Abstract
Groundwater contributions to streamflow significantly influence the structure and function of riverine ecosystems, particularly in glacierized catchments where there are marked differences in water sources and subsurface flow paths. Here, we investigated spatial and temporal variation in relationships between water sources, flow paths, physical and chemical processes, organic matter, microbial biofilms, and macroinvertebrates across groundwater-fed streams in the glacierized Toklat River catchment of Denali National Park, Alaska. Streams fed predominantly by seepage from the valley sides were perennial, whereas streams sustained by glacial meltwater seepage were ephemeral. Differences in environmental conditions between flow regimes appeared to influence spatial and temporal patterns of organic matter, linking to macroinvertebrate community dynamics. Macroinvertebrates in perennial streams were supported by fine particulate organic matter from subsurface flow paths during summer, transitioning to a combination of fine particulate matter and leaf litter in autumn. In comparison, macroinvertebrates inhabiting ephemeral streams, which only flowed during autumn, were supported by leaf litter. Some macroinvertebrate taxa were unaffected by turnover in organic matter, indicating potential plasticity in organic matter resource use. Findings highlight the importance of considering spatial and temporal variation in groundwater-fed streams, considering that projected hydrological changes under a changing climate may have significant implications for these systems.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Groundwater contributions to streamflow in glacierized catchments are vitally important and typically create biodiversity hotspots (Murray et al., 2004; Boulton & Hancock, 2006; Murray et al., 2006) by sustaining low variability in flow and water temperature regimes, and high water clarity, when compared to other water sources (Füreder et al., 2001; Malard et al., 2001; Barquin & Death, 2006). Nevertheless, groundwater-fed streams vary in their degree of flow permanence ranging from ephemeral to perennial (Wood et al., 2005; Brown et al., 2007a). In glacierized catchments, this variability in groundwater contributions is a significant driver of macroinvertebrate community composition, yet this topic has generally been overlooked.
Groundwater flow pathways in glacierized catchments typically lie within the most permeable deposits of the valley bottom, valley side, and in bedrock aquifers (Robinson et al., 2008). As a result, features such as river terraces, which are often composed of coarse fluvial deposits, may support groundwater upwellings in glacierized river catchments (Malard, 2003). Groundwater permeating these deposits can be derived from multiple sources, for example, glacial meltwater can infiltrate the underlying alluvial aquifer and then discharge to the surface further down the valley (Malard et al., 1999; Crossman et al., 2011). Alternatively, precipitation can percolate through colluvial and alluvial deposits and remerge at lower elevations when the water table intersects the surface (Crossman et al., 2011). Furthermore, groundwaters can be routed through deposits via different flow pathways (Anderson, 1989), resulting in multiple different types of streams originating on river terraces. The combined effects of water sources and flow paths lead to marked heterogeneity in instream conditions within river terrace streams (e.g., hydrology and physical and chemical processes), as well as influencing the riparian ecotones surrounding the stream channels (Stromberg & Merritt, 2016). This variation in streams and riparian habitats across river terraces is responsible for their role as hotspots of terrestrial and aquatic biodiversity in an otherwise fluvial landscape (Crossman et al., 2012).
Across streams, there may be physical and environmental differences resulting from variation in the relative contributions of groundwater to total stream flow (Kath et al., 2016), the interactive effects of distinct groundwater sources (e.g., snowmelt, surface seepage, and glacial melt), and associated water flow pathways (e.g., deep or shallow subsurface flows) (Khamis et al., 2016). Although physical and chemical variables are generally more stable in groundwater-fed streams than in other stream types, there can still be considerable variability in these variables in groundwater-fed streams in glacierized catchments (Robinson et al., 2008; Windsor et al., 2017; Durkota et al., 2019). Notably, habitat patches within streams can differ in their environmental conditions in response to their proximity to groundwater discharges (Mathers et al., 2017; Crespo-Pereez et al., 2020). Patches close to areas of groundwater exhibit different organic matter and macroinvertebrate community dynamics depending upon the source of groundwater. These characteristics generate variability across spatial and temporal scales in both physical and chemical habitat (flow permanence and environmental stability) and resource availability (nutrient and organic matter concentrations) within and across stream systems (Boulton & Hancock, 2006; Sertić Perić et al., 2021). In turn, this variation influences the biotic communities inhabiting these systems, including microbial biofilms (Colls et al., 2019) and macroinvertebrates (Windsor et al., 2017).
Climatic and associated hydrological change threatens the ecological integrity of groundwater-fed streams in glacierized catchments. For example, estimates in the Toklat catchment in Denali National Park (Alaska, USA) suggest that snow- and glacial meltwater may contribute to an increase in groundwater flow by 2070 (Crossman et al., 2013a). As such, changes in the contributions of groundwater from different water sources are likely to influence the environmental and biological conditions present. Here, we investigate the complex spatial and temporal dynamics of the relationship between macroinvertebrate communities and their physiochemical environment. In doing so, we highlight the role of potential climate change-induced stresses on freshwater communities within glacierized catchments. The specific objectives of the study were to assess:
-
(i)
The spatial and temporal variability in environmental and physical variables (e.g., water source contributions, flow regime, water temperature) in different groundwater-fed streams across the Toklat Catchment in Denali National Park, and;
-
(ii)
The relationships between environmental, physical, and chemical variables and benthic macroinvertebrate communities (overall structure and individual taxa)
Our hypothesis was that we would be able to distinguish between the physical and chemical variables and macroinvertebrate communities associated with different groundwater types. We accomplished these objectives during an intensive summer field campaign sampling perennial and ephemeral streams in a glacierized valley of the Toklat River in Alaska.
Methods
Site description
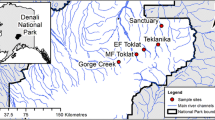
The Toklat River is a braided north-flowing, glacier-fed tributary of the Yukon River which derives a substantial percentage of its total flow from valley glaciers in its headwaters. The upstream catchment is ~ 115 km2 in area, ranging in elevation from 1,835 to 1,197 m. Mean annual precipitation is ~ 2,400 mm of which ~ 2,000 mm falls as snow, while mean annual June to September daily air temperature in 2008 (recorded 5 km to the North at the Toklat Road Camp) was 11.8 °C (day) and 4.7 °C (night) (WRCC, 2008). The underlying geology is dominated by Devonian and Silurian clastic and carbonate rocks, with Quaternary and Neogene sediments along the valley sides (Gilbert et al., 1984). Extensive debris fans and talus cones have formed along the valley margins with glacial and fluvial-glacial deposits in the valley bottom.
From May 30th to September 8th, 2008, we studied seven sites across four groundwater-fed streams arising across a fluvial terrace (0.9 km2 area) on the floodplain of the middle fork of the Toklat River in Denali National Park, Alaska (63°29′19.54″ N, 149°57′54.05″ W). These streams, in comparison to a barren active floodplain, support a dense marginal vegetative community including soapberry [Shepherdia canadensis (L.) Nutt.] and dwarf birch (Betula nana L.), alongside other riparian specialist plants. Sites were selected to represent different groundwater and riparian conditions (see Table 1). Upstream (A2, A5, B2) and downstream (A1, A4, B3) sites within the same streams were used to compare local variations in the levels of groundwater influence as determined by local flow paths, which were in turn related to the position of the site on the alluvial terrace (Fig. 1). The riparian vegetation associated with individual streams varied, depending on proximity to the active channel, and also the hydrological conditions in the surrounding terrace sediments.
To contextualize the following analyses, precise topographical measurements of the study area, including the position of the channels on the terrace and the elevation of individual sites, were mapped using a Geo electronic distance measurer (LEICA, Wetzlar, Germany).
Water source contributions
A total of 70 water samples were collected at 14-day intervals across all sites and additional samples were collected higher up the catchment to represent specific water sources (e.g., ice-melt, snowmelt, debris fan seepage flow and rainfall). Stream water samples were collected from the center of the stream using acid-rinsed Nalgene polyethylene bottles. Samples were filtered through 0.45 μm nylon membrane filters and refrigerated prior to analysis. In the laboratory, chloride (Cl−) concentrations (ppm) were measured using an ICS 2000 (Dionex, Sunnyvale, USA). Instrumental precision for the analysis of Cl− was 0.25 ppm.
Concentrations of Cl− in deep groundwater from the debris fan site (DFSdeep) were approximately 85 times greater than in samples of snow and glacial meltwater; therefore, we used Cl− measurements across stream samples and water sources to estimate the proportional contribution of DFSdeep from the debris fan to the total flow for each groundwater stream. Estimates were made using a groundwater mixing model, with the groundwater end-member estimated from high Cl− concentrations observed in waters from DFSdeep:
where DFSdeep% was the percentage contribution of water at each site that was derived from DFSdeep, Su was the concentration of Cl− at upwelling sites, and SD was the mean Cl− concentration of debris fan seepage (Crossman et al., 2011).
Flow pathways
Stream depth and hydraulic head were measured twice daily from June 17th to September 2nd, 2008, using nested piezometers inserted to depths of 50 cm and 100 cm at each sample site (Fig. 1). Within the piezometers, Level TROLL 500 pressure transducers (In-Situ Inc., Fort Collins, USA) were used to log water level and the direction of subsurface water movement was determined by calculating the hydraulic gradient at each piezometer nest (dividing the difference between stream water level and the water level in the piezometers at 50 cm depth by the vertical distance). A positive gradient indicated groundwater discharge. To provide additional context and confirm the contributions of different flow paths, pH and electrical conductivity (EC) were monitored monthly using a HI 98,127 multimeter (Hanna Instruments, Woonsocket, USA).
Physical and chemical variables
Stream morphology
Stream channel dimensions, water depth, and flow velocity were measured at 1/6 depth every 3 weeks over the study period, and stream discharge and flow duration curves were subsequently estimated. At each site, predominant substrate size was determined by measuring the b-axis of 100 randomly selected stones (Burgher et al., 2002) and D50 was calculated. The bottom component of the Pfankuch Stability Index was also determined, incorporating scores for rock angularity, brightness, particle consolidation and size distribution, scouring and deposition, and aquatic vegetation (Pfankuch, 1975). The bottom component of this index describes the stability of the material in the streams that is almost always underwater with higher scores being more unstable.
Stream discharge
Stream stage (m) was measured across streams using Level TROLL 500 pressure transducers (In-Situ Inc., Fort Collins, USA). These transducers were placed at the surface of the stream bed, and at a depth of 50 cm, across five of the sample sites (A2, A5, B1, B2, and B3), and mean stream stage was logged at 15-min intervals. Stream stage data were combined with stream rating equations derived for each cross section, enabling stream discharge to be estimated for each site, over the entire study period.
Water temperature
Stream water temperature (°C) was measured using Tinytag Plus thermistors (Gemini, Chichester, UK), inserted at the surface across five of the sample sites (A2, A5, B1, B2, and B3) at 15-min intervals. Perennial streams were identified as those where in-stream temperatures exceeded 0 °C throughout the year (Brown et al., 2006a).
Diurnal variations in water temperature, stream stage, and discharge were assessed using an index of variation (Fowler & Death, 2001). The diurnal stage variation index (DSVI) was derived using the following equation:
where Smin and Smax were the minimum and maximum stage values for every 24-h period throughout the study season (24 h was chosen because of the strong diurnal variation in flow) (Crossman et al., 2013b). Similarly, indices for Diurnal Temperature Variation (DTVI) and Diurnal Discharge Variation (DDVI) were determined.
Microbial biofilms
Samples of microbial biofilms were collected monthly at each site following the protocol described in Ledger and Hildrew (1998). For each sample, the surface area (cm2) of four randomly selected stones was calculated by tracing the stone outline onto an acetate sheet. Surfaces were then scraped into a 50 ml tube frozen and stored in the dark until analysis. The sample mass was measured using a microbalance (Ohaus, Parsippany, USA) to estimate the biofilm mass per unit area (g cm−2). A 10 ml subsample was freeze dried and chlorophyll pigments were extracted in 90% acetone for 24 h. Absorbance at 750, 664, 647, and 630 nm was determined using a spectrophotometer (Hanna Instruments, Woonsocket, USA), and total chlorophyll was calculated (Sterman 1988).
Benthic macroinvertebrates
Five replicate benthic invertebrate samples were collected monthly at each site using a Surber sampler (0.093 m2, mesh 330 µm) and preserved in 90% ethanol. Flotation was subsequently used to separate macroinvertebrates from gravel and heavier organic materials, by the addition of a saturated solution of MgSO4. The remaining mineral portion of the sample was examined for cased-caddisfly larvae and other macroinvertebrates that were not extracted via flotation.
Macroinvertebrate taxa were identified using Thorp and Covich (2001). Individuals were identified to species, wherever possible, but Simuliidae and Chironomidae were only identified to family. The abundance of different taxa was enumerated, and macroinvertebrate diversity (H) was calculated using Shannon’s Index of Diversity (Shannon & Weaver, 1949)
where Pi is the relative abundance of each taxon, calculated as the proportion of individuals of a given taxon to the total abundance within the sample.
Organic matter
Organic material collected in the Surber samples was first dried at 65 °C and then sieved into coarse (> 1 mm) and fine (< 1 mm) fractions. The weight of each fraction was measured using a microbalance (Ohaus, Parsippany, USA) prior to ashing in a furnace at 540 °C for 2 h. Ash-free dry mass was then determined by re-weighing the different organic matter fractions. From these data, the ash-free dry mass of FPOM and CPOM, as well as total organic matter (TOM), was calculated per unit area (mg m−2).
Data analysis
Objective 1: Spatial patterns in environmental and physical variables
Patterns of environmental and physical variables across sites were evaluated in relation to their elevation by correlating different variables (e.g., TOM, FPOM, CPOM, site elevation, and flow permeance) using a series of Spearman’s rank-order correlations.
Objective 2: Relationships between environmental variables and macroinvertebrate communities
Metrics for macroinvertebrate communities (total abundance and Shannon–Weaver index) were related to water source contributions, TOM, CPOM, FPOM, and site elevation using Spearman’s rank-order correlations. A Mann–Whitney U test was carried out between the macroinvertebrate diversity of ephemeral and perennial streams as diversity data were not normally distributed. Comparisons of variance in macroinvertebrate abundance between sites within and between months were completed using ANOVA, with P-values generated using F-tests.
Variation in macroinvertebrate community structure and relationships with physical and chemical variables were analyzed using detrended correspondence analysis (DCA; Hill & Gauch, 1980). DCA is an ordination technique that detrends (divides the axes into segments and centers the second axis on zero) and equalizes the weighted variance of taxon scores along the axis segments. Similar communities occur closer together on both axes. Axis 1 and Axis 2 sample scores from these analyses were correlated against physical and chemical variables (discharge, DDVI, DSVI, pH, EC, Cl− concentration, chlorophyll, FPOM, CPOM, HG, sediment particle size, site elevation, and the Pfankuch Stability Index). A DCA with independent non-parametric analysis of additional variables was selected, rather than a detrended canonical correspondence analysis which uses multiple linear regression to directly constrain relationships between sites and samples to the measured variables (Ter Braak, 1986), as it is more appropriate for smaller datasets. Water temperature data (DTVI) were not included in the DCA as this was only measured at five sites, and thus, temperature was correlated separately using Spearman’s rank-order correlation.
To determine the significance of environmental variables for individual taxa abundance, the six most dominant taxa (96% of total abundance) were correlated individually with each variable for each month (due to the strong seasonal gradient of macroinvertebrate distribution correlations) using Spearman’s rank-order correlation.
Results
Water source contributions and flow paths
Characteristics of the seven sample sites were spatially and temporally variable at a range of scales (Table 1). Sites at the low elevation end of the terrace (A1–A5 and B1) were perennial, with high percent DFSdeep. In contrast, sites further up the terrace (B2 and B3) were ephemeral, reflecting their higher elevation, with markedly lower DFSdeep contributions (Fig. 2). As glacial seepage (GMWriv) contributed more flow to the ephemeral streams, flow was only initiated after the infiltration of seasonal meltwater and precipitation raised the water table to intersect the surface.
Organic matter was similarly variable: TOM and FPOM were negatively correlated with elevation in June (P < 0.01; Fig. 3a). FPOM generally increased in all streams from June to August (Fig. 3c) and total chlorophyll in microbial biofilms increased in August by a factor of ~ 4.5 (Fig. 3d). Notably, FPOM, total chlorophyll, and CPOM were lowest in almost all sites in June (Fig. 3e).
Seasonal heterogeneity in physical and chemical characteristics of groundwater-fed streams. A Correlation between elevation (cm relative to lowest point on terrace) and mean TOM and mean FPOM in June. B Correlation between elevation and mean CPOM in July. C Seasonal changes in FPOM throughout study period. D Temporal variation in chlorophyll concentrations in August. E Site-specific increases in CPOM
Variation in macroinvertebrate communities
Macroinvertebrate diversity was significantly correlated with percent DFSdeep (P < 0.01; Fig. 2) and was higher in perennial channels than ephemeral channels (P < 0.01). The relationship between macroinvertebrate diversity and site elevation (a proxy for flow permanence; see Water source contributions and flow paths) was also significant (P < 0.05), with the highest diversity at lower elevation sites (perennial streams).
At all sites except A4, macroinvertebrate abundance was highest during August and significantly higher at B2 and B3 (P < 0.01). During June, macroinvertebrate abundance (Fig. 4a, b) was significantly correlated with percent DFSdeep and FPOM (P < 0.05; Fig. 4c). FPOM and percent DFSdeep were themselves significantly correlated (P < 0.05). During July and August, macroinvertebrate abundance was not significantly associated with DFSdeep. However, macroinvertebrate abundance was still significantly correlated with FPOM and also CPOM (P < 0.05, Fig. 4d, e).
Relationships between flow pathways, organic matter concentrations, and macroinvertebrate abundances. A Macroinvertebrate abundance and %DFSdeep in June. B FPOM and %DFSdeep in June. C Abundance and FPOM in June. D Abundance and FPOM in July. E Abundance and CPOM in July. F Abundance and TOM in July. Lines represent relationships and are not statistically derived from Spearman’s rank correlations
Seasonal variation at each of the seven sites was evident in the DCA (Fig. 5a). Sites on Axis 1 were plotted primarily according to the time of sampling, and on Axis 2 to site location (cumulative variance 90%). Perennial streams were grouped into clearly defined monthly clusters (June, July, and August). Ephemeral streams were grouped into two clusters, June and July/August. This seasonal gradient for both stream types along Axis 1 accounted for 58% of the variance in taxa distribution.
Community variation in macroinvertebrates. A Detrended correspondence analysis of macroinvertebrate taxa and sample sites. B Ordination biplot of detrended correspondence analysis, with independently correlated physical and chemical variables. Taxa: 1 Ceratopogonidae, 2 Hexatoma spp., 3 Megaleuctra spp., 4 Oligochaetae spp. A, 5 Baetis bicaudatus, 6 Chelifera spp., 7 Zapada haysi, 8 Plumiperla diversa, 9 Simuliidae, 10 Ephydra spp., 11 Limniphora spp., 12 Ecclisomyia spp., 13 Physchodidae, 14 Tipula spp., 15 Hydracarina, 16 Collembola, 17 Chironomidae, 18 Oligochaetae spp. B, 19 Alaskaperla ovibovis, 20 Baetis tricaudatus, 21 Clinocera spp., 22 Oreogeton spp., 23 Isoperla petersoni
Ephemeral streams in June and perennial streams in June and July supported similar macroinvertebrate community structure (Fig. 5b), characterized by a higher abundance of Baetis tricaudatus Dodds, 1923, Baetis bicaudatus Dodds, 1923, Clinocera spp., Zapada haysi (Ricker, 1952), and Alaskaperla ovibovis (Ricker, 1965). In late July/August, ephemeral channels supported a higher abundance of Chironomidae and Collembola whereas perennial channels supported a higher abundance of Oreogeton spp., Ecclisomyia spp., Isoperla petersoni Needham & Christenson, 1927, and Plumiperla diversa (Frison, 1935). The strong seasonal gradient of DCA Axis 1 indicated that significantly correlated variables influenced taxa on a seasonal basis, while variables that correlated more strongly with DCA Axis 2 were accounted for by between site differences (Table 2). DFSdeep% (positively correlated on DCA Axis 1 and negatively on DCA Axis 2) therefore influenced macroinvertebrate communities, both seasonally (increasing in concentration from June to August) and between sites (with greater DFSdeep seepage concentrations in perennial streams).
Patterns of dominant macroinvertebrate taxa
The relationship of the six most dominant taxa with physical and chemical variables varied seasonally (Table 3). In June, several taxa (Z. haysi, B. bicaudatus, Chironomidae, and Simuliidae [the latter found only at site A4 during June]) were correlated to water flow pathway (DFSdeep%), FPOM, and site elevation (Fig. 6a–c). Ecclisomyia spp., although low in abundance in June and only collected at sites A4 and B1, had a significant correlation with CPOM. The abundance of the predator Oreogeton spp. was significantly associated with abundance of their prey Chironomidae throughout June and July (P ≤ 0.05) (Fig. 6d, e).
Seasonal variability in distribution of individual taxa with relation to physiochemical variables. A Zapada haysi, B Chironomidae, and C Baetis Bicaudatus to DFSdeep%. D and E The distribution of Oreogeton spp. associated with that of Chironomidae throughout June and July, respectively. F and G Association of Chironomidae and Baetis bicaudatus with resource availability throughout August. H Difference between association of Oreogeton spp. with Chironomidae within perennial and ephemeral streams during August
In July, despite the change in FPOM availability, several taxa continued to demonstrate positive associations with TOM (Z. haysi, Chironomidae, B. bicaudatus, Ecclisomyia spp., and Oreogeton spp.), predominantly related to CPOM availability. Ecclisomyia spp. abundance increased markedly to become established in four of the seven stream sites. Despite initially establishing in the site furthest downstream in June (A4), Simuliidae were increasingly found at upstream sites, demonstrating a positive correlation with stream elevation throughout July and August.
Fewer taxa were associated with organic matter in August, with only Chironomidae and Baetis bicaudatus continuing to demonstrate a positive correlation (Fig. 6f, g). B. bicaudatus also demonstrated a strong association with chlorophyll, sediment grain size, and the Pfankuch Stability Index during this month. Oreogeton spp. maintained significant associations with Chironomidae in perennial streams during August but not in ephemeral streams (Fig. 6h). Ecclisomyia spp., found at all seven stream sites, was significantly associated with the Pfankuch Stability Index.
Discussion
Groundwater-fed streams were strongly influenced by local variation in water source and flow path dynamics. Here, we found that in a glacierized catchment spatial and temporal variation in these variables influenced physical and chemical conditions and as a result the macroinvertebrate communities. Across the stream sites studied, macroinvertebrate communities were related to the duration of active flow in the channels, and its subsequent influence on physical and chemical processes and organic matter dynamics. Overall, these findings highlight the combined influence of flow regime stability and organic matter content as drivers of macroinvertebrate community structure and abundance across groundwater-fed streams in glacierized catchments.
Spatial and temporal variation in environmental conditions
Spatial variation in the physical and environmental conditions present across groundwater steams on the river terrace was high, with the largest differences occurring between perennial and ephemeral streams. Perennial streams were at lower elevations on the river terrace in comparison with ephemeral streams, and there were differences in water source contributions with perennial streams supported by groundwater from DFSdeep (seepage from the valley sides), whereas ephemeral channels water source was predominantly GWriver (DFSdeep, snow and glacial meltwater) groundwater associated with the alluvial aquifer. These patterns align well with findings from previous studies which have also found spatial variation in water sources contributions at local scales in glacierized catchments (Alther et al., 2019; Zuecco et al., 2019), which concluded that flow permanence strongly influenced environmental conditions within streams (Brown et al., 2006a).
Temporal variation in environmental conditions within streams was also high. Ephemeral streams only started flowing in late summer with increases in glacial meltwater contributions to groundwater. More specifically, changes in water sources and flow paths influenced organic matter dynamics, with increases in TOM and total chlorophyll (from microbial biofilms) with highest levels observed in August. This pattern coincides with leaf fall and in-stream degradation, as well as peak primary productivity. Patterns such as this were expected in this study, but yet are uncommon for streams in glacierized catchments, which are typically supported by low levels of allochthonous organic matter due to poorly developed riparian ecotones on highly disturbed glacial floodplains (Zah & Uehlinger, 2001).
Factors influencing variation in macroinvertebrate communities
Macroinvertebrate abundance, diversity, and community composition varied spatially, across streams with different degrees of flow permanence, demonstrating the important influence of groundwater dynamics. Overall, perennial streams maintained a higher macroinvertebrate abundance and diversity in comparison with ephemeral groundwater-fed streams. Higher macroinvertebrate diversity in perennial streams can be attributed to the greater channel stability and the permanence of flow (Wood et al., 2005; Brown et al., 2006b, 2007b; Milner et al., 2020). Conversely, the low macroinvertebrate diversity observed in ephemeral channels is likely a result of flow intermittency.
There were, however, temporal variations in macroinvertebrate communities across the stream sites. For example, diversity in perennial channels increased throughout the summer (June to August) likely due to the response of macroinvertebrate taxa to increasing OM availability, particularly CPOM, in July and August. In contrast, diversity in ephemeral streams was invariant, likely reflecting limitations on colonization due to distance from the macroinvertebrate colonizing pool in the perennial streams (McArthur and Barnes 1985; Fritz and Dodds 2005) as well as the influence of short summer seasons and thus opportunities for dispersal. Colonization of ephemeral stream reaches predominantly depends upon seasonal upstream migration from perennial reaches (William & Hynes, 1976; Fengolio et al., 2002) and aerial dispersal, which across the streams studied here already had low macroinvertebrate abundance. Although flow stability is likely to be the main determinant of spatial and temporal variations in macroinvertebrate community composition (see Milner et al., 2020), a series of co-varying physical and chemical variables may be responsible for the patterns observed across these groundwater-fed streams.
Organic matter dynamics was particularly variable across and within streams. The strong positive relationship between FPOM and DFSdeep%, and subsequently macroinvertebrate abundance across flowing streams in June, indicates the potential for FPOM transport to streams by groundwater flowing through colluvial deposits on the floodplain margins (Boissier & Fontvielle, 1995). Certainly, FPOM entrainment by waters percolating through the more densely vegetated and deeper soils on the valley sides would increase the FPOM content of DFSdeep waters providing a valuable carbon resource to macroinvertebrates during periods when CPOM was limited (Richardson, 1991; Dobson & Hildrew, 1992). Furthermore, increases in CPOM due to leaf litter inputs in late July and August would explain the shift in the association of macroinvertebrates from FPOM in June to CPOM in late July and August across stream sites.
Irrespective of the organic matter source (FPOM or CPOM), there was a continued correlation between macroinvertebrate taxa and organic matter, indicative of resource tracking (Hart & Robinson, 1990; Rowe & Richardson, 2001). Two dominant mechanisms of tracking have been identified: (1) active (a deliberate emigration from sites of low to high resource availability) and (2) passive (accidental dislodgement) (Richardson, 1991; Kohler, 1985; Rowe & Richardson, 2001). However, the facultative feeding nature of some macroinvertebrates may complicate the resource tracking relationship where taxa alter their diet according to resource availability or developmental stage (Mihuc & Minshall, 1995; Moore, 1977). Here, this may be the case as the relationships between TOM and macroinvertebrates are most marked in resource-depleted environments, such as arctic and alpine streams (Tiegs et al., 2008).
Conclusions
Our findings presented here demonstrate that groundwater sources and flow paths are key variables influencing macroinvertebrate community structure in groundwater-fed streams. Our results also highlight the importance of changes in the source and timing of groundwater flow, in the context of wider uncertainties of the extent (and reliability) of the contribution to flow from snow- and ice-melt and summer rainfall. Climate change is likely to affect these streams by altering the proportional contribution of individual water sources to streamflow. This may be highly complex (e.g., Liu et al., 2020) and at present is poorly known (Jyvasjarvi et al., 2015). Given projections that maximum glacier water yield in Alaska will occur in 2060 under a RCP of 2.6 and 2090 under a RCP of 8.5 (Huss & Hock, 2018), glacier meltwater is likely to increase throughout the second part of this century in Alaska. Consequently, ephemeral streams, fed through the snow and glacial melt water, are likely to run longer over the summer and with potentially greater flow.
To determine the full extent of climatic influences on these terraces, further research is needed into the relative importance of winter macroinvertebrate diversity and abundance on summer community composition and into seasonal changes in groundwater sources associated with receding glaciers and snowpacks feeding deep aquifers.
Data availability
The data behind the paper are available on application to the corresponding author.
References
Alther, R., C. Thompson, B. Lods-Crozet & C. T. Robinson, 2019. Macroinvertebrate diversity and rarity in non-glacial Alpine streams. Aquatic Sciences 81: 42.
Anderson, M. P., 1989. Hydrogeologic facies models to delineate large-scale spatial trends in glacial and glaciofluvial sediments. Geological Society of America Bulletin 101: 505–511.
Barquin, J. & R. G. Death, 2006. Spatial patterns of macroinvertebrate diversity in New Zealand springbrooks and rhithral streams. Journal of the North American Benthological Society 25: 768–786.
Boissier, J. M. & D. Fontvieille, 1995. Biological characteristics of forest soils and seepage waters during simulated rainfalls of high intensity. Soil Biology and Biochemistry 27: 139–145.
Boulton, A. J. & P. J. Hancock, 2006. Rivers as groundwater-dependent ecosystems: a review of degrees of dependency, riverine processes and management implications. Australian Journal of Botany 54: 133–144.
Brown, L. E., D. M. Hannah & A. M. Milner, 2006a. Thermal variability and stream flow permanency in an alpine river system. River Research and Applications 22: 493–501.
Brown, L. E., D. M. Hannah & A. M. Milner, 2006b. Stability and persistence of alpine stream macroinvertebrate communities and the role of physicochemical habitat variables. Hydrobiologia 560: 159–173.
Brown, L. E., D. M. Hannah & A. M. Milner, 2007a. Vulnerability of alpine stream biodiversity to shrinking glaciers and snowpacks. Global Change Biology 13: 958–966.
Brown, L. E., A. M. Milner & D. M. Hannah, 2007b. Groundwater influence on alpine stream ecosystems. Freshwater Biology 52: 878–890.
Burgher, P., J. V. Ward & C. T. Robinson, 2002. Seasonal variations in zoobenthos across habitat gradients in an alpine glacial floodplain (Val Roseg, Swiss Alps). Journal of the North American Benthological Society 21: 561–575.
Colls, M., X. Timoner, C. Font, S. Sabater & V. Acuña, 2019. Effects of duration, frequency, and severity of the non-flow period on stream biofilm metabolism. Ecosystems 22: 1393–1405.
Crespo-Perez, V., O. Dangles, C. Ibarra, R. Espinsosa, P. Andino, D. Jacobsen & S. Cauvy-Fraunie, 2020. Functional structure and diversity of invertebrate communities in a glacierized catchment of the tropical Andes. Freshwater Biology 65: 1348–1362.
Crossman, J., C. Bradley, I. Boomer & A. M. Milner, 2011. Water flow dynamics of groundwater-fed streams and their ecological significance in a glacierized catchment. Arctic, Antarctic and Alpine Research 43: 364–379.
Crossman, J., C. Bradley, J. N. W. David & A. M. Milner, 2012. Use of remote sensing to identify areas of groundwater upwelling on active glacial floodplains: their frequency, extent and significance on a landscape scale. Remote Sensing and the Environment 123: 116–126.
Crossman, J., M. N. Futter & P. G. Whitehead, 2013a. The significance of shifts in precipitation patterns: modelling the impacts of climate change and glacier retreat on extreme flood events in Denali National Park, Alaska. PLoS ONE 8: e74054.
Crossman, J., C. Bradley, A. M. Milner & G. Pinay, 2013b. Influence of environmental instability of groundwater-fed streams on hyporheic fauna, on a glacial floodplain, Denali National Park, Alaska. River Research and Application 29: 548–559.
Dobson, M. & A. G. Hildrew, 1992. A test of resource limitation among shredding detritivores in low order streams in Southern England. Journal of Animal Ecology 61: 68–77.
Durkota, J. M., P. J. Wood, T. Johns, J. R. Thompson & R. J. Flower, 2019. Distribution of macroinvertebrate communities across surface and groundwater habitats in response to hydrological variability. Fundamental and Applied Limnology 193: 79–92.
Fengolio, S., P. Agosta, B. Tiziano & M. Cucco, 2002. Field experiments on colonization and movements of stream invertebrates in an Apennine river (Visone, NE Italy). Hydrobiologia 474: 125–130.
Fowler, R. T. & R. G. Death, 2001. The effect of environmental variability on hyporheic community structure. Hydrobiologia 445: 85–95.
Fritz, K. M. & W. K. Dodds, 2005. Harshness: characterisation of intermittent stream habitat over space and time. Marine and Freshwater Research 56: 13–23.
Füreder, L., C. Schütz, M. Wallinger & R. Burger, 2001. Physico-chemistry and aquatic insects of a glacier-fed and a spring-fed alpine stream. Freshwater Biology 46: 1673–1690.
Gilbert W. G., C. J. Nye & K. W. Sherwood, 1984. Stratigraphy, petrology and geochemistry of upper Triassic rocks from the Pingston and McKinley terrains, Central Alaska Range. Division of Geological and Geophysical Surveys State of Alaska Department of Natural Resources, Fairbanks, AK, USA.
Hart, D. D. & C. T. Robinson, 1990. Resource limitation in a stream community: phosphorus enrichment effects on periphyton and grazers. Ecology 71: 1494–1502.
Hill, M. O. & H. G. Gauch, 1980. Detrended correspondence analysis: an improved ordination technique. Advances in Vegetation Science 2: 47–58.
Huss, M. & R. Hock, 2018. Global-scale hydrological response to future glacier mass loss. Nature Climate Change 8: 135–148.
Jyvasjarvi, J., H. Marttila, P. M. Rossi, P. Ala-Aho, B. Olofsson, J. Nisell, B. Backman, J. Ilmonen, R. Virtanen, L. Paasivirta, R. Britschgi, B. Kløve & T. Muotka, 2015. Climate-induced warning imposes a threat to north European spring ecosystems. Global Change Biology 21: 4561–4569.
Kath, J., E. Harrison, B. J. Kefford, L. Moore, P. J. Wood, S. B. Schafer & F. Dyer, 2016. Looking beneath the surface: using hydroecology and traits to explain flow variability effects on stream invertebrates. Ecohydrology 9: 1480–1495.
Khamis, K., L. E. Brown, D. M. Hannah & A. M. Milner, 2016. Glacier-groundwater stress gradients control alpine river biodiversity. Ecohydrology 9: 1263–1275.
Kohler, S. L., 1985. Identification of stream drift mechanisms: an experimental and observational approach. Ecology 66: 1749–1761.
Ledger, M. E. & A. G. Hildrew, 1998. Temporal and spatial variation in epilithic biofilm of an acid stream. Freshwater Biology 40: 655–670.
Liu, W., R. T. Bailey, H. E. Andersen, E. Jeppesen, A. Nielsen, K. Peng, E. Molina-Navarro, S. Park, H. Thodsen & D. Trolle, 2020. Quantifying the effects of climate change and stream biota in a groundwater-dominated catchment: a modeling approach combining SWAT MODFLOW with flow-biota empirical models. Science of the Total Environment 745: 140933.
Malard, F., 2003. Groundwater-surface water interactions. In Ward, J. V. & U. Uehlinger (eds), Ecology of a Glacial Floodplain. Aquatic Ecology Series 1 Springer, New York: 37–56.
Malard, F., K. Tockner & J. V. Ward, 1999. Shifting dominance of subcatchment water sources and flow paths in a glacial floodplain, Val Roseg, Switzerland. Arctic, Antarctic and Alpine Research 31: 135–150.
Malard, F., A. Mangin, U. Uehlinger & J. V. Ward, 2001. Thermal heterogeneity in the hyporheic zone of a glacial floodplain. Canadian Journal of Fisheries and Aquatic Sciences 58: 1319–1335.
Mathers, K. L., M. J. Hill & P. Wood, 2017. Benthic and hyporheic macroinvertebrate distribution within the heads and tails of riffles during baseflow conditions. Hydrobiologia 794: 17–30.
McArthur, J. V. & J. R. Barnes, 1985. Patterns of macroinvertebrate colonisation in an intermittent rocky mountain stream in Utah. Great Basin Naturalist 45: 117–123.
Mihuc, T. B. & G. W. Minshall, 1995. Generalists vs. trophic specialists: implications for food web dynamics in post-fire streams. Ecology 76: 2361–2372.
Milner, A. M., C. Docherty, F. M. Windsor & K. Tojo, 2020. Macroinvertebrate communities in streams with contrasting water sources in the Japanese Alps. Ecology and Evolution 10: 7812–7825.
Moore, J. W., 1977. Some factors affecting algal consumption in subarctic Ephemeroptera, Plecoptera and Simuliidae. Oecologia 27: 261–273.
Murray, B., M. J. B. Zeppel, G. C. Hose & D. Eamus, 2004. Groundwater-dependent ecosystems in Australia: it’s more than just water for rivers. Ecological Management and Restoration 4: 110–113.
Murray, B. R., G. C. Hose, D. Eamus & D. Licari, 2006. Valuation of groundwater-dependent ecosystems: a functional methodology incorporating ecosystem services. Australian Journal of Botany 54: 221–229.
Pfankuch, D. J., 1975. Stream Reach Inventory and Channel Stability Evaluation, Department of Agriculture Forest Service Region, Missoula:
Richardson, J. S., 1991. Seasonal food limitation of detritivores in a montane stream: an experimental test. Ecology 72: 873–887.
Robinson, Z. P., I. J. Fairchild & A. J. Russel, 2008. Hydrological implications of landscape evolution at Skeðaràrsandur, SE Iceland. Geomorphology 97: 218–236.
Rowe, L. & J. S. Richardson, 2001. Community responses to experimental food depletion: resource tracking by stream invertebrates. Oecologia 129: 473–480.
Sertić, Perić M., J. M. Nielsen, C. J. Schubert & C. T. Robinson, 2021. Does rapid glacial recession affect feeding habits of alpine stream insects? Freshwater Biology 66: 114–129.
Shannon, C. E. & W. Weaver, 1949. The Mathematical Theory of Communication, University of Illinois Press, Urbana:
Sterman, N. T., 1988. Spectrophotometric and fluorometric chlorophyll analysis. In Loban, C. S., D. J. Hapman & B. P. Kremer (eds), Experimental Phycology: A Laboratory Manual Cambridge University Press, Cambridge: 35–46.
Stromberg, J. C. & D. M. Merritt, 2016. Riparian plant guilds of ephemeral, intermittent and perennial rivers. Freshwater Biology 61: 1259–1275.
ter Braak, C. J. F., 1986. Canonical correspondence analysis: a new eigenvector technique for multivariate direct gradient analysis. Ecology 67: 1167–11779.
Thorp, J. H. & A. P. Covich, 2001. Ecology and Classification of North American Freshwater Invertebrates, 2nd ed. Academic Press, San Diego:
Tiegs, S. D., F. D. Peter, C. T. Robinson, U. Uehlinger & M. O. Gessner, 2008. Leaf decomposition and invertebrate colonization responses to manipulated litter quantity in streams. Journal of the North American Benthological Society 27: 321–331.
Western Regional Climate Centre, 2008. Toklat Alaska; Station Daily Time Series. [available on internet at: http://www.raws.dri.edu].
Williams, D. D. & H. B. N. Hynes, 1976. The recolonization methods of stream benthos. Oikos 27: 265–272.
Windsor, F. M., M. T. Grocott & A. M. Milner, 2017. An inter-catchment assessment of macroinvertebrate communities across groundwater-fed streams within Denali National Park, interior Alaska. Hydrobiologia 785: 373–384.
Wood, P. J., J. Gunn, H. Smith & A. Abas-Kutty, 2005. Flow permanence and macroinvertebrate community diversity within groundwater dominated headwater streams and springs. Hydrobiologia 545: 55–64.
Zah, R. & U. Uehlinger, 2001. Particulate organic matter inputs to a glacial stream ecosystem in the Swiss Alps. Freshwater Biology 46: 1597–1608.
Zuecco, G., L. Carturan, F. De Blasi, R. Seppi, T. Zanoner, D. Penna, M. Borga, A. Carton & G. Dalla Fontana, 2019. Understanding hydrological processes in glacierized catchments: evidence and implications of highly variable isotopic and electrical conductivity data. Hydrological Processes 33: 816–832.
Acknowledgements
We would like to thank Lewis Blake, Nicholas Hale, and Daniel Woodhouse for their field assistance, and Pamela Sousanes, Lucy Tyrell, and Craig Brandt of Denali National Park for help with logistics and equipment and also thank Melanie Bickerton for laboratory and data analysis and Richard Johnson for technical support at UoB. Fieldwork for this research was sponsored by a NERC research studentship, with additional support provided by the Denali Foundation.
We would also like to thank the anonymous reviewers, associate editor, and editor for their helpful comments on the manuscript. This paper is dedicated to the memory of Jill Crossman: Jill studied the ecohydrology of upwelling streams in Denali National Park, Alaska for her PhD dissertation at the University of Birmingham UK. On leaving Birmingham, Jill undertook postdocs at the universities of Oxford (UK) and Trent (Canada) and was then appointed to a permanent position as Assistant Professor at the University of Windsor in Canada. Jill was a member of the Great Lakes Institute for Environmental Research (GLIER) where her research focused on lake nutrient dynamics and microplastics. No matter if it was fieldwork or a social occasion, life was never dull around Jill – she was exuberant, genial, and spontaneous, but also an excellent and dedicated scientist who will be sorely missed by all who knew her.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest.
Additional information
Handling editor: Sally A. Entrekin
This paper is dedicated to the memory of Jill Crossman and was revised and published after her untimely passing.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Crossman, J., Bradley, C., Windsor, F.M. et al. Water source dynamics influence macroinvertebrate communities across groundwater-fed streams in a glacierized catchment. Hydrobiologia 850, 1801–1816 (2023). https://doi.org/10.1007/s10750-023-05182-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10750-023-05182-x