Abstract
Ecological succession within lakes generally proceeds as eutrophication and shallowing occur and the aquatic biota of the lakes is altered. The eutrophication rate depends on the lake size and the area and fertility of the watershed. If the watershed fertility of several different lakes is equivalent and the lakes’ formation time and initial environment are similar, the trophic status of each lake is expected to increase over time depending on the ratio of the watershed area to lake size. To test this supposition, we surveyed the topography and water quality of 10 lakes of varying sizes in Akan Caldera, Japan, which were formed thousands of years ago by fragmentation due to volcanic eruptions within the caldera. The ratio of the accumulated watershed area to lake size was positively correlated with total phosphorus concentration, an indicator of trophic status, and lake types were classified as oligotrophic, mesotrophic, eutrophic, and dystrophic. In addition, 21 species of macrophytes were found in the lakes, and the species composition of each lake was divided into five types corresponding to combinations of the lake types. The discovery of such diversity in a group of lakes with a similar origin paves the way for new comparative studies.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Ecological succession within lakes is not only a major component of freshwater ecology and limnology but also a platform for the diagnosis and management of aquatic environments, which are deteriorating worldwide due to human activity in the twentieth and twenty-first centuries (Whittaker, 1975; Sakamoto, 1976; Vollenweider & Kerekes, 1982; Owen, 1985; Horne & Goldman, 1994; Wetzel, 2001; Schindler & Vallentyne, 2008). In general, lake succession proceeds as follows: in a newly formed lake, the water is oligotrophic and contains a paucity of biota; subsequently, the trophic status increases owing to nutrient inflow from the watershed, and the lake basin shallows due to the accumulation of sediments; the diversity of biota and biomass increases or fluctuates throughout this process and eventually the lake attains the state of a bog or marsh (Lindeman, 1942; Whittaker, 1975; Sakamoto, 1976; Owen, 1985; Horne & Goldman, 1994). Forel, considered the father of limnology (Horne & Goldman, 1994), explained this transition using the analogy of human aging from adolescence to old age (Forel, 1901), and this concept has been widely accepted owing to both intuitive and experiential supporting evidence (Yoshimura, 1937; Whittaker, 1975; Sakamoto, 1976; Horne & Goldman, 1994). Forel also stated that it is impossible to follow a normal lake’s evolution because of the vast timescale over which a lake disappears due to sedimentation (Forel, 1901). Therefore, a general understanding of long-term succession has been indirectly formed by comparing many lakes under different trophic conditions (Dachnowski, 1912, 1926; Pearsall, 1920, 1921; Yoshimura, 1937; Kormondy, 1969; Odum, 1971), conducting follow-up surveys of relatively small dams or reservoirs (Sakamoto, 1976; Morishita, 1983), studying experimental microcosms and mesocosms (Margalef, 1968; Odum, 1969; Horne & Goldman, 1994; Wetzel & Likens, 2000; Dodson, 2005; Dodds & Whiles, 2010), and performing historical reconstructions of sedimentation processes (Smith et al., 1991; Wetzel, 2001). Nevertheless, the perception of ecological succession in large lakes still relies on many assumptions because of their long-aging process (Margalef, 1968; Sakamoto, 1976; Sayer et al., 1999; Johnson & Miyanishi, 2008). Thus, new approaches to studying lake succession, including the succession of large lakes, are required.
In principle, the rate of eutrophication, a major factor in lake succession, is determined by lake size, watershed area, and the fertility of the watershed (Horne & Goldman, 1994). Therefore, if the fertility is equivalent among different watersheds, eutrophication rates, i.e., the rates of nutrient increase, should vary depending on the ratio of the watershed area to lake size of each lake. In addition, in a group of lakes with a similar formation time and initial environment, the trophic statuses of the lakes are expected to change with positive correlations among the ratios of the watershed area to lake size. Such lakes can be considered a series that evolved to obtain different trophic statuses, providing a new approach to studying lake succession. However, in practice, this relationship has not been demonstrated because nutrient loading from the watershed is generally influenced by indigenous variables, such as land form, soil, erosion, local climate (temperature and rainfall), vegetation, and land use (farmland, factories, and urbanization) in addition to time (Yoshimura, 1937; Whittaker, 1975; Sakamoto, 1976; Owen, 1985; Horne & Goldman, 1994). Therefore, in the current study, we offer new insights into this area of research by comparing a group of lakes within a large caldera.
The lakes of Akan Caldera, Hokkaido, Japan were formed thousands of years ago by volcanic eruptions that divided a huge caldera lake into several lakes of various sizes (Satoh, 1965; Tamada & Nakagawa, 2009; Hasegawa & Nakagawa, 2016). Although these lakes share a similar terrestrial environment within the caldera, their trophic statuses are known to be diverse, ranging from oligotrophic to eutrophic (Tanaka, 1992; Maeda Ippoen Foundation, 1994; Hino et al., 1998). In the present study, we determined the topography and water quality of the Akan Caldera lakes to test the hypothesis that the trophic status is positively correlated with the ratio of the watershed area to lake size. Changes in aquatic vegetation are also an observable feature of lake succession (Yoshimura, 1937; Sakamoto, 1976; Wetzel, 2001). Indeed, previous studies on the Akan Caldera lakes suggested that aquatic macrophyte vegetation differs among the lakes (Kawakami, 1898; Tanakadate, 1925; Takayasu et al., 1930; Katagiri et al., 2001). Therefore, we also investigated the decision framework for vegetation by examining the relationship between the number and species composition of macrophytes and the topography and water quality of the lakes, including their trophic statuses.
Materials and methods
Study area
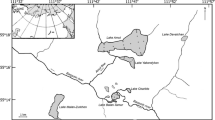
Akan Caldera is situated at the southern end of the Akan-Shiretoko Volcanic Chain, a volcanic region in eastern Hokkaido, Japan (Hasegawa & Nakagawa, 2016; Hasegawa et al., 2022). The outer shape of the caldera is oblong (24 km east–west × 13 km north–south) and a central cone, i.e., Mt. O-akan, rises at the center of its inner basin (Fig. 1). Within the caldera, lakes of various sizes surround Mt. O-akan and are interspersed with wetlands, grasslands, and forests that appear to have been formed after the deposition of smaller lakes (Satoh, 1965; Maeda Ippoen Foundation, 1994). Lake watersheds are isolated from external input by the caldera wall (Tanakadate, 1925; Takayasu et al., 1930), and the water systems, connected by rivers or underground flow (Takayasu et al., 1930; Takayasu & Kondo, 1936; Satoh, 1965), are roughly divided north–south and join at the southern foot of Mt. O-akan where they discharge through Akan River, i.e., the notch in the south caldera wall (Fig. 1a).
Map and landscape of the Akan Caldera lakes. a Watershed of Akan Caldera is isolated by a caldera wall shown by the red-dotted line; however, the western caldera wall was obscured by volcanic eruptions that occurred after the formation of the caldera. Ten lakes surround Mt. O-akan within the caldera and are connected by rivers and underground flow from water systems to the north and south. This map was generated using the map software Kashmir 3C (Sugimoto, 2010). b Land area is mostly covered with subarctic forests, except for a town south of Lake Akan (Akanko-onsen). L lake, P pond
Large lakes are distributed from west to northeast of Mt. O-akan, whereas smaller lakes are localized from south to east (Fig. 1a). This peculiar lake arrangement is a result of the formation history of the caldera and Mt. O-akan. In the Akan region, more than 10 large volcanic eruptions have occurred in the last 1.5 million years, and the present oblong-shaped caldera was formed by the largest eruption 200,000 years ago (Satoh, 1965; Hasegawa & Nakagawa, 2016). After the last large eruption (150,000 years ago), a huge lake, Ko-akanko (ancient Lake Akan), was generated in the caldera (Satoh, 1965; Tamada & Nakagawa, 2009; Hasegawa & Nakagawa, 2016). This lake was narrowed by postcaldera volcanic activity in the southwest part of the caldera; by 110,000 years ago, the landform of the inner caldera wall was almost complete (Satoh, 1965; Tamada & Nakagawa, 2009; Hasegawa & Nakagawa, 2016). A minor eruption of Mt. O-akan occurred 13,000 years ago slightly southeast of the center of the caldera and stopped when the lava flow reached the caldera wall; hence, Ko-akanko was separated into large and small basins (Satoh, 1965; Tamada & Nakagawa, 2009; Hasegawa & Nakagawa, 2016).
The developmental history of Mt. O-akan also suggests that Pond Hyotan and Pond Junsai of the southern water system were first divided from Ko-akanko, and the other lakes of the northern water system were formed 5000–2500 years ago (Satoh, 1965; Tamada & Nakagawa, 2009; Hasegawa & Nakagawa, 2016). The depth charts of the large lakes, namely Akan, Panke, and Penke, show the remains of valleys at the bottom of the inner caldera wall extending as far as the base of Mt. O-akan (Tanakadate, 1925; Takayasu & Kondo, 1936; Satoh, 1965), suggesting that the water level of Ko-akanko was extremely low or the basin was exposed by discharge through the notch before the eruption of Mt. O-akan (Satoh, 1965). Therefore, the lakes of the northern water system are thought to have been formed by reflooding after damming by Mt. O-akan, although the timing of lake formation and the developmental processes are not fully understood.
Akan Caldera occupies a part of Akan-Mashu National Park (designated in 1934) and is mostly covered with subarctic forests, except for a town on the south side of Lake Akan (Akanko-onsen) (Fig. 1b) (Maeda Ippoen Foundation, 1994). Only Lake Akan has been developed as a sightseeing area owing to the presence of Marimo, Aegagropila linnaei Kütz., a ball-shaped green alga designated as a Japanese natural treasure (Maeda Ippoen Foundation, 1994; Umekawa et al., 2021). Since the 1950s, the increase in tourism has resulted in eutrophication from sewage discharge, which continued until the 1980s when a public sewage treatment service was provided (Igarashi et al., 2000).
Topography of lakes and watersheds
Lake area, including the island areas, and boundary length were calculated using ARCGIS10 (Esri Japan Co.) based on the 1/25,000 numerical map data of the Geographical Survey Institute, Japan. Land watershed area was computed using the digital elevation model (DEM) 10-m data of the Geographical Survey Institute. After altitude data were changed into raster (altitude grid), subtle undulations were removed using the Fill tool, and the bearing azimuth of the flow was computed using the Flow Direction tool (north: 64; northeast: 128; east: 1; southeast: 2; south: 4; southwest: 8; west: 16; northwest: 32). Accumulation values (the number of cells accumulated toward the direction of flow) computed via the Flow Accumulation tool were extracted at accumulation values of > 30,000 (sl30000) and > 200 (sl200) using the Reclass command, and each watershed (ws30000 and ws200, respectively) was computed via grouping according to every feeder of sl30000 and sl200 using the Stream Link tool. Finally, the ws raster was converted into the polygon, and ws30000 and ws200 were manually divided as a watershed for each lake according to the DEM.
Lake volume and mean depth were calculated based on lake charts. Depth sounding for chart drawing was performed in the autumn of 2014 in eight lakes, excluding Lakes Akan and Panke for which the lake charts were already available. The depth of the entire lake was uniformly measured using a GPS fish finder (Lowrance HDS-8, Navico) on a motor boat, and the depth-sounding data were converted into contour drawings via the chart drawing software Reefmaster (ReefMaster Software Ltd.). The elevation of the lake surface was obtained using a global navigation satellite system survey. Residence time was calculated as lake volume divided by inflow, where inflow was equal to annual rainfall at 1200 mm (Maeda Ippoen Foundation, 1994) multiplied by the accumulated watershed area (see the “Statistical analysis” section).
Water quality
Measurements of physical and chemical variables and the collection of lake water were performed twice (in October–November 2013 and July 2014) at the surface of a deep area in each of the 10 studied lakes, and the data are shown as an average in the water quality section of Online Resource 1. Secchi depth (m), water temperature (Temp; °C), electrical conductivity (EC; mS m−1), and pH were directly recorded using a Secchi disk and portable sensors. Water was collected using 2-l polycarbonate bottles and taken immediately to the laboratory. Dissolved oxygen (DO; mg l−1) and chemical oxygen demand (COD; mg l−1) were measured via titration with a standard sodium thiosulfate solution and a potassium permanganate solution, respectively (K0102, 2013). Total nitrogen (TN; mg l−1) and total phosphorus (TP; mg l−1) were measured spectrophotometrically using an autoanalyzer (AACS-II, Bran + Luebbe Ltd.) (Grasshoff et al., 2009). Additionally, an aliquot of each water sample was filtered using Whatman GF/F glass fiber filters, and suspended solids (SS; mg l−1) were measured gravimetrically after drying at 110 °C for 2 h (K0102, 2013). Chlorophyll-a (Chl-a; μg l−1) concentrated onto a Whatman GF/F glass filter was quantified using a spectrophotometer (UV-1600, Shimadzu Co. Ltd.) after extraction using 100% methanol (SCOR-UNESCO, 1966). The filtrate and a TOC meter (TOC VC, Shimadzu Co. Ltd.) were used for measuring dissolved organic carbon (DOC; mg l−1) according to the high-temperature combustion method (Fukushima et al., 1996).
Macrophyte survey
Macrophytes were surveyed in the 10 lakes of Akan Caldera via SCUBA diving or snorkeling in October–November 2013 and July 2014. The survey area covered the entire littoral zone of each lake to the deepest point where vegetation was observed, and the maximum depth surveyed in this study was 18 m in Lake Panke. As Lake Jiro, Pond Kyodai-naka, and Pond Kyodai-minami are small and shallow and their lake bottoms in the limnetic zone were visible (Online Resource 1), their entire limnetic zones were included in the survey. Although aquatic plants, including emerged plants and macroalgae, were collected and recorded in the survey, the community analyses of the differences in growth and distribution factors were only performed for the submerged, floating-leaved, and free-floating plants.
Statistical analysis
In total, 31 variables were analyzed (Online Resource 1). Lake topographic factors included 11 variables: elevation, boundary length, lake area (LA), shore line development, maximum depth, mean depth, lake volume (LV), residence time, land watershed area (LWA), total watershed area (TWA, i.e., the sum of LA and LWA), and accumulated watershed area (AWA, i.e., the sum of the TWA above a given lake). LA and LWA correspond to the aquatic and terrestrial watershed areas specific to individual lakes, respectively, whereas AWA indicates the total watershed area including the upstream portion (Online Resource 2). Six variables reflected the inflow and outflow of materials: the LWA–LA ratio (LWA/LA), LWA–LV ratio (LWA/LV), TWA–LA ratio (TWA/LA), TWA–LV ratio (TWA/LV), AWA–LA ratio (AWA/LA), and AWA–LV ratio (AWA/LV) (Owen, 1985). Water chemistry parameters consisted of 10 variables: Temp, pH, DO, EC, SS, Chl-a, DOC, COD, TN, and TP. The macrophyte communities were defined by four variables: the number of submerged species, floating-leaved species, free-floating species, and total species. Secchi depth was excluded from the analysis because of missing data (Online Resource 1).
To explore the relationship among topography, water quality, and the macrophyte community, a correlation matrix containing these standardized data was constructed (Online Resource 2), and variables with strong correlation coefficients (|r| ≥ 0.7) were investigated in detail. In lakes where macrophytes occurred, the presence or absence of individual species was converted into binary data (1 or 0), and cluster analysis was conducted using the Ward method with the Euclidean distance. An add-in program for Microsoft Excel, BellCurbe (Social Survey Research Information) was used for these analyses.
Results and discussion
Water quality and lake types
The trophic statuses of the 10 lakes in Akan Caldera were diverse (Online Resource 1), i.e., the lakes were classified as oligotrophic (2 lakes), mesotrophic (5 lakes), and eutrophic (3 lakes) lakes (Fig. 2a) according to the Chl-a- and TP concentration-based criteria specified by the Organization for Economic Co-Operation and Development (Vollenweider & Kerekes, 1982). Among these lakes, Pond Junsai (eutrophic) had brownish water with high TN (0.438 mg l−1), DOC (7.1 mg l−1), and Chl-a (12.5 μg l−1) concentrations (Online Resource 1) indicative of dystrophic properties (Wetzel, 2001; Dodson, 2005). Thus, we consider Pond Junsai, a dystrophic lake.
Aspects of trophic status in the Akan Caldera lakes. a Lake types categorized by chlorophyll-a and total phosphorous (TP). The lakes are classified into the following types: oligotrophic, mesotrophic, and eutrophic, and Pond Junsai (red square) unites dystrophic characteristics (Online Resource 1). b Relationship between the ratio of the accumulated watershed area to lake volume (AWA/LV) and TP. Only Lake Akan (red square), which has been subjected to anthropogenic eutrophication in the past, shows an unusually high TP concentration. L lake, P pond
Among 10 water quality variables and 17 topographic characters, strong (|r| ≥ 0.7) and significant (P < 0.050 according to a test of no correlation) correlations were found between TP and AWA, TP and AWA/LV, EC and AWA, EC and AWA/LA, EC and AWA/LV, pH and shoreline development, DO and AWA/LV, and Temp and elevation (Table 1). Although the coefficients of the correlations between TP and AWA/LA and DO and AWA/LA did not reach 0.7 and − 0.7, respectively, both correlations were statistically significant (Table 1).
When two-dimensional plots of these 10 combinations were produced, Lake Akan was discriminated as an outlier in TP and AWA/LA, TP and AWA/LV, EC and AWA/LA, and EC and AWA/LV (Table 1, Fig. 2b). Lake Akan was subject to anthropogenic eutrophication in the second half of the twentieth century (Igarashi et al., 2000); thus, we estimated the phosphorus concentration of Lake Akan before eutrophication. The oldest P2O5 measurement taken in Lake Akan in 1931 (Miyadi, 1932), i.e., 0.010 mg l−1, gave a phosphorus concentration of 0.004 mg l−1 according to a conversion formula in which the P2O5 value was divided by 2.29 (Yoshimura, 1937). This calculated phosphorus concentration was close to the regression lines of TP and AWA/LA (r = 0.787, P < 0.050) and TP and AWA/LV (r = 0.881, P < 0.010) in the analysis of nine lakes, excluding Lake Akan (Fig. 3a). The difference (0.022 mg l−1) between the calculated phosphorus concentration and the observed concentration (0.026 mg L−1; Online Resource 1) was likely due to the increase caused by eutrophication, suggesting that the AWA/LA and AWA/LV of the lakes including Lake Akan likely had a tighter linear regression relationship with TP before eutrophication. Conversely, without including the outlier in the case of TP and AWA and replacing the Lake Akan TP data with the calculated value of 0.004 mg l−1, the resulting plot was distant from the regression line (r = 0.795, P < 0.050; Fig. 3b).
Estimation of the phosphorus concentration in Lake Akan in 1931 before eutrophication. Each regression line is drawn for nine lakes (circles), excluding Lake Akan. a In the relationship between the ratio of the accumulated watershed area (AWA) to lake volume and total phosphorus (TP) level, the 1931 estimate of the phosphorus concentration of Lake Akan (white square) is in proximity to the regression line. b Conversely, in the relationship between AWA and TP, the same 1931 estimate is distant from the regression line. L lake, P pond
A strong correlation was found between EC and TP (r = 0.821, P < 0.010; Online Resource 3). According to the regression formula (y = 819.023x − 2.343), the estimated EC of Lake Akan was 0.933 mS m−1, which was near the regression lines of EC and AWA/LA (r = 0.973, P < 0.001) and EC and AWA/LV (r = 0.979, P < 0.001) for nine lakes when TP was given as 0.004 mg l−1 (Fig. 4a). In contrast, the calculated EC of Lake Akan was distant from the regression line between EC and AWA without the outlier (r = 0.988, P < 0.001; Fig. 4b), suggesting that the involvement of AWA may be invalid, along with the results of the similar substitution in the correlation between TP and AWA (Fig. 3b).
Estimation of the electrical conductivity (EC) in Lake Akan in 1931 before eutrophication. Each regression line is drawn for nine lakes (circles), excluding Lake Akan. a In the relationship between the ratio of the accumulated watershed area (AWA) to lake volume and EC, the 1931 estimate of EC for Lake Akan (white square) is in proximity to the regression line, like in the case of phosphorus (Fig. 3). b Conversely, in the relationship between AWA and EC, the same 1931 estimate is distant from the regression line, like in the case of phosphorus (Fig. 3). L lake, P pond
The regression line of pH and shoreline development was also produced without including the outlier (Fig. 5). Because shoreline development is proportional to the length of the horizontal littoral zone, its correlation with pH might be related to the consumption of CO2 through hydrophytic photosynthesis (Sakamoto, 1976; Horne & Goldman, 1994; Brönmark & Hansson, 2005), although this assumption requires further investigation.
The regression lines of DO and AWA/LA and DO and AWA/LV exhibited a slight negative slope due to the low DO values of Lake Jiro (Fig. 6); thus, these regressions may not indicate accurate environmental gradients. Lake Jiro has no inflow and outflow rivers (Fig. 1a), and the water appears to be supplied through underground flow from the upstream Lake Akan (Motoda, 1950). However, in addition to DO, the Chl-a, DOC, and COD values of Lake Jiro were the lowest among the Akan Caldera lakes, whereas the TP concentration was the highest (Online Resource 1). Notably, a portion of Lake Jiro’s surface does not freeze in winter (Motoda, 1950). Collectively, these results suggest that other water sources, such as groundwater, may be involved in water formation in Lake Jiro.
Finally, regardless of lake size, water temperature decreased with elevation upstream, and the difference in water temperature was 3.5 °C for a change in elevation of about 150 m (Fig. 7).
Distribution and species composition of macrophytes
We recorded 21 species of macrophytes (excluding emergent plants and macroalgae) in 7 lakes, whereas no macrophyte species were observed in 3 lakes without inflow rivers (Online Resource 1). The correlation matrix of the number of macrophyte species was strong and significant with the following variables among the abovementioned lake topographic characters (Online Resource 3): boundary length (r = 0.720, P < 0.050), shoreline development (r = 0.703, P < 0.050), maximum depth (r = 0.924, P < 0.001), mean depth (r = 0.928, P < 0.001; Fig. 8), and residence time (r = 0.921, P < 0.001). Several previous studies on relatively shallow or small lakes and ponds found that the number of macrophyte species is correlated with lake area, and MacArthur and Wilson’s “theory of island biogeography,=,” which theorizes that larger islands are more biodiverse than smaller islands, was also applicable in these studies (Wilson & MacArthur, 1967; Møller & Rørdam, 1985; Rørslett, 1991; Weiher & Boylen, 1994; Toivonen & Huttunen, 1995; Linton & Goulder, 2000; Mäkelä et al., 2004; Søndergaard et al., 2005). However, in the present study, no significant correlation was found between lake area and the number of macrophyte species (Online Resource 3). Boundary length and shoreline development are parameters that affect the horizontal length of the littoral zone, and the magnitude of the maximum and mean depths and the residence time related to the depth and volume of the lake basin contribute to the expansion of the vertical littoral zone under conditions of higher water clarity. Therefore, the number of macrophyte species in the large lakes of Akan Caldera is presumed to be more closely related to the littoral zone area than the lake area.
Effect of lake size on the number of aquatic macrophyte species in the Akan Caldera lakes. In total, 21 species were observed in 7 lakes. Pond Junsai (red square) exhibits dystrophic water quality and is dominated by floating-leaved and free-floating plants, whereas most other lakes have a submerged plant community. L lake, P pond
Two-dimensional plots of the number of macrophyte species and the five abovementioned topographic characters revealed that Pond Junsai, a dystrophic lake, was an outlier (Fig. 8). Furthermore, when individual species were classified as submerged, floating-leaved, and free-floating, the correlation coefficient was significantly higher when the number of species of submerged plants was included in the correlation (Online Resource 3). The number of species of floating-leaved and free-floating plants was significantly and strongly correlated with the water quality parameters Chl-a, DOC, COD, and TN (Online Resource 3), and most of these species were localized in Pond Junsai. Thus, we conducted a cluster analysis to determine the species composition of macrophytes in each lake (Fig. 9). The cluster was first divided into two groups: 6 species of floating-leaved and free-floating plants in Pond Junsai and 14 submerged and 1 floating-leaved plant species in the other lakes. Pond Junsai contained the following indicator species of dystrophic water: Brasenia schreberi J. F Gmel. (Fig. 10), Nuphar pumila (Timm) DC. var. pumila, Nymphaea tetragona Georgi var. tetragona, and Utricularia macrorhiza Leconte (Kadono, 2014). The remaining species recorded from the other lakes were classified as oligotrophic, mesotrophic, oligo-mesotrophic, and oligo-meso-eutrophic according to the lake types based on trophic statuses (Fig. 2a). Ranunculus nipponicus Nakai var. submersus H. Hara (Fig. 10), Potamogeton alpinus Balb., and Isoetes asiatica (Makino) Makino, which are typically found in more pristine water (Lacoul & Freedman, 2006; Søndergaard et al., 2010; Kadono, 2014), were designated as oligotrophic. Myriophyllum spicatum L. (Fig. 10), Hydrilla verticillata (L. f.) Royle, Potamogeton compressus L., Potamogeton pectinatus L., and Ceratophyllum demersum L., which occur in more eutrophic settings (Egertson et al., 2004; Lacoul & Freedman, 2006; Kadono, 2014), were designated as oligo-mesotrophic. Potamogeton crispus L. (Fig. 10), which is distributed in a variety of water environments (Kadono, 2014), was designated as mesotrophic.
Cluster analysis of the distribution and species composition of aquatic macrophytes in the Akan Caldera lakes. The tree diagram was drawn using the Ward method with Euclidean distance based on the 1/0 data converted from the presence/absence data of the seven lakes in which macrophytes were found. According to the lake habitat types (Fig. 2a), clusters are classified into five vegetation types: oligotrophic, mesotrophic, oligo-mesotrophic, oligo-meso-eutrophic, and dystrophic. L lake, P pond
Determination model for macrophyte species composition corresponding to trophic types in the Akan Caldera lakes. Individual macrophyte species belong to any one of the five occurring macrophyte types, which are accompanied by inherent trophic type(s) and range. Macrophytes of oligotrophic-, mesotrophic-, and dystrophic occurrence types are only distributed within oligotrophic-, mesotrophic-, and dystrophic-type lakes, respectively. In contrast, within oligo-mesotrophic-, and oligo-meso-eutrophic occurrence types, macrophytes are able to occur widely in oligotrophic/mesotrophic- and oligotrophic/mesotrophic/eutrophic-type lakes. Accordingly, the species composition of macrophytes in oligotrophic- and mesotrophic-type lakes is determined by a combination of the occurrence types, i.e., oligotrophic/oligo-mesotrophic/oligo-meso-eutrophic and mesotrophic/oligo-mesotrophic/oligo-meso-eutrophic. Oligo-meso-eutrophic and dystrophic occurrence types are distributed in eutrophic and dystrophic lakes, respectively. L lake, P pond
Prior to anthropogenic eutrophication in the second half of the twentieth century, the phosphorus level of Lake Akan appears to have been much lower than the current level (Fig. 3). Thus, the current aquatic flora data of Lake Akan were compared with the results of the oldest known vegetation survey conducted in 1897 (Kawakami, 1898). In the present study, ten species of macrophytes were observed in Lake Akan (Fig. 9). However, two oligotrophic species, namely R. nipponicus var. submersus H. Hara and I. asiatica, which were recorded in 1897, were not found, whereas the presence of a mesotrophic species, P. crispus, in Lake Akan was discovered in this study. These results suggest a shift to a more eutrophic vegetation type occurred over time.
Uniqueness of the Akan Caldera lakes
To summarize, although the lakes of Akan Caldera have the same origin, after they were divided by volcanic eruptions, they developed into a series of oligotrophic, mesotrophic, eutrophic, and dystrophic lakes (Fig. 2a). The trophic status of each lake indicated by TP concentration was closely associated with the ratio of the watershed area (AWA) to lake size (LA and LV) (Fig. 2b). These results suggest that the rate of eutrophication differed among the lakes, and we observe the various stages of lake succession in progress in this system. However, the observed TP concentrations of Lake Akan and its downstream neighbors, Lake Jiro and Lake Taro, are assumed to have been affected by anthropogenic eutrophication in the past (Fig. 3a). Furthermore, the eruption history of Mt. O-akan suggests that Pond Hyotan and Pond Junsai may have formed earlier than the other lakes (Satoh, 1965; Tamada & Nakagawa, 2009; Hasegawa & Nakagawa, 2016). As shown in Fig. 2a, the Chl-a levels of these two ponds are higher than those of the other lakes, perhaps owing to differences in their formation times. To determine the long-term changes in nutrient loading, including TP, and primary production, the correct formation time and subsequent eutrophication history of each lake should be clarified through studies on, for example, lake sediment. In the present study, the linear regressions between TP and AWA/LA and AWA/LV (Fig. 2b) indicated that the indigenous environmental variables of the watersheds may vary only slightly. However, the actual geology and vegetation within the Akan Caldera are nonuniform (Satoh, 1965; Maeda Ippoen Foundation, 1994; Hasegawa et al., 2022); thus, further studies are needed to determine the actual phosphorus loading from the watersheds. Such studies will provide a clearer picture of the diversification of trophic statuses in the Akan Caldera lakes, including the abovementioned lake-specific events. Furthermore, comparing the succession process among the lakes might help determine whether the eutrophication rate in the large and oligotrophic and mesotrophic lakes Akan, Panke, and Penke is simply lower or mediated by unique processes, e.g., the phosphorus cycle, which varies with lake size, trophic status, and aquatic biota (Horne & Goldman, 1994; Wetzel, 2001).
Macrophyte species composition varied among the lakes according to their trophic conditions (Fig. 9). Many species of macrophytes can be classified into various types according to the environmental characteristics of habitats, including trophic status (Seddon, 1972; Wiegleb, 1984; Mäkirinta, 1989; Toivonen & Huttunen, 1995; Heegaard et al., 2001; Schneider & Melzer, 2003; Lacoul & Freedman, 2006). For example, Schneider and Melzer (2003) proposed seven categories ranging from oligotrophic to eutrophic, including polytrophic types. Lacoul and Freedman (2006) simplified this list into three categories, i.e., oligotrophic, eutrophic, and general types, based on the rationale that many macrophytes have a broad ecological range and occur with many trophic statuses, whereas other species have a narrower distribution. In the present study, all of the observed species belonged to one of five occurrence types, and trophic status exhibited a species-specific range (Fig. 10). Thirteen species were distributed among the oligotrophic, mesotrophic, and dystrophic lake types, whereas eight species occurred in the oligotrophic/mesotrophic and oligotrophic/mesotrophic/eutrophic lakes with wider trophic ranges. These results indicate how the compositions of macrophyte species in Akan Caldera lakes with different trophic types are determined by combinations of species with different trophic requirements. Notably, we found vegetation changes in Lake Akan caused by anthropogenic eutrophication. Such changes were characterized by the disappearance of oligotrophic-type species, the appearance of mesotrophic-type species, and the survival of oligo-mesotrophic- and oligo-meso-eutrophic-type species when the trophic condition of Lake Akan shifted from oligotrophic to mesotrophic, as shown in Fig. 10. Further investigation is necessary to determine whether this is a universal phenomenon in limnology.
Trophic status is not the only factor known to affect macrophyte distribution and species composition. At a smaller scale, the following physical factors can produce environmental gradients between or within lakes: topography, geological qualities, inflow waters as physical factors in the watershed, lake basin morphology (mainly depth and area), water temperature, light conditions, turbidity, current flow, and substrate (sediment). Additionally, the chemical factors that affect such gradients include inorganic ions, salinity, organic matter, conductivity, alkalinity, pH, and nutrients (Pearsall, 1920, 1921; Spence, 1967; Seddon, 1972; Hutchinson, 1975; Kadono, 1982; Wiegleb, 1984; Rørslett, 1991; Toivonen & Huttunen, 1995; Vestergaard & Sand-Jensen, 2000; Dodson, 2005; Søndergaard et al., 2005, 2010; Lacoul & Freedman, 2006; Lukács et al., 2015). Thus, elucidating the precise mechanisms underlying the distribution and species composition of macrophytes is challenging owing to multiple influencing factors. In the Akan Caldera lakes, species distribution was associated with trophic conditions (Figs. 9, 10), whereas the number of species was related to lake size (Fig. 8). In general, species diversity increases depending on spatial environmental heterogeneity (Wright et al., 1993; Miyashita & Noda, 2003). Lakes Akan, Panke, and Penke, which are all large and oligotrophic and mesotrophic lakes with high species counts, appear to provide a variety of the abovementioned physical and chemical factors. The topography of capes, bays, and islands in these lakes diversifies the wave action and substrates by varying wind–wave parameters and fetch, i.e., the length of the lake surface over which wind blows (Horne & Goldman, 1994; Lacoul & Freedman, 2006). Inflow rivers locally alter substrates, current, and water quality; deeper water lowers water temperature and reduces substrate grain size; and oligotrophic or mesotrophic water allows sunlight to penetrate deeper into the littoral zone, leading to a gradual gradient in the light environment (Hutchinson, 1975; Horne & Goldman, 1994; Weiher & Boylen, 1994; Toivonen & Huttunen, 1995; Wetzel, 2001; Dodson, 2005; Søndergaard et al., 2005, 2010; Lacoul & Freedman, 2006). Such environmental variability likely leads to the provision of habitats for many macrophytes in these large lakes. Conducting comparative studies with smaller lakes will help determine the effects of lake size on the development and change in macrophyte communities in the Akan Caldera lakes.
Pond Junsai was dominated by floating-leaved and free-floating plants and is the only example of a lake with dystrophic water quality in the Akan Caldera. Pond Junsai’s brownish water, derived from high DOC levels associated with an abundance of humic substances, causes substantial light absorption that suppresses submerged plant growth (Toivonen & Huttunen, 1995; Wetzel, 2001; Dodson, 2005; Lacoul & Freedman, 2006; Søndergaard et al., 2010). Humic substances originate from the decomposition of terrestrial and littoral plant materials (Wetzel, 2001; Brönmark & Hansson, 2005; Lacoul & Freedman, 2006; Dodds & Whiles, 2010). Comparative studies involving other lakes are also warranted to determine how and why Pond Junsai has evolved uniquely as well as the developmental processes of terrestrial and littoral vegetation and the history of humic substance loading.
Conclusion
The present study revealed that the Akan Caldera lakes are composed of four lake types with different trophic statuses (oligotrophic, mesotrophic, eutrophic, and dystrophic types) and five vegetation types corresponding to the lake types (oligotrophic, mesotrophic, oligo-mesotrophic, oligo-meso-eutrophic, and dystrophic types), despite having the same origin and being formed at almost the same time. The ratio of the accumulated watershed area to lake size (area and volume) was positively correlated with TP concentration, an indicator of trophic status, confirming our earlier prediction. Moreover, these results suggest that the eutrophication rates unique to each lake were determined by the watershed and lake basin size, and these rates have led to a variety of lake ecosystems that differ in their water quality and aquatic vegetation. This case can be likened to a large-scale experiment leading to the current statuses of the lakes, which have evolved over thousands of years after the massive container of the caldera was divided into large and small basins and watersheds by volcanic eruptions; thus, the Akan Caldera region has exceptional potential in terms of research and education in lake ecology. The actual rate and process of eutrophication and changes in aquatic vegetation in individual lakes will be revealed by conducting further studies on watersheds and lake sediments. This approach is expected to facilitate comparative studies on the spatiotemporal fluctuations in aquatic environments and biodiversity in large and small lakes.
Data availability
The datasets generated during and/or analyzed during the current study are available in the Zenodo repository, https://zenodo.org/record/7282382#.Y2hZYHbP1aQ.
References
Brönmark, C. & L.-A. Hansson, 2005. The Biology of Lakes and Ponds, 2nd ed. Oxford University Press, Oxford: 285.
Dachnowski, A., 1912. Peat Deposits of Ohio: Their Origin, Formation and Uses, Division of Geological Survey, Columbus:, 424.
Dachnowski, A. P., 1926. Factors and Problems in the Selection of Peat Lands for Different Uses, United States Department of Agriculture, New York:, 24.
Dodds, W. K. & M. R. Whiles, 2010. Freshwater Ecology, 2nd ed. Academic Press, Cambridge: 811.
Dodson, S. I., 2005. Introduction to Limnology, McGraw-Hill, New York:, 400.
Egertson, C. J., J. A. Kopaska & J. A. Downing, 2004. A century of change in macrophyte abundance and composition in response to agricultural eutrophication. Hydrobiologia 524: 145–156. https://doi.org/10.1023/B:HYDR.0000036129.40386.ce.
Forel, F. A., 1901. Handbuch der Seenkunde: Allgemeine Limnologie, J. Engelhorn, Berlin:, 249.
Fukushima, T., A. Imai, K. Matsushige, M. Aizaki & A. Otsuki, 1996. Freshwater DOC measurements by high-temperature combustion: comparison of differential (DTC-DIC) and DIC purging methods. Water Research 30: 2717–2722. https://doi.org/10.1016/S0043-1354(96)00198-4.
Grasshoff, K., K. Kremling & M. Ehrhardt, 2009. Methods of Seawater Analysis, 3rd ed. Wiley, Berlin: 632.
Hasegawa, T. & M. Nakagawa, 2016. Large scale explosive eruptions of Akan volcano, eastern Hokkaido, Japan: a geological and petrological case study for establishing tephro-stratigraphy and -chronology around a caldera cluster. Quaternary International 397: 39–51. https://doi.org/10.1016/j.quaint.2015.07.05.
Hasegawa, T., M. Nakagawa, H. Kamiyama & A. Yamamoto, 2022. Geological, geophysical, and geochemical constraints on the time-space evolution of Akan composite caldera, Hokkaido, Japan. Frontiers in Earth Science 10: 953152. https://doi.org/10.3389/feart.2022.953152.
Heegaard, E., H. H. Birks, C. E. Gibson, S. J. Smith & S. Wolfe-Murphy, 2001. Species-environmental relationships of aquatic macrophytes in Northern Ireland. Aquatic Botany 70: 175–223. https://doi.org/10.1016/S0304-3770(01)00161-9.
Hino, S., H. Mikami, J. Arisue, Y. Ishikawa, K. Imada, R. Yasutomi, Y. Kawamura & K. Takano, 1998. Limnological characteristics and vertical distribution of phytoplankton in oligotrophic lake Akan-Panke. Japanese Journal of Limnology 59: 263–279. https://doi.org/10.3739/rikusui.59.263.
Horne, A. J. & C. R. Goldman, 1994. Limnology, 2nd ed. McGraw-Hill, New York: 576.
Hutchinson, G. E., 1975. A Treatise on Limnology, Volume III, Limnological Botany, Wiley, New York:, 660.
Igarashi, S., Y. Ishikawa & H. Mikami, 2000. Long-term changes in limnological characteristics in Lake Akan, Hokkaido. Research Report from the National Institute for Environmental Studies, Japan 153: 34–54.
JIS K0102, 2013. Testing Methods for Industrial Wastewater, Japanese Standards Association, Tokyo:
Johnson, E. A. & K. Miyanishi, 2008. Testing the assumptions of chronosequences in succession. Ecology Letters 11: 419–431. https://doi.org/10.1111/j.1461-0248.2008.01173.x.
Kadono, Y., 1982. Occurrence of aquatic macrophytes in relation to pH, alkalinity, Ca++, Cl− and conductivity. Japanese Journal of Ecology 32: 39–44. https://doi.org/10.18960/seitai.32.1_39.
Kadono, Y., 2014. A Field Guide to Aquatic Plants of Japan, Bun-ichi Sogo Shuppan, Misato:, 326.
Katagiri, K., T. Ito & H. Kawamura, 2001. Water plants in the Lakes Akan and Akan-Panke, Hokkaido, Northern Japan. Scientific Reports of the Hokkaido Fish Hatchery 55: 41–48.
Kawakami, T., 1898. Collection record in Akan, Kushiro Province. Botanical Magazine 12: 220–225.
Kormondy, E. J., 1969. Concepts of Ecology, 2nd ed. Prentice-Hall, Upper Saddle River: 209.
Lacoul, P. & B. Freedman, 2006. Environmental influences on aquatic plants in freshwater ecosystems. Environmental Reviews 14: 89–136. https://doi.org/10.1139/a06-001.
Lindeman, R. L., 1942. The trophic dynamic aspect of ecology. Ecology 23: 399–417.
Linton, S. & R. Goulder, 2000. Botanical conservation value related to origin and management of ponds. Aquatic Conservation: Marine and Freshwater Ecosystems 10: 77–91. https://doi.org/10.1002/(SICI)1099-0755(200003/04)10:2%3c77::AID-AQC391%3e3.0.CO;2-Y.
Lukács, B. A., B. Tóthmérész, G. Borics, G. Várbíró, P. Juhász, B. Kiss, Z. Müller, G. László & T. Erős, 2015. Macrophyte diversity of lakes in the Pannon Ecoregion (Hungary). Limnologica 53: 74–83. https://doi.org/10.1016/j.limno.2015.06.002.
Maeda Ippoen Foundation, 1994. The Nature of Akan National Park, 1993, Maeda Ippoen Foundation, Tokyo:, 1289.
Mäkelä, S., E. Huitu & L. Arvola, 2004. Spatial patterns in aquatic vegetation composition and environmental covariates along chains of lakes in the Kokemäenjoki watershed (S. Finland). Aquatic Botany 80: 253–269. https://doi.org/10.1016/j.aquabot.2004.08.006.
Mäkirinta, U., 1989. Classification of South Swedish Isoetid vegetation with the help of numerical methods. Vegetatio 81: 145–157. https://doi.org/10.1007/978-94-009-2432-1_12.
Margalef, R., 1968. Perspectives in Ecological Theory, University of Chicago Press, Chicago:, 1119.
Miyadi, D., 1932. Studies on the bottom fauna of Japanese lakes. VII. Lakes of Hokkaido. Japanese Journal of Zoology 4: 223–252.
Miyashita, T. & T. Noda, 2003. Community Ecology, University of Tokyo Press, Tokyo:, 187.
Møller, T. R. & C. P. Rørdam, 1985. Species numbers of vascular plants in relation to area, isolation and age of ponds in Denmark. Oikos 45: 8–16. https://doi.org/10.2307/3565216.
Morishita, I., 1983. Dam Lake Ecology, Sankaidou, Tokyo:, 191.
Motoda, S., 1950. The lakes in Hokkaido. Scientific Reports of the Hokkaido Fish Hatchery 5: 1–96.
Odum, E. P., 1969. The strategy of ecosystem development. Science 164: 262–270.
Odum, E. P., 1971. Fundamentals of Ecology, 3rd ed. Saunders, New York: 574.
Owen, O. S., 1985. Natural Resource Conservation: An Ecological Approach, 4th ed. Macmillan, New York: 657.
Pearsall, W. H., 1920. The aquatic vegetation of the English lakes. Journal of Ecology 8: 163–201. https://doi.org/10.2307/2255612.
Pearsall, W. H., 1921. The development of vegetation in the English lakes, considered in relation to the general evolution of glacial lakes and rock basins. Proceedings of the Royal Society B 92: 259–284. https://doi.org/10.1098/rspb.1921.0024.
Rørslett, B., 1991. Principal determinants of aquatic macrophyte richness in northern European lakes. Aquatic Botany 39: 173–193. https://doi.org/10.1016/0304-3770(91)90031-Y.
Sakamoto, M., 1976. Ecological Succession, II, Kyoritsu-Shuppan, Bunkyo:, 234.
Satoh, H., 1965. Akanko, Explanatory Text of the Geological Map of Japan, Kushiro, No. 7. Geological Survey of Japan: 90.
Sayer, C., N. Roberts, J. Sadler, C. David & P. Wade, 1999. Biodiversity changes in a shallow lake ecosystem: a multi-proxy palaeolimnological analysis. Journal of Biogeography 26: 97–114. https://doi.org/10.1111/j.1365-2699.1999.00298.x.
Schindler, D. W. & J. R. Vallentyne, 2008. The Algal Bowl: Overfertilization of the World’s Freshwaters and Estuaries, University of Alberta Press, Alberta:, 323.
Schneider, S. & A. Melzer, 2003. The trophic index of macrophytes (TIM)—a new tool for indicating the trophic state of running waters. International Review of Hydrobiology 88: 49–67. https://doi.org/10.1002/iroh.200390005.
SCOR-UNESCO, 1966. Determination of Photosynthetic Pigments in Seawater. Monographs on Oceanographic Methodology 1, UNESCO Publications Center, New York:, 69.
Seddon, B., 1972. Aquatic macrophytes as limnological indicators. Freshwater Biology 2: 107–130. https://doi.org/10.1111/j.1365-2427.1972.tb00365.x.
Smith, J. P., P. G. Appleby, R. W. Battarbee, J. A. Dearing, R. Flower, E. Y. Haworth, F. Oldfield & P. E. O’Sullivan, 1991. Environmental History and Palaeolimnology, Springer, Tokyo:, 382.
Søndergaard, M., E. Jeppesen & J. P. Jensen, 2005. Pond or lake: does it make any difference? Archiv Für Hydrobiologie 162: 143–165. https://doi.org/10.1127/0003-9136/2005/0162-0143.
Søndergaard, M., L. S. Johansson, T. L. Lauridsen, T. B. Jørgensen, L. Liboriussen & E. Jeppesen, 2010. Submerged macrophytes as indicators of the ecological quality of lakes. Freshwater Biology 55: 893–908. https://doi.org/10.1111/j.1365-2427.2009.02331.x.
Spence, D. H. N., 1967. Factors controlling the distribution of freshwater macrophytes with particular reference to the lochs of Scotland. Journal of Ecology 55: 147–170. https://doi.org/10.2307/2257723.
Sugimoto, T., 2010. Introduction to Kashmir C3, Jitsugyo no Nihon Sha, Minato:, 191.
Takayasu, S. & K. Kondo, 1936. River and lake survey, Lake Panke and Lake Penke. Scientific Reports of Hokkaido Fisheries Experimental Station 39: 1–24.
Takayasu, S., H. Igarashi & K. Sawa, 1930. Lake survey, Lake Akan. Scientific Reports of Hokkaido Fisheries Experimental Station 21: 67–92.
Tamada, J. & M. Nakagawa, 2009. Eruption history of Oakan volcano, eastern Hokkaido, Japan. Kazan 54: 147–162.
Tanaka, M., 1992. The Lakes in Japan, Nagoya University Press, Nagoya:, 530.
Tanakadate, S., 1925. Research Report on the Volcano Lakes in Hokkaido, Hokkaido Government, Sapporo:, 155.
Toivonen, H. & P. Huttunen, 1995. Aquatic macrophytes and ecological gradients in 57 small lakes in southern Finland. Aquatic Botany 51: 197–221. https://doi.org/10.1016/0304-3770(95)00458-C.
Umekawa, T., I. Wakana & M. Ohara, 2021. Reproductive behavior and role in maintaining an aggregative form of the freshwater green alga Marimo, Aegagropila linnaei, in Lake Akan, Hokkaido, Japan. Aquatic Botany 168: 103309. https://doi.org/10.1016/j.aquabot.2020.103309.
Vestergaard, O. & K. Sand-Jensen, 2000. Alkalinity and trophic state regulate aquatic plant distribution in Danish lakes. Aquatic Botany 67: 85–107. https://doi.org/10.1016/S0304-3770(00)00086-3.
Vollenweider, R. & J. Kerekes, 1982. Eutrophication of Waters. Monitoring, Assessment and Control, Organization for Economic Co-Operation and Development, Paris:, 154.
Weiher, E. & C. W. Boylen, 1994. Patterns and prediction of α and β diversity of aquatic plants in Adirondack (New York) lakes. Canadian Journal of Botany 72: 1797–1804. https://doi.org/10.1139/b94-221.
Wetzel, R. G., 2001. Limnology, Lake and River Ecosystems, 3rd ed. Academic Press, Cambridge: 1006.
Wetzel, R. G. & G. E. Likens, 2000. Limnological Analyses, 3ed ed. Springer, New York: 429.
Whittaker, R. H., 1975. Communities and Ecosystems, 2nd ed. Macmillan, New York: 385.
Wiegleb, G., 1984. A study of habitat conditions of the macrophytic vegetation in selected river systems in Western Lower Saxony (Federal Republic of Germany). Aquatic Botany 18: 313–352. https://doi.org/10.1016/0304-3770(84)90055-X.
Wilson, E. O. & R. H. MacArthur, 1967. The Theory of Island Biogeography, Princeton, Princeton University Press:, 224.
Wright, D. H., D. J. Currie & B. A. Maurer, 1993. Energy supply and patterns of species richness on local and regional scales. In Ricklefs, R. E. & D. Schluter (eds), Species Diversity in Ecological Communities: Historical and Geographical Perspectives, Vol. 7. University of Chicago Press, Chicago: 66–74.
Yoshimura, S., 1937. Limnology, Sanseido, Chiyoda: 439.
Acknowledgements
“Research of the aquatic plants in Lake Akan and circumference lakes” and “Lake charts drawing of Lake Akan and circumference lakes,” conducted in 2013 and 2014 by the Ministry of Environment of Japan, form part of this study. The City of Kushiro supported the production of this study. Árni Einarrson (Mývatn Research Station, Iceland) provided advice on research design. Takehiko Fukushima (University of Tsukuba) and Koichi Kamiya (Ibaraki Kasumigaura Environmental Science Center) provided support with water analyses. The Hokkaido Shimbun Press contributed to aerial photography. Satoshi Kofuku and Kokoro Kikuchi (IDEA Consultants, Inc.) assisted with literature research. Shuji Hino (Yamagata University) and Yasushi Ishikawa (Hokkaido Research Organization) provided advice regarding the Caldera lakes and water quality determination. Mami Yamazaki (Sapporo Museum Activity Center) provided advice on macrophytes.
Funding
The authors did not receive support from any organization for the submitted work.
Author information
Authors and Affiliations
Contributions
IW, YK, and JU drafted the manuscript. IW designed the study. IW, YT, YS, HY, and YO obtained the data. KW and TH surveyed the formation process of the lakes. IW analyzed the data. MO supervised and administrated the project. All authors discussed the results, contributed critically to the drafts, and gave the final approval for publication.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no competing interests to declare that are relevant to the content of this article.
Additional information
Handling editor: Andre Andrian Padial
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
10750_2023_5231_MOESM1_ESM.xlsx
Supplementary file1 (XLSX 19 KB) Akan Caldera lakes_1 (Online Resource 1): Dataset of topography, water quality, and the number of species of macrophytes in the Akan Caldera lakes.
10750_2023_5231_MOESM2_ESM.pdf
Supplementary file2 (PDF 363 KB) Akan Caldera lakes_2 (Online Resource 2): Diagram illustrating the classification of the watershed variables.
10750_2023_5231_MOESM3_ESM.xlsx
Supplementary file3 (XLSX 21 KB) Akan Caldera lakes_3 (Online Resource 3): Correlation matrix of topography, water quality, and the number of species of macrophytes in the Akan Caldera lakes.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Wakana, I., Kadono, Y., Urabe, J. et al. Varying stages of ecological succession in lakes subdivided by volcanic eruptions at Akan Caldera, Japan. Hydrobiologia 850, 3919–3933 (2023). https://doi.org/10.1007/s10750-023-05231-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10750-023-05231-5