Abstract
Understanding how lakes respond to changes in nutrient loading along a productivity gradient can help identify key drivers of aquatic change, thereby allowing appropriate mitigation strategies to be developed. Physical, chemical and biological water column measurements combined with long-term water monitoring data for six closely located crater lakes, in Southeast Asia, were compared to assess the response of lakes along a productivity gradient equating to a transect of increasing aquaculture intensity. Increasing chlorophyll a (phytoplankton biomass) in the upper waters appeared to modify the thermocline depth and light availability causing a shift from a deep chlorophyll maximum at low aquaculture intensity to the emergence of algal dead zones lower in the water column with high aquaculture intensity. High phosphorus loading and light limitation from enhanced algal biomass, associated with high aquaculture intensity, exacerbated nitrogen drawdown, leading to the prevalence of potentially nitrogen-fixing cyanobacteria. Seasonal overturn during the cooler season resulted in low dissolved oxygen concentrations in the epilimnion, potential harmful algal blooms, a reduction in the habitable depth for fish and ultimately increased mortality amongst farmed fish.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The scale of the human impact on tropical freshwater systems is substantial, often inhibiting their ability to provide the ecosystem services on which local populations depend (Legaspi et al., 2015; Briddon et al., 2020; de Leon et al., 2020; May et al., 2021). In the Philippines, as in many parts of Southeast Asia, the utilisation of freshwater lakes and their surrounding catchments for aquaculture, agriculture (e.g. coconut plantations, Cocos nucifera L.) and supporting human settlements contributes significantly to the economy, food security and employment (Cordero & Baldia, 2015). Freshwater and marine aquaculture production of fish, crustaceans and molluscs contributed to over 1.79 billion USD to the Philippine economy in 2018 (Bureau of Fisheries and Aquatic Resources, 2019). Anthropogenic activities such as deforestation, urbanisation and intensive aquaculture frequently lead to deterioration in lake ecosystem integrity due to increased nutrient inputs (Bannister et al., 2019). This may result in eutrophication, the occurrence of in harmful algal blooms (HABs), overall degradation of water quality (Heisler et al., 2008) and cascading negative impacts on productivity and aquaculture yield in freshwater lakes (Jeppesen et al., 2010).
Aquaculture can cause nutrient enrichment, either directly from the over-use of fish food or in the form of fish waste (Holmer, 2002), leading to eutrophication. Up to 85% of phosphorus (P) and 52 to 95% of nitrate (NO3−) in fish feed from aquaculture is lost to the environment through waste and excretion (Southgate & Lucas, 2003). Eutrophication causes increased hypolimnetic dissolved oxygen (DO) consumption, progressively lowering DO concentrations in the water column and enhancing diurnal DO fluctuations (Mesman et al., 2021). Continued eutrophication can result in the complete exhaustion of DO and the formation of hypoxic or anoxic conditions in the hypolimnion. As many freshwater bodies in the Philippines are the basis of key social-ecological systems, eutrophication not only adversely effects aquatic organisms, but can also jeopardise local livelihoods by reducing revenue from aquaculture (Szekielda et al., 2014).
Latitudinal differences in climatic conditions lead to distinctive seasonality in mixing, stratification and phytoplankton succession in tropical lakes (Cordero & Baldia, 2015), which may have distinctive vertical (water column) structures that are susceptible to seasonal variations (Kraemer et al., 2015; Lau et al., 2020; Mesman et al., 2021). However, the magnitude of seasonal temperature changes is smaller than at higher latitudes, meaning that some tropical lakes never completely mix (Boehrer & Schultze, 2008; Bannister et al., 2019), whilst in others, seasonal mixing may occur as a disturbance effect of monsoonal airflows or cyclones (Klug et al., 2012; Mendoza-Pascual et al., 2021). Lakes at low latitudes are also potentially more sensitive to changes in temperature and nutrient availability than their cooler climate equivalents (Lewis, 1987; Adrian et al., 2009), whilst nitrogen (N) may be more limiting, and nutrient cycling more efficient (Abell et al., 2012; Bannister et al., 2019). N limitation in tropical lakes is enhanced by high internal N loss rates through denitrification (the reduction of NO3− to N2) (Lewis, 2000). Denitrification is stimulated by anoxia and high temperatures, and under these conditions surficial sediments lose NO3− rapidly and planktonic denitrification occurs leading to a further depletion of NO3− (Lewis, 2000). This reduces the amount of NO3− available for redistribution during mixing (Gardner et al., 1998). Furthermore, supply of P through internal loading and natural P supply from chemical weathering in catchments occur at higher rates in warm, humid conditions (Lewis, 2000; Lau et al., 2020; May et al., 2021). Increased thermal stability and prolonged stratification increases sequestration of nutrients in the hypolimnion (Lau et al., 2020), further influencing the nutrient balance in tropical lakes (Gao et al., 2019).
These unique characteristics create challenges for the management and restoration of freshwater ecosystems in the tropics, especially when readily available interventions have been tailored to suit conditions at higher latitudes. Nowhere is this more evident than in tropical Southeast Asia, a region that is undergoing some of the highest rates of environmental change on Earth and where freshwater pollution is commonplace and increasing (Zhou et al., 2020). External stressors on the Seven Crater Lakes of San Pablo on the island of Luzon, the largest of the archipelago of islands that make up the Philippines, arise from multiple forms of rapid environmental changes, and exemplify those facing water bodies throughout the wider region. The pressures are both exogenous and endogenous; cage aquaculture is commonplace, plantation agriculture is a predominant land cover leading to run-off, and both urbanisation and tourism (and the depositing of untreated sewage) and associated demands on resources have expanded rapidly (Brillo, 2015b; Bannister et al., 2019). Monitoring of the lakes by the Laguna Lakes Development Authority (LLDA) 1996–2016 reveals a recent history of significant eutrophication, fish kills, HABs and water hyacinth proliferation (Brillo, 2015a, 2016a, 2016b, 2016c; LLDA, 2009; Santiago & Arcilla, 1993; Zafaralla, 2010). The LLDA uses a DO concentration of < 5 mg/l to delineate “Class C” lakes (Department of Environment and Natural Resources Resources, 2021), below which fish health is affected. All lakes were borderline or fell below this DO criteria over the 1996–2016 monitoring period (LLDA, 2009), indicating significant threats to the sustainability of aquaculture in the lakes. Given that the extent of mixing influences oxygen distribution in the lakes, thereby potentially impacting the viability of aquaculture, a better understanding of interactions between mixing, primary productivity and aquaculture will benefit those involved in lake management in the tropics, including fish-farmers and their dependents.
Here, we determine variations in the vertical profile of physical and chemical characteristics, nutrients and photosynthetic pigments along a gradient of increasing aquaculture intensity (determined using a freedom of information request from the LLDA) and water quality degradation in six of the Seven Crater Lakes of San Pablo. The study aims to provide a basis for assessing the risks posed to aquatic ecosystem functioning and to identify possible interventions aimed at ensuring the resilience and sustainability of these critically important social-ecological systems.
Materials and methods
Study area
Located in Southeast Asia, the Philippines generally experiences a warm, humid climate that is influenced by the effects of seasonal (monsoonal) airflows, topography, location and the tropical Pacific Ocean. The study area—focussed on the Seven Crater Lakes of San Pablo, is found in Laguna Province in the southern part of the island of Luzon (Fig. 1) and has an average precipitation of 3178 mm per annum, with a drier season from January to June (averaging 124 mm per month) and a wetter season from July to December (381 mm per month). Mean annual temperature is c. 26.5 °C and humidity is c. 88% (1901–2017) (Harris et al., 2014), with cooler temperatures in January to February (average of 25.5 °C) and warmer temperatures in April to July (average of 28.3 °C). Tropical storms (typhoons) forming over the northwest Pacific, particularly those that go on to make landfall, are increasing in intensity and destructive potential (Mei & Xie, 2016), with a high proportion passing close to or over the Philippines (Kubota & Chan, 2009; Cinco et al., 2016). Typhoons can occur year-round, although they mainly occur from June through to September. In a typical year, 15 typhoons (or tropical cyclones) enter the Philippines with five or six making landfall (Kubota & Chan, 2009).
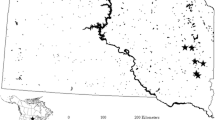
Study area of the Seven Crater Lakes of San Pablo showing A the main land uses and (inset) the location within the Philippines (Geographic coordinate system GCS_WGS_1984; Source: Arc GIS Open Data, ESRI.); B mean fish density for each lake since 1976 to 2018 provided by the LLDA; C photographs of each lake showing the main activities at each site
The Seven Crater Lakes of San Pablo (Bunot, Calibato, Mohicap, Palakpakin, Pandin, Sampaloc and Yambo) are volcanic in origin, relatively small and deep maars (Fig. 1). Calibato was excluded from this study owing to its extreme depth (> 150 m). Geochemical studies of the Taal Volcano (part of the same volcanic field as the study area) show that lava composition ranges from calcareous-alkaline to iron enriched (Miklius et al., 1991). Aquaculture in the lakes mostly involves the cultivation of Tilapia [Oreochromis niloticus (Linnaeus, 1758)], first introduced in the 1970s (Bureau of Fisheries and Aquatic Resources, 2019). Plantations of coconut were introduced much earlier, towards the end of the seventeenth century, and are now extensive in the study area (Migrino, 2017). San Pablo City, which encompasses Lake Sampaloc, is the largest urban centre in the study area, with a population of c. 266,000 (2015; PhilAtlas, 2020). Sampaloc has a number of structures, such as a boardwalk around the edge of the lake, houses, hotels, restaurants and a large church, many of which were constructed in the catchment in the 1980–1990s (Brillo, 2016c). Ecotourism, which initially focussed on Lake Pandin, has expanded to include the other lakes (Brillo, 2017).
Primary and secondary data collection
Previous water quality monitoring
Water quality data for the six study lakes are available from the LLDA from 1996 to 2016 (for the complete methodology see Brillo, 2016c). This data is comprised of chlorophyll a, ammonia (N–NH3), nitrate (N–NO3–) and phosphate (P–PO43−) (Table 1). Comparison with previous water quality monitoring can help determine the changing trends in nutrient concentrations over time and how they compare with the values collected in May 2017. Previous studies have also measured variations of temperature and DO with depth during the cooler and drier (February) and warmer and wetter (August and November) seasons of 2017 (Bannister et al., 2019; Mendoza et al., 2019) (summarised in Figure SI1). Furthermore, Mendoza et al. (2019) reported temperature and DO measurements for all six lakes from August 2018.
High resolution water column sampling
Additional sampling for water, photosynthetic pigments and surface sediments took place between the 10th and 17th May 2017. Physical and chemical measurements of the water column took place in the deepest part of the six lakes (Table 1 for GPS coordinates). A YSI EXO1 multi-probe with chlorophyll a, DO and temperature sensors logged data every second whilst being lowered from the surface to the lake bottom. For the full calibration and measuring procedures see https://www.ysi.com/File%20Library/Documents/Manuals/EXO-User-Manual-Web.pdf. Water clarity was estimated using light intensity readings collected using a LI Cor Sensor Light meter LI192, at 1 m depth intervals.
Water chemistry and nutrient analysis
Two litres of water were collected from the surface, mid and bottom depths of each lake, using a Van Dorn sampler. The water was filtered using a GF/F Whatman filter (0.45 μm) and was analysed for silicate using the molybdate yellow method, soluble reactive phosphorus (SRP) using the molybdenum blue method and ammonium (N-NH4+) using the methods described in Mackereth and Heron (1979). The accuracy of each method was determined using a set of five calibration standards along a concentration gradient. Unfiltered water underwent a persulphate digestion for total phosphorus (TP) analysis.
Photosynthetic pigment sampling and analysis
A measured volume of water was filtered using GF/F Whatman glass-fibre filters (0.45 μm) to collect sufficient filtrate for photosynthetic pigment analysis. Filtrate samples were stored frozen prior to analysis. Surface sediment pigments were acquired from a single sediment core collected (using a UWITEC (gravity) corer) from the deepest part of each lake from Lakes Sampaloc, Palakpakin, Bunot and Mohicap in May 2017 and from Lakes Yambo and Pandin in April 2018 (Table 1). Immediately after collection, the cores were subsampled into 0.5 cm depth intervals which were subsequently analysed for sediment organic content by loss-on-ignition and for chlorophyll and carotenoid pigments. A radiometric isotope chronology was developed to date the sediment horizons (see supplementary information; Figures SI2–13; Tables SI1–12). The chronologies were used to identify the recent years of sediment deposition since 2010 CE. Only sub-samples dated post-2010 were used in this study.
Analysis of photosynthetic pigments (chlorophylls, their degradation products and carotenoids) was conducted on water filtrates and freeze-dried surface sediment samples by reversed-phase HPLC with online photo diode array (PDA) spectrophotometry. Pigments were extracted in HPLC-grade acetone: methanol: water (80:15:5) overnight at − 4 °C, extracts were filtered using a 0.22 µm PTFE syringe filter and dried under N2 gas. Samples were redissolved in an injection solution of acetone: ion pairing reagent (consisting of 1.875 g tetrabutyl ammonium acetate, 19.35 g ammonium acetate and 250 ml deionised water): methanol (70:25:5) (Chen et al., 2001). Pigments were separated using reversed-phase HPLC (McGowan, 2007) with a mobile phase of solvent A (80:20 methanol: 0.5 m ammonium acetate), solvent B (90:10 acetonitrile: deionised water), solvent C (100% ethyl acetate) and a stationary phase consisting of an ODS Hypersil column (25 × 4.5 mm; 5 µm particle size) following a modification to the conditions of Chen et al. (2001). Pigment identification was based on retention time and spectral characteristics and the peak areas were calibrated using commercial standards (DMI Denmark).
Three photosynthetic pigments were chosen to represent the different important primary producer groups; total primary producers (chlorophyll a), cyanobacteria (canthaxanthin) and anaerobic phototroph photosynthetic, purple sulphur bacteria (okenone). A mean of pigment concentrations in all sub-samples dated post-2010 was used to determine how pigment concentrations changed along the aquaculture gradient.
Numerical analyses
To determine if there were significant differences in the nutrient concentrations between lakes, one-way ANOVAs with Tukey-adjusted multiple comparisons were conducted on P–PO43−, N–NO3- and N–NH3 concentrations for all the lakes.
The mixing depth in each water column was determined using the depth of the thermocline in May 2017 as measured by the multiprobe. The thermocline was defined as the thin but distinct layer in which there is a substantial drop in temperature compared to the layers above and below (Fig. 2; Fiedler, 2010). Euphotic depth (the depth to which 1% of incident irradiance penetrates) was calculated from the light intensity measurements collected at 1 m depth intervals. The photic zone was then calculated using the following equation (Degefu et al., 2014):
Defined parameters: Vertical light extinction coefficient ε per metre, Io is surface light irradiance, Iz is light irradiance at depth Z.
The euphotic depth (Zeu) was calculated (Degefu et al., 2014) using:
The upper water column light intensity measurements were used to calculate the photic zone (r2 > 0.8; Figure SI14), excluding values from deeper in the water column that deviated from the linear regression (Staehr et al., 2012; Obrador et al., 2014; Ehrenfels et al., 2020).
Principal components analysis (PCA) was conducted on pigment samples from the water column including chlorophyll a, chlorophyll b, β carotene, pheophyin b, canthaxanthin, diadinoxanthin, diatoxanthin, myxoxanthophyll, peridinin and okenone from all depths. Prior to analysis, all pigment abundances and explanatory variables were standardised using a log(x + 1) transformation to normalise the data and to reduce the asymmetry of distribution (Legendre & Birks, 2012). The transformed pigment records from each of the study lakes were analysed using the indirect ordination method of PCA using CANOCO 5. PCA was carried out on the water column pigment assemblages, to identify trends or clusters in the data. A further redundancy analysis (RDA) ordination (using CANOCO 5) with constrained ordination (using forward selection) was conducted to explore correlations between water chemistry and pigment (phototroph) assemblages. Temperature, DO, TP and nutrients (silicate, N-NH4+) were included individually with only significant variables (P < 0.05) being included in the final RDA.
Results
Previous water quality monitoring
LLDA monitoring data indicated a strong increasing gradient in lake nutrient concentrations Yambo → Pandin → Mohicap → Palakpakin → Sampaloc → Bunot (Table 1) corresponding well to the aquaculture gradient. Concentrations of N–NH3 and P–PO43− levels were statistically lower (P value < 0.05) in the lakes with low levels of aquaculture (Yambo and Pandin) compared to those with higher levels of aquaculture (Mohicap, Sampaloc, Bunot and Palakpakin; see ANOVA results in Tables SI13 and SI14). There were no significant differences in N–NO3- concentrations between the two groups. N–NH3 concentration increased significantly in Palakpakin, Bunot, Sampaloc and Mohicap between 1996 and 2016. There was a significant decline in P–PO43− concentrations in Sampaloc and Pandin over the same period. Nutrient concentrations remained stable in Yambo. Mean chlorophyll a concentrations were similar for all the lakes ranging from 31.22 to 40.44 μg/l (Table 1).
Quarterly temperature profiles reported by Bannister et al. (2019) (Figure SI1) on Mohicap, Yambo and Sampaloc found uniformly mixed temperatures down the water column during the cooler months (February; December in Mohicap and Yambo only). DO concentrations declined between the epilimnion and hypolimnion, coincident with the thermocline in August and November 2017 for all three lakes. In Yambo and Mohicap, the DO concentrations became uniform in February 2017, but this did not happen in Sampaloc. The temperature and DO measurements collected in August 2018 (Mendoza et al., 2019) showed a well-mixed water column in Palakpakin, the shallowest of the six study sites, yet there was a distinct difference between the epilimnia and hypolimnia of the other lakes consistent with the measurements collected in 2017.
Depth profiles in the water column (May 2017)
Vertical variations in temperature, chlorophyll a and DO showed distinct differences across a gradient of increasing intensity of aquaculture (Fig. 2). A distinct thermocline was present in all lakes except Palakpakin (Fig. 2a). The thermocline was deepest in Yambo and Pandin at c.8–9 m, and between 5 and 6 m in Mohicap, Sampaloc and Bunot. The mixing depth of the lake varied in accordance with thermocline depth, as did the depth of the photic zone (Fig. 2).
The euphotic depth ranged from 2.25 m (Palakpakin) to 16.78 m (Yambo) and was ordered from deepest to shallowest as Yambo → Pandin → Mohicap → Sampaloc → Bunot → Palakpakin (Table 1; Fig. 2). The photic zone was deeper than the thermocline in Yambo, Pandin and Mohicap.
Deep-water chlorophyll maxima (DCM) in the form of peak concentrations of chlorophyll a occurred close to the thermocline in Yambo, Pandin and Mohicap (Fig. 2b). In Palakpakin, Bunot and Sampaloc, chlorophyll a concentrations declined gradually in the upper waters with no distinct DCM. Highest chlorophyll a concentrations occurred in Palakpakin (40 μg/l) with the lowest maxima at the DCM in Yambo and Mohicap (c. 8 μg/l). In Pandin, there were two spikes in chlorophyll a concentrations at 5.7 m and 11.1 m depths. In all lakes except Sampaloc and Bunot, the euphotic depth extended below the peak in chlorophyll a concentrations.
DO concentrations were highest in the epilimnion of Bunot (15 mg/l), whereas the highest DO concentrations in the other lakes ranged from 6.97 to 9.22 mg/l. Below the mixing depth, all lakes showed anoxic or near anoxic conditions in the deeper waters, including Palakpakin, (Fig. 2c). The DO profile mirrored the thermocline, with the highest concentrations of DO found in the mixing zone.
Nutrients
TP, SRP, N-NH4+ and silicate concentration maxima occurred deeper in the water column except in Palakpakin, where nutrient concentrations showed little variation with depth (Figure SI15). Highest concentrations of TP (c. 1500 μg/l) and SRP (c. 1000 μg/l) were found in Bunot and Sampaloc, whilst highest N-NH4+ concentrations (1929–1961 μg/l) were found in the hypolimnia of Pandin, Sampaloc and Bunot. Sampaloc was the only lake where N-NH4+ was detected in the surface waters with a concentration of 1929 μg/l. All lakes except Yambo had detectable concentrations of N-NH4+. Silicate concentrations were relatively uniform down the water column for all lakes, with concentrations of 7–9 mg/l in Palakpakin, Bunot, Mohicap and Pandin and c.4 mg/l and c.2 mg/l respectively in Yambo and Sampaloc.
In comparison to the LLDA data, the P–PO43− concentrations in Yambo, Pandin, Mohicap, Bunot and Palakpakin (but not Sampaloc) were substantially lower in May 2017 than those collected by the LLDA. As different forms of N were collected N-NH3 (by the LLDA) and N–NH4+ (in May 2017) it was not possible to compare the two datasets.
Primary producer assemblages
Low concentrations (< 0.03 nmol/l) of β carotene, diatoxanthin, chlorophyll b and pheophytin b pigments were observed throughout the water column of Yambo and Pandin (Figure SI15). In comparison, Sampaloc, Palakpakin and Bunot showed a wider variety and higher concentrations (0.1–3.2 nmol/l) of pigments from algal groups; cyanobacteria (canthaxanthin and myxoxanthophyll), siliceous algae (diatoxanthin and diadinoxanthin) and chlorophytes (lutein and chlorophyll b). These three lakes also had the highest concentrations of chlorophyll a and β carotene (indicators of total algal production), with the highest values in the surface waters of Bunot (3.2 and 0.5 nmol/l, respectively).
PCA identified two main groupings of water column pigments (Fig. 3). The first, samples from Palakpakin were distinguished by high abundances of canthaxanthin (cyanobacteria). The second, containing the remaining samples, was characterised by low abundance of canthaxanthin (cyanobacteria) and the presence of chlorophyll a, β carotene, the degradation product of chlorophyll b, pheophytin b, cyanobacterial pigments (myxoxanthophyll, lutein-zeaxanthin) and okenone (purple sulphur bacteria).
A PCA biplot of the pigment data separated by each lake and depth, B RDA biplot of algal community variability constrained against total phosphorus in the study lakes. Total phosphorus was the only variable significantly correlated with the pigments in the RDA identified through forward selection (P = 0.014). Samples from each lake are coloured: Yambo (purple), Pandin (green), Mohicap (white), Sampaloc (blue), Bunot (black), Palakpakin (red) and sampling depth indicated by shapes (see legend)
For the RDA, forward selection identified TP as the only variable that was significantly (P < 0.05) correlated with photosynthetic pigments in the water samples. The RDA showed a distinct trophic gradient with the oligotrophic/mesotrophic lakes grouped to the upper right quadrant (characterised by low chlorophyll a) and the eutrophic/hypereutrophic lakes grouped to the bottom left quadrant (characterised by high chlorophyll a). Palakpakin was again characterised by high canthaxanthin concentrations.
Surface sediment pigments deposited since 2010
When arranged along the gradient of increasing aquaculture intensity, the mean concentrations of chlorophyll a, canthaxanthin and okenone in surface sediment samples (Fig. 4) initially increased, reaching peak concentrations in Sampaloc, before declining (chlorophyll a and canthaxanthin) or dropping to levels that were undetectable (okenone) in lakes with the highest level of aquaculture (Bunot and Palakpakin).
Discussion
Morphometric conditions are important in determining whether lakes stratify (Boehrer & Schultze, 2008). In addition to being shallow and polymictic, Palakpakin has a relatively low residence time (Brillo, 2016a), which may further encourage lake mixing (supported by the water column measurements taken in August 2018 (Mendoza et al., 2019)). For the other deeper crater lakes, thermocline depth generally correlated with lake depth. In temperate regions, thermocline depth depends mainly on lake surface area because of the importance of fetch for wind mixing (Gorham & Boyce, 1989; Gunkel & Beulker, 2009). The small surface area of the study sites and their generally steep-sided catchments precluded lake area being a factor in determining differences in thermocline depth. Disturbance by strong winds and storms is also unlikely to have been a factor, given that sampling took place outside the main typhoon season and the sheltering effect from the steep walls of the catchments. Yambo, Mohicap, Sampaloc and Bunot have similar maximum depths (between 23 and 36 m) but very different photic zone depths (ranging from 16.7 m in the case of Yambo to 3.25 m in the case of Bunot). There is a markedly deeper thermocline in Yambo (depth of 15 m) compared with the other lakes, suggesting that water clarity could play an important role, as has been reported elsewhere (Kling, 1988; Bartosiewicz et al., 2019). Clearer waters allow greater solar radiation penetration, thereby facilitating deeper water mixing (Heiskanen et al., 2015). Further evidence of the impact of water clarity on thermocline depth is provided in the quarterly monitoring data for Yambo in February 2017 (Bannister et al 2019). The data indicated that a breakdown in stratification resulted in the thermocline most likely reaching the lake bottom (e.g. complete mixing, which was not observed in the other lakes). In the more productive lakes, such as Bunot, increased chlorophyll a concentrations could thus have caused a shallowing of thermocline depth.
Differences in photic zone depth, the part of the water column where photosynthesis takes place (Lewis, 2010), represent a gradient in water clarity, with deeper photic zones in the oligotrophic/mesotrophic lakes. The most likely explanation is that phytoplankton biomass in the epilimnion controls the depth to which light can penetrate (Longhi & Beisner, 2009), with the photic zone either deeper than (Yambo, Pandin), ± equal to (Mohicap) or shallower than (Sampaloc, Bunot, Palakpakin) the mixing depth. In the lakes where the photic zone exceeded the mixing depth (Yambo, Pandin), the highest concentrations of chlorophyll a occurred as a distinctive DCM at the metalimnion (Fig. 5A). Photosynthetic organisms congregating at the metalimnion are able to access more readily available nutrients in the hypolimnion (Descy et al., 2005; Leach et al., 2018). DCM are common in transparent lakes where nutrients are scarce (Saros et al., 2005; Camacho, 2006). The DCM observed in Yambo and Pandin thus reflects their relatively high transparency and low nutrient status. At Mohicap, where the photic zone was equal to the mixed zone, phytoplankton appear positioned to maximise nutrient access, but the limited penetration of light to the bottom of the photic zone of this relatively turbid lake restricts development of a more pronounced DCM (Fig. 5B). Where the mixed layer extends below the photic zone (Sampaloc, Bunot, Palakpakin), the maximum biomass of chlorophyll a is located towards the upper part of the water column, reflecting the greater nutrient availability in the upper waters of these lakes. Nutrient availability facilitates phytoplankton production in the epilimnion, and light limitation from this enhanced algal biomass alters the thermal energy and could shallow the mixing depth, pushing the zone of maximum production towards the surface of the water (Mazumder et al., 1990). This pattern has been noted in other eutrophic tropical lakes such as Lake Victoria (Haande et al., 2011). In the deeper and most nutrient-rich lakes (Sampaloc, Bunot), chlorophyll a peaks associated with the metalimnetic area are likely to be due to the redistribution of algal cells via water mixing, leading to some accumulation of biomass on top of the metalimnion.
Conceptual diagram proposing how a deep (> 10 m) tropical lake responds to increasing levels of aquaculture (represented by the number of fish). A A lake with weak thermal stratification, limited anoxia, reduced algal productivity and the absence of purple sulphur bacteria. B A lake experiencing more stable stratification, increased algal productivity, purple sulphur bacteria and increased nutrient loading. C A lake experiencing increasing eutrophication showing stable stratification, widening of the anoxic layer, abundant purple sulphur bacterial, greater algal productivity and a shallowing of the euphotic depth. D Hypereutrophic conditions resulted in further widening of the anoxic layer, substantial shading of phytoplankton and aphotic dead zones without purple sulphur bacteria and a smaller volume of water which is habitable for fish. The red bars represent the concentration of nutrients present in both the epilimnion and hypolimnion. The yellow triangle represents the light intensity down the water column
The DO profiles indicate that the hypolimnia were completely anoxic in May 2017. However, deep-water anoxia varies seasonally and amongst lakes (Bannister et al., 2019; Mendoza et al., 2019) (Figure SI1). In the oligotrophic lake, Yambo, deep waters became oxygenated during the cooler season in February (Mendoza-Pascual et al., 2021) most likely due to full mixing of the water column. In contrast, homogeneous or near homogeneous water mixing (as inferred by water temperature profiles) resulted in very low DO concentrations (c. 1 mg/l) throughout the water column in Mohicap, and the maintenance of deep-water anoxia in Sampaloc. Temperature and DO profiles collected in 1989–1990 by Santiago & Arcilla (1993) showed year long anoxic conditions in the hypolimnion suggesting the lack of deep-water mixing is a long-term occurrence in Sampaloc. This indicates that the biochemical oxygen demand of deep waters from these lakes exceeded the oxygen supplied by water circulation (Wilhelm & Adrian, 2007; Caliro et al., 2008). Oxygen demand in hypolimnia is fuelled by settling organic material and reduced substances from the sediment, each of which are exacerbated by eutrophication and aquaculture activities (Mesman et al., 2021). The inability to replenish deep waters with oxygen seasonally, despite isothermal mixing, is therefore likely linked to eutrophication. The low DO concentrations during turnover in Mohicap (c. 1 mg/l) in February coincided with fish kills (observed by local fisherfolk) (Bagarinao, 1998; Luther et al., 2004). A further consequence of water overturning is the fresh supply of nutrients from deep-water, which can encourage the development of algal blooms (Mesman et al., 2021). This is consistent with both fish kills and algal blooms having been observed in other tropical lakes such as Lake Chivero following water turnover (Mhlanga et al., 2006; Yang et al., 2018). The elevated epilimnetic DO concentrations in Sampaloc in February 2017 may reflect elevated phytoplankton blooms caused by water turnover.
Measured oxygen profiles in the water column thus appear to be driven by a combination of lake water mixing and phytoplankton productivity. Although deep waters of all lakes were anoxic, measured DO concentrations in surface waters exceeded 5 mg/l in all lakes, indicating habitable conditions for fish (LLDA, 2009). In the oligotrophic/mesotrophic lakes (Yambo, Pandin), the epilimnetic DO concentrations ranged between 5 and 8 mg/l, whilst the concentrations ranged between 7 and 15 mg/l in the eutrophic/hypereutrophic lakes. Greater oxygenated epilimnetic conditions in the eutrophic/hypereutrophic lakes are probably a combination of enhanced photosynthetic activity in the upper waters and shallower mixing zone depths, which distributes atmospheric oxygen within a smaller volume of water than in the deeper mixing and unproductive lakes (De Crop & Verschuren, 2019). In the case of Bunot, where the mixing depth is shallowest and algal biomass is concentrated towards the lake surface, this led to supersaturation of oxygen relative to the atmosphere (15 mg/l). One consequence of the distinctive vertical oxygen distribution is the creation of habitats within the lake that are suitable for anaerobic phototrophs such as Chromatiaceae (purple sulphur bacteria) (Fig. 5C). The lower peak in chlorophyll a registered by the sensors in Pandin could be indicating a peak in bacteriochlorophyll pigments, because the presence of okenone in the metaliminion indicates the presence of purple sulphur bacteria. Chromatiaceae require light and yet are located beneath the photic zone in Pandin, and is presumably additional evidence of their ability to adapt to low light intensities (Kushkevych et al., 2021). Although not detected in water samples from the other lakes, analyses revealed the presence of okenone from Chromatiaceae in surface sediment samples from Yambo, Pandin, Mohicap and Sampaloc, suggesting their presence previously and possibly at other times of the year.
Lakes with the highest aquaculture intensity (Bunot and Sampaloc) have the highest concentrations of P (TP and SRP), suggesting that aquaculture, in combination with increased inputs of P (as shown by an increase in PO43− concentrations; Table 1) from expanding human populations in the lake catchments, could be responsible for eutrophication (Schindler et al., 2016; Briddon et al., 2020; de Leon et al., 2020; May et al., 2021). Different lakes along the aquaculture gradient have distinct pigment assemblages; shallow Palakpakin supports the highest levels of cyanobacteria, the oligotrophic/mesotrophic lakes (Yambo, Pandin) have pigments from dinoflagellates and purple sulphur bacteria, and pigments from chlorophytes and diatoms are more common in the eutrophic/hypereutrophic deep lakes (Mohicap, Sampaloc, Bunot). In lakes with most intensive aquaculture, TP was abundant in dissolved and particulate forms in the bottom waters, whilst it was undetectable in the lake with the lowest intensity of aquaculture (Yambo). TP was the single variable that was significantly correlated with the algal pigments sampled down the water column in each lake. Constraining samples along this TP axis further separates samples from individual lakes within the biplot space, highlighting the strong vertical structuring of both TP and algal pigments down the water column. Taken together, these analyses highlight the important role of TP in influencing the composition of algal assemblages, and that algae and TP both vary vertically in the water column.
Excessive P loads may have exacerbated N limitation in these lakes. In larger lakes with deeper mixed layers, low light availability can constrain N2 fixation and exacerbate N limitation (Ferber et al., 2004). Similarly, light limitation through high algal biomass may also restrict N2 fixation (Maberly et al., 2020). However, light availability and so N2 fixation may increase when a lake has a shallow mixing layer (Mugidde et al., 2003), and consequently canthaxanthin and myxoxanthophyll, indicator pigments of potentially N2-fixing cyanobacteria, were found in the waters of Sampaloc and Bunot and were especially prevalent in Palakpakin. The abundant presence of cyanobacterial canthaxanthin pigments in sediments of all six lakes indicates that cyanobacteria are common in these sites. Most N in the lakes was in the form of NH4+ and usually present in the bottom waters. NH4+ in lakes often derives from the microbial decomposition of nitrogenous compounds in organic matter (ammonification), hence its abundant presence in the hypolimnetic waters of the eutrophic/hypereutrophic lakes. NH4+ is usually the preferred form of N for phytoplankton uptake (Glibert et al., 2016; Lachmann et al., 2019). The availability of hypolimnetic NH4+ deeper in the water column may therefore help to explain why phytoplankton grow close to the metalimnion in these N-limited systems when sufficient light was available. NH4+ concentrations in Sampaloc were also high in the surface waters (1929 μg/l), which could be a result of untreated sewage discharge from the city of San Pablo, leading to high levels of microbial decomposition in the epilimnion (Hampel et al., 2018). NH4+ is toxic to fish at concentrations > 2000 μg/l (Karasu Benli & Köksal, 2005), so its high concentration (1929 μg/l) in the epilimnion of Sampaloc has serious implications for aquaculture. Whilst NH4+ was undetectable in the surface waters of the other lakes, high hypolimnic levels of NH4+ limit the depth to which fish can live (Fig. 5D). In the oligotrophic lake (Yambo), low NH4+ concentrations (0 μg/l) in the hypolimnion are most likely due to the rapid oxidation of NH4+ into NO3− in the well-oxygenated and deep epilimnion.
Implications for lake management
Surveying the depth profiles of water column parameters raises a number of important issues relevant to the sustainability of aquaculture and management of tropical lakes more generally. First, the strong vertical structuring of phytoplankton, the base of aquatic primary productivity, is sensitive to changes in trophic status, demonstrating that phytoplankton growth can be light limiting due to high algal biomass in the mixing zone (Brothers et al., 2014) with evidence of possible “dead zones” due to anoxia (Burson et al., 2018; Conroy et al., 2011; Vadeboncoeur et al., 2021). This research provides evidence of a lack of a linear relationship between nutrients and algal production, supported by other studies that observed a similar relationship between TP and chlorophyll a concentrations in lakes worldwide due to light limitation, which has implications for lake management specifically regarding P management (Quinlan et al., 2021). Second, as lake temperatures are expected to increase in the coming decades (Mesman et al., 2021), water column stratification is likely to become more intense and prevalent (Kraemer et al., 2015). High temperatures in tropical lakes (compared to temperate lakes) mean they are especially prone to anoxic conditions in the hypolimnion, which could reduce the efficiency of TP management because of internal P loading (Lewis, 2000; Quinlan et al., 2021). High temperatures (> 25 °C) can override the lake-specific characteristics, such as morphology and the concentrations of other limiting nutrients, when attempting to control P concentrations (Matzinger et al., 2007; Trolle et al., 2011; Quinlan et al., 2021). This has led many tropical lakes (including the study lakes) to become N-limited. One possible mitigation strategy is to reduce N, rather than attempting a large reduction of P (McCauley et al., 1989). Therefore, mitigation strategies need to go beyond focussing purely on reducing nutrient loading from aquaculture and other activities, such as addressing contamination from domestic wastes, if water quality degradation is to be reversed and lakes, such as those focussed on in this study, are to be restored to ‘pre-eutrophic’ conditions.
Conclusion
Variations in measured variables in the San Pablo crater lakes indicate a strong vertical structuring of physical and chemical conditions and thermal stratification in all except Palakpakin, the shallowest and most mixed of the six lakes studied. The position of a lake along the gradient of increasing aquaculture intensity, which also equates to increasing trophic status, relates to the degree of overlap between the photic zone and mixing zone, with important consequences for the quantity and location of phototrophic production and anoxia. Increasing levels of aquaculture thus highlight a non-linear relationship between nutrient loading and algal production because of changes to nutrient and light availability, and to epilimnion DO concentrations. Continual water quality monitoring of tropical lakes can be useful in identifying changes in seasonal patterns and key drivers of environmental change, in order to develop effective, lake-specific mitigation strategies.
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Abell, J. M., D. Özkundakci, D. P. Hamilton & J. R. Jones, 2012. Latitudinal variation in nutrient stoichiometry and chlorophyll-nutrient relationships in lakes: a global study. Fundamental and Applied Limnology/archiv Für Hydrobiologie 181: 1–14.
Adrian, R., C. M. O’Reilly, H. Zagarese, S. B. Baines, D. O. Hessen, W. Keller, D. M. Livingstone, R. Sommaruga, D. Straile, E. Van Donk, G. A. Weyhenmeyer & M. Winder, 2009. Lakes as sentinels of climate change. Limnology and Oceanography 54: 2283–2297. https://doi.org/10.4319/lo.2009.54.6_part_2.2283.
Bagarinao, T., 1998. The sulfide tolerance of milkfish and tilapia in relation to fish kills in farms and natural waters in the Philippines. Hydrobiologia 382: 97–112.
Bannister, W., S. McGowan, A. C. Santos-Borja, J. Quak, L. S. Fong, M. Mendoza, R. D. S. Papa & D. Taylor, 2019. Potential anthropogenic regime shifts in three freshwater lakes in Tropical East Asia. Freshwater Biology 64: 708–722.
Bartosiewicz, M., A. Przytulska, B. N. Deshpande, D. Antoniades, A. Cortes, S. MacIntyre, M. F. Lehmann & I. Laurion, 2019. Effects of climate change and episodic heat events on cyanobacteria in a eutrophic polymictic lake. Science of the Total Environment 693: 133414.
Boehrer, B. & M. Schultze, 2008. Stratification of lakes. Reviews of Geophysics 46: 1–27. https://doi.org/10.1029/2006RG000210.
Briddon, C. L., S. McGowan, S. E. Metcalfe, V. Panizzo, J. Lacey, S. Engels, M. Leng, K. Mills, M. Shafiq & M. Idris, 2020. Diatoms in a sediment core from a flood pulse wetland in Malaysia record strong responses to human impacts and hydro-climate over the past 150 years. Geo Geography and Environment 7: e00090.
Brillo, B. B. C., 2015a. Development issues regarding Bunot Lake: the Lesser Lake among the seven lakes of San Pablo City, Philippines. Lakes and Reservoirs: Research and Management 20: 155–165. https://doi.org/10.1111/lre.12096.
Brillo, B. B. C., 2015b. The status of Philippine lake studies: Scholarly deficit in social science and small-lake research. Asia-Pacific Social Science Review 15: 78–101.
Brillo, B. B. C., 2016a. Developing a small lake: the case of Palakpakin Lake, San Pablo City, Philippines. Water Resources 43: 611–620. https://doi.org/10.1134/S0097807816040035.
Brillo, B. B. C., 2016b. Developing Mohicap Lake, San Pablo City, Philippines. The Social Sciences 11: 283–290.
Brillo, B. B. C., 2016c. Urban lake governance and development in the Philippines: the case of Sampaloc Lake, San Pablo City. Taiwan Water Conservancy 64: 66–81.
Brillo, B. B. C., 2017. The politics of lake governance: sampaloc lake, pandin lake, and tadlac lake of the laguna de bay region, Philippines. Asia-Pacific Social Science Review 17: 66–79.
Brothers, S., J. Köhler, K. Attermeyer, H. P. Grossart, T. Mehner, N. Meyer, K. Scharnweber & S. Hilt, 2014. A feedback loop links brownification and anoxia in a temperate, shallow lake. Limnology and Oceanography 59: 1388–1398.
Bureau of Fisheries and Aquatic Resources, 2019. [available on internet at https://www.bfar.da.gov.ph/].
Burson, A., M. Stomp, E. Greenwell, J. Grosse & J. Huisman, 2018. Competition for nutrients and light: testing advances in resource competition with a natural phytoplankton community. Ecology 99: 1108–1118. https://doi.org/10.1002/ecy.2187.
Caliro, S., G. Chiodini, G. Izzo, C. Minopoli, A. Signorini, R. Avino & D. Granieri, 2008. Geochemical and biochemical evidence of lake overturn and fish kill at Lake Averno, Italy. Journal of Volcanology and Geothermal Research 178: 305–316. https://doi.org/10.1016/j.jvolgeores.2008.06.023.
Camacho, A., 2006. On the occurrence and ecological features of deep chlorophyll maxima (DCM) in Spanish stratified lakes. Limnetica 25: 453–478.
Chen, N., T. Bianchi, B. McKee & J. Bland, 2001. Historical trends of hypoxia on the Louisiana shelf: application of pigments as biomarkers. Organic Geochemistry 32: 543–561. https://doi.org/10.1016/S0146-6380(00)00194-7.
Cinco, T. A., R. G. de Guzman, A. M. D. Ortiz, R. J. P. Delfino, R. D. Lasco, F. D. Hilario, E. L. Juanillo, R. Barba & E. D. Ares, 2016. Observed trends and impacts of tropical cyclones in the Philippines. International Journal of Climatology 36: 4638–4650. https://doi.org/10.1002/joc.4659.
Conroy, J. D., L. Boegman, H. Zhang, W. J. Edwards & D. A. Culver, 2011. “Dead Zone” dynamics in Lake Erie: the importance of weather and sampling intensity for calculated hypolimnetic oxygen depletion rates. Aquatic Sciences 73: 289–304. https://doi.org/10.1007/s00027-010-0176-1.
Cordero, C. S. & S. F. Baldia, 2015. Temporal variation of phytoplankton community structure in Lake Mohicap, San Pablo City, Laguna, Philippines. International Journal of Pure & Applied Bioscience 3: 377–385.
De Crop, W. & D. Verschuren, 2019. Determining patterns of stratification and mixing in tropical crater lakes through intermittent water-column profiling: a case study in western Uganda. Journal of African Earth Sciences 153: 17–30. https://doi.org/10.1016/j.jafrearsci.2019.02.019.
de Leon, J. R., S. K. P. Guinto, D. T. Tordesillas & R. D. S. Papa, 2020. Depth and productivity as predictive parameters for the widespread invasion of Arctodiaptomus dorsalis (Marsh, 1907) (Copepoda: Calanoida) in tropical lakes in the Philippines. Journal of Crustacean Biology 40: 512–519. https://doi.org/10.1093/jcbiol/ruaa052.
Degefu, F., A. Herzig, F. Jirsa, F. Jirsa, F. Degefu & M. Schagerl, 2014. First limnological records of highly threatened tropical high-mountain crater lakes in Ethiopia. Tropical Conservation Science 7: 365–381. https://doi.org/10.1177/194008291400700302.
Department of Environment and Natural Resources Resources, 2021. Philippine government classification of water quality guidelines (Administrative Order 2021-19).
Descy, J. P., M. A. Hardy, S. Sténuite, S. Pirlot, B. Leporcq, I. Kimirei, B. Sekadende, S. R. Mwaitega & D. Sinyenza, 2005. Phytoplankton pigments and community composition in Lake Tanganyika. Freshwater Biology 50: 668–684. https://doi.org/10.1111/j.1365-2427.2005.01358.x.
Ehrenfels, B., M. Bartosiewicz, A. Mbonde, K. Baumann, C. Dinkel, J. Junker, T. Kamulali, I. Kimirei, D. Odermatt, F. Pomati, E. Sweke & B. Wehrli, 2020. Thermocline depth and euphotic zone thickness regulate the abundance of diazotrophic cyanobacteria in Lake Tanganyika. Biogeosciences Discussions 2020: 1–21. https://doi.org/10.5194/bg-2020-214.
Ferber, L. R., S. N. Levine, A. Lini & G. P. Livingston, 2004. Do cyanobacteria dominate in eutrophic lakes because they fix atmospheric nitrogen? Freshwater Biology 49: 690–708. https://doi.org/10.1111/j.1365-2427.2004.01218.x.
Fiedler, P. C., 2010. Comparison of objective descriptions of the thermocline. Limnology and Oceanography: Methods 8: 313–325. https://doi.org/10.4319/lom.2010.8.313.
Gao, Y., Y. Jia, G. Yu, N. He, L. Zhang & B. Zhu, 2019. Anthropogenic reactive nitrogen deposition and associated nutrient limitation effect on gross primary productivity in inland water of. Journal of Cleaner Production 208: 530–540. https://doi.org/10.1016/j.jclepro.2018.10.137.
Gardner, W. S., J. F. Cavaletto, H. A. Bootsma, P. J. Lavrentyevy & F. Troncone, 1998. Nitrogen cycling rates and light effects in tropical Lake Maracaibo, Venezuela. Limnology and Oceanography 43: 1814–1825.
Glibert, P. M., F. P. Wilkerson, R. C. Dugdale, J. A. Raven, C. L. Dupont, P. R. Leavitt, A. E. Parker, J. M. Burkholder & T. M. Kana, 2016. Pluses and minuses of ammonium and nitrate uptake and assimilation by phytoplankton and implications for productivity and community composition, with emphasis on nitrogen-enriched conditions. Limnology and Oceanography 61: 165–197. https://doi.org/10.1002/lno.10203.
Gorham, E. & F. M. Boyce, 1989. Influence of lake surface area and depth. Journal of Great Lakes Research 15: 233–245.
Gunkel, G. & C. Beulker, 2009. Limnology of the crater lake cuicocha, ecuador, a cold water tropical lake. International Review of Hydrobiology 94: 103–125. https://doi.org/10.1002/iroh.200811071.
Haande, S., T. Rohrlack, R. P. Semyalo, P. Brettum, B. Edvardsen, A. Lyche-Solheim, K. Sørensen & P. Larsson, 2011. Phytoplankton dynamics and cyanobacterial dominance in Murchison Bay of Lake Victoria (Uganda) in relation to environmental conditions. Limnologica 41: 20–29.
Hampel, J. J., M. J. McCarthy, W. S. Gardner, L. Zhang, H. Xu, G. Zhu & S. E. Newell, 2018. Nitrification and ammonium dynamics in Taihu Lake, China: seasonal competition for ammonium between nitrifiers and cyanobacteria. Biogeosciences 15: 733–748. https://doi.org/10.5194/bg-15-733-2018.
Harris, I., P. D. Jones, T. J. Osborn & D. H. Lister, 2014. Updated high-resolution grids of monthly climatic observations–the CRU TS3. 10 Dataset. International Journal of Climatology 34: 623–642.
Heiskanen, J. J., I. Mammarella, A. Ojala, V. Stepanenko, K. Erkkilä, H. Miettinen, H. Sandström, W. Eugster, M. Leppäranta & H. Järvinen, 2015. Effects of water clarity on lake stratification and lake-atmosphere heat exchange. Journal of Geophysical Research: Atmospheres 120: 7412–7428.
Heisler, J., P. M. Glibert, J. M. Burkholder, D. M. Anderson, W. Cochlan, W. C. Dennison, Q. Dortch, C. J. Gobler, C. A. Heil & E. Humphries, 2008. Eutrophication and harmful algal blooms: a scientific consensus. Harmful Algae 8: 3–13.
Holmer, M., 2002. Impacts of milkfish (Chanos chanos) aquaculture on carbon and nutrient fluxes in the Bolinao area, Philippines. Marine Pollution 44: 685–696.
Jeppesen, E., M. Meerhoff, K. Holmgren, I. González-Bergonzoni, F. Teixeira-de Mello, S. A. J. Declerck, L. De Meester, M. Søndergaard, T. L. Lauridsen & R. Bjerring, 2010. Impacts of climate warming on lake fish community structure and potential effects on ecosystem function. Hydrobiologia 646: 73–90.
Karasu Benli, A. Ç. & G. Köksal, 2005. The acute toxicity of ammonia on Tilapia (Oreochromis niloticus L.) larvae and fingerlings. Turkish Journal of Veterinary and Animal Sciences 29: 339–344.
Kling, G. W., 1988. Comparative transparency, depth of mixing, and stability of stratification in lakes of Cameroon, West Africa. Limnology and Oceanography 33: 27–40. https://doi.org/10.4319/lo.1988.33.1.0027.
Klug, J. L., D. C. Richardson, H. A. Ewing, B. R. Hargreaves, N. R. Samal, D. Vachon, D. C. Pierson, A. M. Lindsey, D. M. O’Donnell & S. W. Effler, 2012. Ecosystem effects of a tropical cyclone on a network of lakes in northeastern North America. Environmental Science & Technology 46: 11693–11701.
Kraemer, B. M., O. Anneville, S. Chandra, M. Dix, E. Kuusisto, D. M. Livingstone, A. Rimmer, S. G. Schladow, E. Silow & L. M. Sitoki, 2015. Morphometry and average temperature affect lake stratification responses to climate change. Geophysical Research Letters 42: 4981–4988. https://doi.org/10.1002/2015GL064097.
Kubota, H. & J. C. L. Chan, 2009. Interdecadal variability of tropical cyclone landfall in the Philippines from 1902 to 2005. Geophysical Research Letters 36: 2–5. https://doi.org/10.1029/2009GL038108.
Kushkevych, I., V. Bosáková, M. Vítězová & S. K. M. R. Rittmann, 2021. Anoxygenic photosynthesis in photolithotrophic sulfur bacteria and their role in detoxication of hydrogen sulfide. Antioxidants 10: 1–11. https://doi.org/10.3390/antiox10060829.
Lachmann, S. C., T. Mettler-Altmann, A. Wacker & E. Spijkerman, 2019. Nitrate or ammonium: influences of nitrogen source on the physiology of a green alga. Ecology and Evolution 9: 1070–1082. https://doi.org/10.1002/ece3.4790.
Lau, M. P., G. Valerio, M. Pilotti & M. Hupfer, 2020. Intermittent meromixis controls the trophic state of warming deep lakes. Scientific Reports 10: 1–16. https://doi.org/10.1038/s41598-020-69721-5.
Leach, T. H., B. E. Beisner, C. C. Carey, P. Pernica, K. C. Rose, Y. Huot, J. A. Brentrup, I. Domaizon, H. P. Grossart, B. W. Ibelings, S. Jacquet, P. T. Kelly, J. A. Rusak, J. D. Stockwell, D. Straile & P. Verburg, 2018. Patterns and drivers of deep chlorophyll maxima structure in 100 lakes: the relative importance of light and thermal stratification. Limnology and Oceanography 63: 628–646. https://doi.org/10.1002/lno.10656.
Legaspi, K., A. Y. A. Lau, P. Jordan, A. Mackay, S. Mcgowan, G. Mcglynn, S. Baldia, R. D. Papa & D. Taylor, 2015. Establishing the impacts of freshwater aquaculture in tropical Asia: the potential role of palaeolimnology. Geo: Geography and Environment 2: 148–163. https://doi.org/10.1002/geo2.13.
Legendre, P. & H. Birks, 2012. From classical to canonical ordination. Tracking Environmental Change Using Lake Sediments. https://doi.org/10.1007/978-94-007-2745-8_8.
Lewis, W. M., 1987. Tropical limnology. Annual Review of Ecology and Systematics 18: 159–184. https://doi.org/10.2307/1941343.
Lewis, W. M., Jr., 2000. Basis for the protection and management of tropical lakes. Lakes & Reservoirs: Research & Management 5: 35–48.
Lewis, W. M., Jr., 2010. Biogeochemistry of tropical lakes. Internationale Vereinigung Für Theoretische Und Angewandte Limnologie: Verhandlungen 30: 1595–1603.
LLDA, 2009. Water Quality Report of the Seven Crater Lakes 2006–2008, Laguna Lake Development Authority-Environmental Quality Management Division, Rizal, Philippines.
Longhi, M. L. & B. E. Beisner, 2009. Environmental factors controlling the vertical distribution of phytoplankton in lakes. Journal of Plankton Research 31: 1195–1207. https://doi.org/10.1093/plankt/fbp065.
Luther, G. W., S. Ma, R. Trouwborst, B. Glazer, M. Blickley, R. W. Scarborough & M. G. Mensinger, 2004. The roles of anoxia, H2S, and storm events in fish kills of dead-end canals of Delaware inland bays. Estuaries 27: 551–560. https://doi.org/10.1007/BF02803546.
Maberly, S. C., J. A. Pitt, P. S. Davies & L. Carvalho, 2020. Nitrogen and phosphorus limitation and the management of small productive lakes. Inland Waters 10: 159–172. https://doi.org/10.1080/20442041.2020.1714384.
Mackereth, J. & J. F. T. Heron, 1979. Water analysis: some revised methods for limnologists. The Quarterly Review of Biology 54: 203. https://doi.org/10.1086/411247.
Matzinger, A., M. Schmid, E. Veljanoska-Sarafiloska, S. Patceva, D. Guseska, B. Wagner, B. Müller, M. Sturm & A. Wüest, 2007. Eutrophication of ancient Lake Ohrid: global warming amplifies detrimental effects of increased nutrient inputs. Limnology and Oceanography 52: 338–353.
May, L., C. M. Aura, V. Becker, C. L. Briddon, L. R. Carvalho, A. J. Dobel, P. Jamwal, B. Kamphuis, M. M. Marinho & S. McGowan, 2021. Getting into hot water: water quality in tropical lakes in relation to their utilisation. IOP Conference Series: Earth and Environmental Science 789: 12021.
Mazumder, A., W. D. Taylor, D. J. McQueen & D. R. S. Lean, 1990. Effects of fish and plankton on lake temperature and mixing depth. Science 247: 312–315.
McCauley, E., J. A. Downing & S. Watson, 1989. Sigmoid relationships between nutrients and chlorophyll among lakes. Canadian Journal of Fisheries and Aquatic Sciences 46: 1171–1175.
McGowan, S., 2007. Pigments in sediments of aquatic environments. Encyclopedia of Quaternary Science 2013: 2062–2074.
Mei, W. & S. P. Xie, 2016. Intensification of landfalling typhoons over the northwest Pacific since the late 1970s. Nature Geoscience 9: 753–757.
Mendoza-Pascual, M. U., M. Itoh, J. I. Aguilar, K. S. A. R. Padilla, R. D. S. Papa & N. Okuda, 2021. Controlling factors of methane in tropical lakes of different depths. Journal of Geophysical Research: Biogeosciences 126: 1–15. https://doi.org/10.1029/2020JG005828.
Mendoza, M. U., J. C. A. Briones, M. Itoh, K. Sophia, A. R. Padilla, J. I. Aguilar, N. Okuda & R. D. Papa, 2019. Small Maar Lakes of Luzon Island, Philippines : their limnological status and implications on the management of tropical lakes—A review. Philippine Journal of Science 148: 565–578.
Mesman, J. P., J. A. A. Stelzer, V. Dakos, S. Goyette, I. D. Jones, J. Kasparian, D. F. McGinnis & B. W. Ibelings, 2021. The role of internal feedbacks in shifting deep lake mixing regimes under a warming climate. Freshwater Biology 66: 1021–1035. https://doi.org/10.1111/fwb.13704.
Mhlanga, L., J. Day, M. Chimbari, N. Siziba & G. Cronberg, 2006. Observations on limnological conditions associated with a fish kill of Oreochromis niloticus in Lake Chivero following collapse of an algal bloom. African Journal of Ecology 44: 199–208. https://doi.org/10.1111/j.1365-2028.2006.00625.x.
Migrino, L. M. S., 2017. Chronology of Events in the History of San Pablo (San Pablo).
Miklius, A., M. F. J. Flower, J. P. P. Huijsmans, S. B. Mukasa & P. Castillo, 1991. Geochemistry of Lavas from Taal Volcano, Southwestern Luzon, Philippines: evidence for multiple Magma supply systems and mantle source heterogeneity. Journal of Petrology 32: 593–627. https://doi.org/10.1093/petrology/32.3.593.
Mugidde, R., R. E. Hecky, L. L. Hendzel & W. D. Taylor, 2003. Pelagic nitrogen fixation in Lake Victoria (East Africa). Journal of Great Lakes Research 29: 76–88. https://doi.org/10.1016/S0380-1330(03)70540-1.
Obrador, B., P. A. Staehr & J. P. C. Christensen, 2014. Vertical patterns of metabolism in three contrasting stratified lakes. Limnology and Oceanography 59: 1228–1240. https://doi.org/10.4319/lo.2014.59.4.1228.
PhilAtlas., 2020. PhilAtlas. https://www.philatlas.com/luzon/r04a/laguna/san-pablo.html. Its population as determined by the 2020 Census was 285%2C348.
Quinlan, R., A. Filazzola, O. Mahdiyan, A. Shuvo, K. Blagrave, C. Ewins, L. Moslenko, D. K. Gray, C. M. O’Reilly & S. Sharma, 2021. Relationships of total phosphorus and chlorophyll in lakes worldwide. Limnology and Oceanography 66: 392–404. https://doi.org/10.1002/lno.11611.
Santiago, A. E. & R. P. Arcilla, 1993. Tilapia cage culture and the dissolved oxygen trends in Sampaloc Lake, the Philippines. Environmental Monitoring and Assessment 24: 243–255.
Saros, J. E., S. J. Interlandi, S. Doyle, T. J. Michel & C. E. Williamson, 2005. Are the deep chlorophyll maxima in alpine lakes primarily induced by nutrient availability, not UV avoidance? Arctic, Antarctic, and Alpine Research 37: 557–563. https://doi.org/10.1657/1523-0430(2005)037[0557:ATDCMI]2.0.CO;2.
Schindler, D. W., S. R. Carpenter, S. C. Chapra, R. E. Hecky & D. M. Orihel, 2016. Reducing phosphorus to curb lake eutrophication is a success. Environmental Science & Technology 50: 8923–8929.
Southgate, P. C. & J. S. Lucas, 2003. Reproduction, Life Cycles and Growth, Blackwell Publishing, Hoboken:
Staehr, P. A., J. P. A. Christensen, R. D. Batt & J. S. Read, 2012. Ecosystem metabolism in a stratified lake. Limnology and Oceanography 57: 1317–1330. https://doi.org/10.4319/lo.2012.57.5.1317.
Szekielda, K. H., E. Espiritu & N. Lagrosas, 2014. Eutrophication of Manila Region, Philippines. International Journal of Geology Earth & Environmental Sciences 4: 2277–2081.
Trolle, D., D. P. Hamilton, C. A. Pilditch, I. C. Duggan & E. Jeppesen, 2011. Predicting the effects of climate change on trophic status of three morphologically varying lakes: implications for lake restoration and management. Environmental Modelling & Software 26: 354–370. https://doi.org/10.1016/j.envsoft.2010.08.009.
Vadeboncoeur, Y., M. V. Moore, S. D. Stewart, S. Chandra, K. S. Atkins, J. S. Baron, K. Bouma-Gregson, S. Brothers, S. N. Francoeur, L. Genzoli, S. N. Higgins, S. Hilt, L. R. Katona, D. Kelly, I. A. Oleksy, T. Ozersky, M. E. Power, D. Roberts, A. P. Smits, O. Timoshkin, F. Tromboni, M. J. Vander-Zanden, E. A. Volkova, S. Waters, S. A. Wood & M. Yamamuro, 2021. Blue waters, green bottoms: benthic filamentous algal blooms are an emerging threat to clear lakes worldwide. BioScience 71: 1011–1027. https://doi.org/10.1093/biosci/biab049.
Vollenweider, R. & J. Kerekes, 1982. Eutrophication of Waters. Monitoring, Assessment and Control, Organization for Economic Co-Operation and Development (OECD), Paris:, 156.
Wilhelm, S. & R. Adrian, 2007. Impact of summer warming on the thermal characteristics of a polymictic lake and consequences for oxygen, nutrients and phytoplankton. Freshwater Biology 53: 226–237. https://doi.org/10.1111/j.1365-2427.2007.01887.x.
Yang, Z., P. Xu, D. Liu, J. Ma, D. Ji & Y. Cui, 2018. Hydrodynamic mechanisms underlying periodic algal blooms in the tributary bay of a subtropical reservoir. Ecological Engineering 120: 6–13. https://doi.org/10.1016/j.ecoleng.2018.05.003.
Zafaralla, A., 2010. Plankton biodiversity, water quality and environmental status of Los Baños’ Crocodile Lake and San Pablo City’s seven lakes (Lakes Sampaloc and Pandin) [Philippines].
Zhou, Q., N. Yang, Y. Li, B. Ren, X. Ding, H. Bian & X. Yao, 2020. Total concentrations and sources of heavy metal pollution in global river and lake water bodies from 1972 to 2017. Global Ecology and Conservation 22: e00925.
Acknowledgements
The authors express their gratitude to Chen Qinqin Christina, Joeline Lim and Letisha Fong for their assistance and support with sample collection during fieldwork in San Pablo, the Philippines. We would like to acknowledge the people of San Pablo, particularly the locals living by the lakes who assisted us with the fieldwork and Milette Mendoza-Pascual for collecting the water column stratification profiles from all lakes. This work was supported by a NERC ENVISION DTP studentship (award number: NE/L002604/1), the Norway Grants Call 2019 (RO-NO 2019)—Collaborative Research Projects, no. 28/2020 and was made possible by a 2017 Memorandum of Understanding between the University of Santo Tomas and the National University of Singapore. We are grateful to the reviewers for their valuable comments which helped to improve the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no competing interests to declare that are relevant to the content of the article.
Ethical approval
The authors confirm that this manuscript is original and has not been published or is currently under consideration for publication elsewhere. The author can also confirm that the manuscript has been read and approved by all named authors.
Additional information
Handling editor: Judit Padisák
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Briddon, C.L., Metcalfe, S., Taylor, D. et al. Changing water quality and thermocline depth along an aquaculture gradient in six tropical crater lakes. Hydrobiologia 850, 283–299 (2023). https://doi.org/10.1007/s10750-022-05065-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10750-022-05065-7