outcome of eight decades of trout stocking in the mountain national parks, Canada. North American Journal of Fish Management 7(4): 545–553. https://doi.org/10.1577/1548-8659(1987)7%3c545:aotooe%3e2.0.co;2.
Donald, D. B. & S. R. Anderson, 2003. Resistance of the prey-to-predator ratio to environmental gradients and to biomanipulations. Ecology 84(9): 2387–2394. https://doi.org/10.1890/02-3067.
Donald, D. B., R. S. Anderson & D. W. Mayhood, 1980. Correlations between brook trout growth and environmental, variables for mountain lakes in Alberta. Transactions of the American Fisheries Society 109(6): 603–610. https://doi.org/10.1577/1548-8659(1980)109%3c603:CBBTGA%3e2.0.CO;2.
Donald, D. B., R. D. Vinebrooke, R. S. Anderson, J. Syrgiannis & M. D. Graham, 2001. Recovery of zooplankton assemblages in mountain lakes from the effects of introduced sport fish. Candian Journal of Fisheries & Aquatic Sciences 58: 1822–1830. https://doi.org/10.1139/f01-121.
Eby, L. A., W. J. Roach, L. B. Crowder & J. A. Stanford, 2006. Effects of stocking-up freshwater food webs. Trends in Ecology and Evolution 21(10): 576–584. https://doi.org/10.1016/j.tree.2006.06.016.
Gilinski, E., 1984. The role of fish predation and spatial heterogeneity in determining benthic community structure. Ecology 65(2): 455–468. https://doi.org/10.2307/1941408.
Gleason, J. E., V. Elbrecht, T. W. A. Braukmann, R. H. Hanner & K. Cottenie, 2020. Assessment of stream macroinvertebrate communities with eDNA is not congruent with tissue-based metabarcoding. Molecular Ecology 30(13): 3239–3251. https://doi.org/10.1111/mec.15597.
Hall, D.L. 1991. Growth, fecundity, and recruitment responses of stunted brook trout populations to density reduction. Ph.D. dissertation, University of British Columbia. https://doi.org/10.14288/1.0100445
He, X., S. R. Gilmore, T. F. Sutherland, M. Hajibabaei, K. M. Miller, K. M. Westfall, J. Pawlowksi & C. L. Abbott, 2021. Biotic signals associated with benthic impacts of salmon farms from eDNA metabarcoding of sediments. Molecular Ecology 30(13): 3158–3174. https://doi.org/10.1111/mec.15814.
Hebert, P. D. N., A. Cywinska, S. L. Ball & J. R. DeWaard, 2003. Biological identifications through DNA barcodes. Proceedings of the Royal Society B 270(1512): 313–321. https://doi.org/10.1098/rspb.2002.2218.
Holmes, T., W. Tonn, C. A. Paszkowski & G. Scrimgeour, 2017. Limited effects of non-native trout on microcrustacean zooplankton in boreal foothills lakes: comparisons of stocked, unstocked, and fishless lakes. Canadian Journal of Fisheries & Aquatic Sciences 74(4): 598–608. https://doi.org/10.1139/cjfas-2015-0495.
Hume, J. & T. Northcote, 1985. Initial changes in use of space and food by experimentally segregated populations of Dolly-Varden (Salvelinus malma) and cutthroat trout (Salmo clarkii). Canadian Journal of Fisheries and Aquatic Sciences 42(1): 101–109. https://doi.org/10.1139/f85-013.
Knapp, R. A. & O. Sarnelle, 2008. Recovery after local extinction: factors affecting re-establishment of alpine lake zooplankton. Ecological Applications 18(8): 1850–1859. https://doi.org/10.1890/08-0044.1.
Knapp, R. A., P. S. Corn & D. E. Schindler, 2001a. The introduction of non-native fish into wilderness lakes: good intentions, conflicting mandates, and unintended consequences. Ecosystems 4(4): 275–278. https://doi.org/10.1007/s10021-001-0009-0.
Knapp, R. A., K. R. Matthews & O. Sarnelle, 2001b. Resistance and Resilience of Alpine Lake Fauna to fish introductions. Ecological Monographs 71(3): 401–421. https://doi.org/10.1890/0012-9615(2001)071[0401:RAROAL]2.0.CO;2.
Kratina, P., M. Vos, A. Bateman & B. R. Anholt, 2009. Functional responses modified by predator density. Oecologia 159(2): 425–433. https://doi.org/10.1007/s00442-008-1225-5.
Lamontagne, S., D. B. Donald & D. W. Schindler, 2008. The distribution of four Chaoborus species (Diptera: Chaoboridae) along an elevation gradient in Canadian Rocky Mountain lakes. Canadian Journal of Zoology 72(9): 1531–1537. https://doi.org/10.1139/z94-203.
Leese, F., M. Sander, D. Buchner, V. Elbrecht, P. Haase & V. M. A. Zizka, 2021. Improved freshwater macroinvertebrate detection from environmental DNA through minimized nontarget amplification. Environmental DNA 3(1): 261–276. https://doi.org/10.1002/edn3.177.
Legendre, P. & E. D. Gallagher, 2001. Ecologically meaningful transformations for ordination of species data. Oecologia 129: 271–280. https://doi.org/10.1007/s004420100716.
Leray, M., J. Y. Yang, C. P. Meyer, S. C. Mills, N. Agudelo, V. Ranwez, J. T. Boehm & R. Machida, 2013. A new versatile primer set targeting a short fragment of the mitochondrial COI region for metabarcoding metazoan diversity: application for characterizing coral reef fish gut contents. Frontiers in Zoology 10(1): 1–14. https://doi.org/10.1186/1742-9994-10-34.
Littlefair, J. E., A. Zander, C. de Sena Costa & E. L. Clare, 2019. DNA metabarcoding reveals changes in the contents of carnivorous plants along an elevation gradient. Molecular Ecology 28(2): 281–292. https://doi.org/10.1111/mec.14832.
Loewen, C. J. G. & R. D. Vinebrooke, 2016. Regional diversity reverses the negative impacts of an alien predator on local species-poor communities. Ecology 97(10): 2740–2749. https://doi.org/10.1002/ecy.1485.
Loewen, C. J. G., A. L. Strecker, G. L. Larson, A. Vogel, J. M. Fischer & R. D. Vinebrooke, 2019. Macroecological drivers of zooplankton communities across the mountains of western North America. Ecography 42(4): 791–803. https://doi.org/10.1111/ecog.03817.
Loewen, C. J. G., A. L. Strecker, B. Gilbert & D. A. Jackson, 2020. Climate warming moderates the impacts of introduced sportfish on multiple dimensions of prey biodiversity. Global Change Biology 26(9): 4937–4951. https://doi.org/10.1111/gcb.15225.
Ludgate, B.G., & G. Closs, 2003. Responses of fish communities to sustained removals of perch (Perca fluviatilis). New Zealand Department of Conservation, Wellington https://www.doc.govt.nz/globalassets/documents/science-and-technical/SFC210.pdf
Luikart, G., N. Ryman, D. A. Tallmon, M. K. Schwartz & F. W. Allendorf, 2010. Estimation of census and effective population sizes: The increasing usefulness of DNA-based approaches. Conservation Genetics 11(2): 355–373. https://doi.org/10.1007/s10592-010-0050-7.
MacLennan, M. M., C. Dings-Avery & R. D. Vinebrooke, 2015. Invasive trout increase the climatic sensitivity of zooplankton communities in naturally fishless lakes. Freshwater Biology 60(8): 1502–1513. https://doi.org/10.1111/fwb.12583.
Martin, G. K., B. E. Beisner, F. J. J. Chain, M. E. Cristescu, P. A. del Giorgio & A. M. Derry, 2021. Freshwater zooplankton metapopulations and metacommunities respond differently to environmental and spatial variation. Ecology 102(1): 1–11. https://doi.org/10.1002/ecy.3224.
Matte, J. M. O., D. M. Glaser, J. R. Post & D. J. Fraser, 2023. Contrasting demographic responses to size-selective harvesting among neighbouring wild fish populations. Journal of Applied Ecology 60(7): 1302–1313. https://doi.org/10.1111/1365-2664.14420.
Mazerolle, M. J. 2020. AICcodavg: Model selection and multimodel inference based on (Q)AIC(c). R package version 2. 3–1. https://cran.rproject.org/package=AICcmodavg
McGann, B. N. & A. L. Strecker, 2022. Zooplankton recovery from a whole-lake disturbance: examining roles of abiotic factors, biotic interactions, and traits. Ecosphere 13(4): 1–26. https://doi.org/10.1002/ecs2.3983.
McMurdie, P. J. & S. Holmes, 2013. Phyloseq: an R package for reproducible interactive analysis and graphics of microbiome census data. PLoS ONE. https://doi.org/10.1371/journal.pone.0061217.
McNaught, A. S., D. W. Schindler, B. R. Parker, A. J. Paul, R. S. Anderson, D. B. Donald & M. Agbeti, 1999. Restoration of the food web of an alpine lake following fish stocking. Limnology & Oceanography 44(1): 127–136. https://doi.org/10.4319/lo.1999.44.1.0127.
McNaught, A. S., D. Pavlik & D. W. Schindler, 2000. Patterns of zooplankton biodiversity in the mountain lakes of Banff National Park. Canada. SIL Proceedings 1922–2010 27(1): 494–499. https://doi.org/10.1080/03680770.1998.11901280.
Meredith, C., J. Hoffman, A. Trebitz, E. Pilgrim, S. Okum, J. Martinson & E. S. Cameron, 2021. Evaluating the performance of DNA metabarcoding for assessment of zooplankton communities in Western Lake Superior using multiple markers. Metabarcoding & Metagenomics 5: 83–97. https://doi.org/10.3897/mbmg.5.64735.
Messner, J. S., M. M. Maclennan & R. D. Vinebrooke, 2013. Higher temperatures enhance the effects of invasive sportfish on mountain zooplankton communities. Freshwater Biology 58(2): 354–364. https://doi.org/10.1111/fwb.12062.
Nelson-Chorney, H. T., C. S. Davis, M. S. Poesch, R. D. Vinebrooke, C. M. Carli & M. K. Taylor, 2019. Environmental DNA in lake sediment reveals biogeography of native genetic diversity. Frontiers in Ecology and the Environment 17(6): 313–318. https://doi.org/10.1002/fee.2073.
Okasen, J., Blanchet, F. G., Friendly, M., Kindgt, R., Legendre, P., McGlinn, D., Minchin, P. R., O’Hara, R. B., Simpson G. L., Solymos, P., Stevens, M. H. H., Szoecs, E., & H. Wagner. 2020. Vegan: Community Ecology Package. R package version 2.5–7. https://CRAN.R-project.org/package=vegan
Peters, R. H., 1983. The ecological implications of body size, Cambridge University Press, Cambridge:
Pinheiro J, Bates D, R Core Team (2022). nlme: Linear and Nonlinear Mixed Effects Models. R package version 3.1–157, https://CRAN.R-project.org/package=nlme.
Post, J. R., E. A. Parkinson & N. T. Johnston, 1999. Density-dependent processes in structured fish populations: interaction strengths in whole-lake experiments. Ecological Monographs 69(2): 155–175. https://doi.org/10.1890/0012-9615(1999)069[0155:DDPISF]2.0.CO;2.
Pukk, L., J. Kanefsky, A. L. Heathman, E. M. Weise, L. R. Nathan, S. J. Herbst, N. M. Sard, K. T. Scribner & J. D. Robinson, 2021. eDNA metabarcoding in lakes to quantify influences of landscape features and human activity on aquatic invasive species prevalenceand fish community diversity. Diversity & Distributions 27(10): 2016–2031. https://doi.org/10.1111/ddi.13370.
R Core Team (2021). R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. URL https://www.R-project.org/
Redmond, L. E., C. J. G. Loewen & R. D. Vinebrooke, 2018. A functional approach to zooplankton communities in mountain lakes stocked with non-native sportfish under a changing climate. Water Resources Research 54(3): 2362–2375. https://doi.org/10.1002/2017WR021956.
Reid, A. J., A. K. Carlson, I. F. Creed, E. J. Eliason, P. A. Gell, P. T. J. Johnson, K. A. Kidd, T. J. MacCormack, J. D. Olden, S. J. Ormerod, J. P. Smol, W. W. Taylor, K. Tockner, J. C. Vermaire, D. Dudgeon & S. J. Cooke, 2019. Emerging threats and persistent conservation challenges for freshwater biodiversity. Biological Reviews 94(3): 849–873. https://doi.org/10.1111/brv.12480.
Ruppert, K. M., R. J. Kline & M. S. Rahman, 2019. Past, present, and future perspectives of environmental DNA (eDNA) metabarcoding: a systematic review in methods, monitoring, and applications of global eDNA. Global Ecology and Conservation 17: e00547. https://doi.org/10.1016/j.gecco.2019.e00547.
Sakata, M. K., S. Yamamoto, R. O. Gotoh, M. Miya, H. Yamanaka & T. Minamoto, 2020. Sedimentary eDNA provides different information on timescale and fish species composition compared with aqueous eDNA. Environmental DNA 2(4): 505–518. https://doi.org/10.1002/edn3.75.
Sarnelle, O. & R. A. Knapp, 2004. Zooplankton recovery after fish removal: limitations of the egg bank. Limnology & Oceanography 49: 1382–1392. https://doi.org/10.4319/lo.2004.49.4_part_2.1382.
Schindler, D. W., 2000. Aquatic problems caused by human activities in Banff National Park, Alberta Canada. Ambio 29(7): 401–407. https://doi.org/10.1579/004-7447-29.8.401.
Schindler, D. W. & B. R. Parker, 2002. Biological pollutants: alien fishes in mountain lakes. Water Air & Soil Pollution 2(2): 379–397. https://doi.org/10.1023/A:1020187532485.
Schindler, D. W., R. A. Knapp & P. R. Leavitt, 2001. Alteration of nutrient cycles and algal production resulting from fish introductions into mountain lakes. Ecosystems 4: 308–321. https://doi.org/10.1007/s10021.
Schmidt-Nielsen, K. & S. N. Knut, 1984. Scaling: why is animal size so important?, Cambridge University Press:
Schnee, M. E., N. G. Clancy, M. C. Boyer & S. L. Bourret, 2021. Recovery of freshwater invertebrates in alpine lakes and streams following eradication of nonnative trout with rotenone. Journal of Fish & Wildlife Management 12(2): 475–484. https://doi.org/10.3996/JFWM-20-040.
Simon, K. S. & C. R. Townsend, 2003. Impacts of freshwater invaders at different levels of ecological organisation, with emphasis on salmonids and ecosystem consequences. Freshwater Biology 48(6): 982–994. https://doi.org/10.1046/j.1365-2427.2003.01069.x.
Strock, K. E., N. Theodore, W. G. Gawley, A. C. Ellsworth & J. E. Saros, 2017. Increasing dissolved organic carbon concentrations in northern boreal lakes: Implications for lake water transparency and thermal structure. Journal of Geophysical Research: Biogeosciences 122: 1022–1035. https://doi.org/10.1002/2017JG003767.
Symons, C. C. & J. B. Shurin, 2016. Climate constrains lake community and ecosystem responses to introduced predators. Proceedings of the Royal Society B 283(1832): 1–8. https://doi.org/10.1098/rspb.2016.0825.
Tiberti, R., A. von Hardenberg & G. Bogliani, 2014. Ecological impact of introduced fish in high altitude lakes: a case of study from the European Alps. Hydrobiologia 724(1): 1–19. https://doi.org/10.1007/s10750-013-1696-1.
Tiberti, R., M. Rolla, S. Brighenti & R. Iacobuzio, 2016. Changes in the insect emergence at the water–air interface in response to fish density manipulation in high altitude lakes. Hydrobiologia 779(1): 93–104. https://doi.org/10.1007/s10750-016-2801-z.
Tiberti, R., G. Bogliani, S. Brighenti, R. Iacobuzio, K. Liautaud, M. Rolla, A. von Hardenberg & B. Bassano, 2019. Recovery of high mountain Alpine lakes after the eradication of introduced brook trout Salvelinus fontinalis using non-chemical methods. Biological Invasions 21: 875–894. https://doi.org/10.1007/s10530-018-1867-0.
Tiberti, R., T. Buchaca, E. Cruset, L. Iacobelli, M. Maini, V. Osorio, M. Puig, Q. Pou-Rovira, I. Sabás & M. Ventura, 2022. Evaluation of visual encounter surveys as a method for the rapid assessment of fish presence and relative density in high mountain lakes. Aquatic Conservation: Freshwater and Marine Ecosystems 32(9): 1520–1528. https://doi.org/10.1002/aqc.3868.
Trépanier-Leroux, D., M. C. Yates, L. Astorg, D. J. Fraser, S. Humphries, M. E. Cristescu & A. M. Derry, 2023. Density-dependent effects of exotic brook trout on aquatic communities in mountain lakes revealed by environmental DNA and morphological taxonomy ver 2. Environmental Data Initiative. https://doi.org/10.6073/pasta/9fae641495fea570c60f0ad2777344b8.
Vanni, M. J., 2002. Nutrient cycling by animals in freshwater ecosystems. Annual Reviews in Ecology, Evolution & Systematics 33: 341–370. https://doi.org/10.1146/annurev.ecolsys.33.010802.150519.
Vinson, M. R., E. C. Dinger & D. K. Vinson, 2010. Piscicides and invertebrates: after 70 years, does anyone really know? Fisheries (bethesda) 35(2): 61–71. https://doi.org/10.1577/1548-8446-35.2.61.
Weidman, P. R., D. W. Schindler & R. D. Vinebrooke, 2011. Pelagic food web interactions among benthic invertebrates and trout in mountain lakes. Freshwater Biology 56(6): 1081–1094. https://doi.org/10.1111/j.1365-2427.2010.02552.x.
Weiss, S., Z. Z. Xu, S. Peddada, A. Amir, K. Bittinger, A. Gonzalez, C. Lozupone, J. R. Zaneveld, Y. Vázquez-Baeza, A. Birmingham & E. R. Hyde, 2017. Normalization and microbial differential abundance strategies depend upon data characteristics. Microbiome 5: 1–18. https://doi.org/10.1186/s40168-017-0237-y.
Xie, R., G. Zhao, J. Yang, Z. Wang, Y. Xu, X. Zhang & Z. Wang, 2021. eDNA metabarcoding revealed differential structures of aquatic communities in a dynamic freshwater ecosystem shaped by habitat heterogeneity. Environmental Research 201: 111602. https://doi.org/10.1016/j.envres.2021.111602.
Yates, M. C., D. M. Glaser, J. R. Post, M. E. Cristescu, D. J. Fraser & A. M. Derry, 2021. The relationship between eDNA particle concentration and organism abundance in nature is strengthened by allometric scaling. Molecular Ecology 30(13): 3068–3082. https://doi.org/10.1111/mec.15543.
Acknowledgements
We thank M. Lagacé, M. Salamon, S. Urschel, and A. Yaghi for field assistance. A. Zhukova contributed to enumeration of macroinvertebrate samples. We thank J. Littlefair and G. Martin for their laboratory assistance with eDNA metabarcoding. We thank D. Glaser, B. Brookes, J. Farkas, T. Ridgeon, N. Dupont, T. Bernos, L. Bogaard, H. Tunna, B. Kelley, M. Lagacé, and M. Tissier for conducting the mark-recapture experiments (Concordia University Animal Ethics permit #30007841). We thank anonymous reviewers whose detailed and constructive comments helped us to improve our manuscript.
Funding
This project was funded by a Fonds de Recherche du Québec—Nature et Technologie (FRQNT) team grant 254557 to AMD, MEC, and DJF, and by an NSERC Strategic Grant STPGP 494015–16 to AMD and DJF. DTL received a scholarship from the Quebec Fonds de Recherche du Québec—Recherche et Technologie (FRQNT) excellence center Groupe Interuniversitaire en Limnologie (GRIL), Grant No. GRIL-PCP-19E02.
Author information
Authors and Affiliations
Contributions
DTL, DJF, MEC, and AMD: conceptually designed the project. DJF: was responsible for leading the collection of brook trout population demography data. DTL: enumerated morphological community samples and conducted the eDNA laboratory research. MCY: conducted eDNA field sampling and calculated brook trout demography metrics. LA: conducted the metabarcoding sequence bioinformatics. DTL: conducted statistical analyses with the help of MCY, and she produced the figures. AMD: led the writing and all authors contributed to editing the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare there are no conflicts of interest. The authors have no relevant financial or non-financial interests to disclose. The authors have no competing interests to declare that are relevant to the content of this article. All authors certify that they have no affiliations with or involvement in any organization or entity with any financial interest or non-financial interest in the subject matter or materials discussed in this manuscript. The authors have no financial or proprietary interests in any material discussed in this article.
Additional information
Handling editor: Xavier Pochon.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Trépanier-Leroux, D., Yates, M.C., Astorg, L. et al. Density-dependent effects of exotic brook trout on aquatic communities in mountain lakes revealed by environmental DNA and morphological taxonomy. Hydrobiologia 851, 1489–1512 (2024). https://doi.org/10.1007/s10750-023-05398-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10750-023-05398-x