Abstract
Ponds are aquatic habitats defined by their small size. Although small they are found on every continent, they are disproportionately rich in aquatic biodiversity, benefit terrestrial wildlife and have important ecosystem function benefits. One of these benefits might be carbon sequestration, a possibility suggested by (1) their abundance, (2) the intensity of their biogeochemical activity. Whilst greenhouse gas fluxes from ponds have been monitored widely, quantifying the stocks of organic carbon buried in sediment is a gap in our knowledge. Here we summarise measures of organic carbon in pond sediments cores from a diverse range of lowland ponds in England. We estimate a general measure of 9.38 kg OC in a 1 m2 × 20 cm block of pond sediment and scale this up to an overall estimate for Great Britain of 2.63 million tons of OC in pond sediment, with 95% CI of 1.41 to 3.84 million tons. The relationship between sediment carbon and gas fluxes remains a significant unknown.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Ponds are the quintessential small water body, often defined by their small size relative to lakes. Exactly what the precise size threshold should be varies, for example < 2 ha in area (Williams et al., 2010) or 1 m2 to 5 ha (Céréghino et al., 2008), all the way up to 8 ha in the 1971 Ramsar Convention on Wetlands. Size-based definitions are difficult because pond area and depth are confounded with biogeochemical functions (Sondergard et al., 2005). Additionally many cultures have customary definitions of what constitutes a lake versus a pond, for example in Nepal where the categorisation of a site changes as glacial melt changes the area of a pond (Poudel, 2018). Ponds are a habitat found on every continent (Epele et al., 2022) as well as on remote islands and from rain forest and desert to the tops of glaciers. Ponds are biodiversity hotspots at the landscape scale, in rural and urban settings (for example Davies et al., 2008; Hill et al., 2016), temperate, tropical and polar (Martinez-Sanz et al., 2012; Allende & Mataloni, 2013; Jeffries et al., 2016), and lowland or upland landscapes (Hinden et al., 2005; Usio et al., 2017). Along with other ‘Small Natural Features’, (Hunter et al., 2017) such as field margins and rocky outcrops, ponds have significant ecological roles.
However, ponds’ essential small size makes them vulnerable to being unappreciated, undocumented, lacking legal protections and vulnerable to degradation and destruction (Calhoun et al., 2017; Hunter et al., 2017). They may suffer from active dislike, being seen as a source of disease (Jeffries et al., 2016) and are therefore filled or drained. They remain rather overlooked by researchers (Oertli et al., 2009; Hill et al., 2021), perhaps because of their familiarity and small size (Jeffries, 2012) or the belief that most are made by humans and therefore not so interesting (Downing, 2010), these biases now explicitly acknowledged not just in Europe but more widely, for example India (Manoj & Padhy, 2015) and the USA (Berg et al., 2016). Ponds are often missed from international and national nature conservation legislation (Hill et al., 2017; Oertli, 2018).
This oversight remains despite our growing awareness of the importance of ponds over the last twenty years. Ponds are increasingly recognized for providing a range of ecosystem services such as flood water retention, nutrient sequestration and pollinator feeding stations (Céréghino et al., 2014; Biggs et al., 2017; Riley et al., 2018). Ponds are biodiversity hotspots, their role extending beyond the aquatic to include benefits to pollinators, insectivorous birds, even terrestrial spiders (Avila et al., 2017; Vickruck et al., 2019; Lewis-Phillips et al., 2020). Ponds bring social, cultural and economic benefits such as amenity value, well-being and livelihoods (Bastien et al., 2012; Huq, 2017; Higgins et al., 2019) and significant elements within historical landscapes, for example in central Europe or the Amazon (Heckenberger et al., 2007; Frajer & Fiedor, 2018). Ponds are biogeochemical hotspots too, defined by their disproportionately high rates of geochemical cycling relative to their small size (McClain et al., 2003) or potential to trap catchment sediment that may otherwise enter water courses (Berg et al., 2016). The importance of ponds for their services and benefits to humanity is beginning to take centre stage in contemporary pond conservation strategies (Céréghino et al., 2014; Biggs et al., 2017; Hill et al., 2017).
Amongst their diverse biogeochemical functions the potential importance of ponds in the carbon cycle was highlighted by Downing et al. (2008) and Downing (2010). The heart of Downing et al.’s argument combined two key elements; (1) the increasing intensity of biogeochemical activity in water bodies the smaller they became (e.g., 8, Downing, 2010) and (2) the sheer number of ponds on planet Earth. In addition, Downing et al. (2008) provided data from pond and lake sediments showing high levels of organic carbon in sediments, with the claim “the world’s farm ponds alone may bury 4 times as much carbon (C) as the world’s oceans”. Downing (2010) combined evidence for the potential intensity of carbon cycling within ponds with the estimates of global pond numbers to suggest that ponds play an “unexpectedly major role” in the global carbon cycle. Downing’s striking suggestion prompted a great deal of the subsequent interest in the role of ponds.
Subsequent estimates of the number and overall area of small water bodies suggests that Downing over-estimated the number of ponds (Seekell & Pace, 2011; Seekell et al., 2013; Polishchuk et al., 2018). Small ponds and wetlands remain cryptic (Pitt et al., 2012), standard remote sensing, lidar and air images all tending to miss small ponds although ground truthing and local knowledge can reveal surprisingly large numbers (Pitt et al., 2012; Jeffries et al., 2016). Whilst the number of ponds remains uncertain, over the last decade there has been a considerable advance in quantifying greenhouse gas fluxes, primarily CO2 and CH4, between ponds and the atmosphere. There is increasing evidence that ponds represent an overlooked source of greenhouse gas emissions, notably CH4, from diverse habitats including artificial rural ponds in Australia (Grinham et al., 2018), temperate ponds in NE USA (Kifner et al., 2018) and urban ponds in Berlin (Ortega et al., 2019). Holgerson and Raymond’s (2016) synthesis of data from freshwaters suggested that small ponds may be a significant source of carbon to the atmosphere and studies of boreal and arctic ponds provide compelling evidence for their importance as sources of CO2 and CH4, (Abnizova et al., 2012; Wik et al., 2016; Kuhn et al., 2018), which is only likely to increase with climate change warming of these higher latitudes (Wik et al., 2016). Although much remains uncertain, for example our poor knowledge of emissions from dried out systems (Marcé et al., 2019), the evidence generally supports ponds’ role as a significant source to the atmosphere (Torgersen & Branco, 2008). Downing’s suggestion that ponds may be important is proving correct, especially as a source for methane, rather than an overall sink of organic carbon.
Whilst examples of green-house gas flux measures from ponds are increasing, the organic carbon stock currently stored in pond sediments and how fast this is buried, remains largely unknown, missing from carbon budgets and subsequently land use policy relating to climate change mitigation. Taylor et al. (2019) estimated carbon burial rates from sediments of lowland ponds in the north-east of England which suggested burial rates higher than other terrestrial habitats, although Gilbert et al. (2016), working on the same ponds showed very rapid switches from being net sinks to net source as the ponds dried out. These recent carbon flux and burial rates from ponds show that the role of ponds may vary greatly between particular sites and times.
Small ponds can take their place as part of what Cole et al. (2007) called the global carbon cycle’s plumbing; the freshwater ecosystems, from large rivers and lakes to small ponds and wetlands responsible for transporting significant amounts of carbon, for example in lotic flows or as gas fluxes. Reviewing our developing understanding of carbon in freshwaters Travnik et al. (2018) notes the progress from small-scale studies of individual systems, to a holistic global view of freshwaters as “collectors and reactors”, not just passive recipients of carbon, but as active transporters, sources and sinks. Understanding the distribution of carbon within and among differing pond types is crucial to accurately quantifying the total carbon stocks within pond sediments, for upscaling studies to regional, national, and global estimates, and their successful integration into carbon budgets. The potential role of small ponds as carbon mitigation sinks, or perhaps problematic sources, needs investigation.
Our purpose here is to bring together recent advances in our knowledge of temperate pond carbon, primarily focusing on stocks and burial rates, using data from typical lowland temperate ponds in England. We consider data allowing:
-
1.
Quantification of organic carbon (OC) from sediments of differing pond types, defined by being from markedly different land use, supporting distinctly different vegetation and some permanent versus temporary habitats in north-east England (Gilbert et al., 2021), along with some new data from three other regions of England.
-
2.
Quantification of OC burial rates in sediment from small ponds of precisely known age, in north-east England (Taylor et al., 2019).
-
3.
Comparison to soil OC stocks and burial rates across broad terrestrial habitats types in the UK collated specifically to promote nature-based solutions to help mitigate climate change (Gregg et al., 2021; Rewilding Britain, 2021; Stafford et al., 2021).
The sediment carbon stock data are used to provide an estimate of overall organic carbon stocks in pond sediments in the Great Britain.
Methodology
Our review draws upon two, linked studies of lowland ponds in England; quantification of organic carbon stocks in ponds sediment (Gilbert et al., 2021) and organic carbon burial rates (Taylor et al., 2019). Both of the original papers provide much more detail on the sampling design, practice and analyses. Here we provide a brief summary of the key methodological strategies and methods, along with detail for additional sites not included in these previous studies.
Pond sediment carbon stocks
Sample sites and biogeographical regions
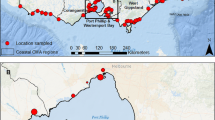
Organic carbon in pond sediments was measured in forty lowland ponds in Northumberland, north-east England, along with five ponds from each of three other biogeographically distinct regions of England: Askham Bog near York (lowland peat bog, northern England), Thompson Common Breckland pingo ponds (post glacial Breckland, east Anglia) and The Lizard Peninsular (lowland heathland, south west England); see Fig. 1 for locations. These three regions were chosen because their biogeography and climates are distinctly different to lowland Northumberland and to one another. All four are lowland regions, with elevations around the sample ponds of < 100 m (Northumberland), < 30 m (Askham), < 50 m (Thompson Common) and < 80 m (Lizard). All four are dominated by agricultural land use, land classified as Grade 2–4 agricultural comprising 64–84% of their areas. Each of the four fall within distinct National Character Areas, (NCAs) which are biogeographically coherent areas of landscape defined by topography, land use and habitats (Natural England n.d.). The precise NCAs are No13 South Northumberland Coastal Plain, No28 Vale of York, No85 The Brecks and No157 The Lizard, the first three characterised as low-lying, whilst the Lizard’s cliff top locality belies the low overall altitude. We acknowledge that the focus on lowland ponds, however diverse in their biogeography, is an important constraint on our data.
Gilbert et al. (2021) provides detail on the sampling and analytical protocols used, as well tests of variation within and between ponds for the Northumberland sites. Our analyses of carbon stocks uses the carbon density, mg C cm3, although we also show data for the carbon as % of sediment dry weight for comparison.
Here we briefly outline the sampling and analysis of the Northumberland pond types and give an overview of the Askham Bog, Thompson Common and Lizard Peninsula sites, these last three being sources of the new data.
Northumberland, Druridge Bay
Druridge Bay is in south-east Northumberland, a cool and dry part of England. The ponds were all in the lowland coastal plain, some in nature reserves other on farmland. We intentionally sampled ponds from four distinct land use types, defined by the surrounding landscape and management: (1) ponds in arable fields, with no buffer between the crops and the pond, and most sites ploughed every year, (2) ponds in livestock pasture fields, again lacking any buffer and accessible to the cattle and sheep, (3) ponds in sand dune slacks, with typical dune slack vegetation often some slight brackish water influence and (4) ponds embedded within more extensive, natural wetlands which provided a buffer between the pond and other land uses and not managed. These four pond types have distinctly different plant communities (Jeffries, 2012) and we hypothesised that they would have significantly different carbon stocks.
The other English regions
Askam Bog
15,000 year old remnant lowland fen in Yorkshire, now a Yorkshire Wildlife Trust Nature reserve and Site of Special Scientific Interest (Fitter et al., 1980). The region is cool and wet compared to the others. The samples were taken from five ponds in the peat bog, including natural pools and some created by historical peat excavation that occurred between Roman times and the seventeenth century.
Thompson Common
The Common is within the Brecks of Norfolk, on a Norfolk Wildlife Trust Reserve and Site of Special Scientific Interest. The region is heavily influenced by continental air masses (Hallett et al., 2004), hotter in summer but colder in winter, and drier than the other regions. The Common is a mosaic of woodland, meadow and wetland, noted for pingo ponds created by the retreat of the Devensian ice sheet approximately 11,000 years ago (Foster, 1993; Walmsley, 2008; Clay, 2015). We cored five pingo ponds.
Lizard Peninsula
The Lizard National Nature Reserve is managed by multiple conservation organisations. The site is famed for its unusual Serpentine geology and unusual temporary ponds, many associated with trackways and others that have been classified as Mediterranean Temporary Ponds. The climate is milder but wetter than the other regions. The ponds support nationally rare flora and fauna (Bilton et al., 2009) some associated with very small trackway pools (Scott et al., 2012). Again, five ponds were cored, from within heathland and grassland habitats.
Sediment coring and quantifying carbon density
Pond sediment in all four regions was sampled using a vanadium steel corer, pushed into the sediment in the wetted area of the sample ponds. The corer was driven manually, typically reaching the more compacted soil base that acts as a plug to seal in the softer sediment layers above. Upon removal of the corer excess water was drained via a small hole at the top, and the length of the core was measured via the internal plunger, allowing for calculation of compaction during the removal of the sediment core. The corer length was 50 cm. Core depths in Northumberland varied between 9.2 to 33 cm, 12.5 cm to 36 cm at Askham, 19 cm to 34.5 cm at Thompson Common and 13 cm to 26 cm at The Lizard. The sediment core was extruded and cut into 1 cm lengths at a time, measured by 1 cm markings along the length of the internal plunger. Slicing the core in this manner was found to be more accurate than extruding the core intact and dissecting in the lab. Upon dissection each section was wrapped in foil and placed in a paper sample bag and transported back to Northumbria University, Newcastle upon Tyne, and stored in refrigeration prior to analysis.
All sediment cores were collected between April and December 2014, and while many ponds dried during summer months, all ponds had standing water at the time of sampling. Sediment cores were collected from the centre of each pond, or as close to the centre as possible where water level was above the height of waders. In some cases the samples were in amongst the vegetation whilst others were from open water.
Within 24 h of coring, individual samples were weighed to acquire the wet weight of each section. Samples were then dried, dry bulk density calculated, the samples ground and analysis of total carbon was performed by dry combustion using Total Elemental Analysis (TEA), specifically a Thermo Scientific FLASH 2000 Series Organic Elemental Analyser.
For comparison between ponds we took one core from each pond: see Gilbert et al. (2021) for further details on field sampling, sample preparation, laboratory analysis and analysis of intra and inter pond variation. Organic carbon is presented as C density in mg C cm−3. The Northumberland ponds could be characterized either by surrounding land use (arable, pasture, naturalistic wetland or sand dune), drying regime (never dry, sometimes dry, dry every year, based on twenty years of working on the sites) or plant communities defined by TWINSPAN classification (Gilbert et al., 2021). Here we analysed the Northumberland data-based dividing the ponds into four groups based on the surrounding land use categories. The Askham Bog, Thompson Common and Lizard ponds were not distributed between different land uses, nor do we have plant community types or drying regime data for these sites so each were treated as single sets. The four Northumberland and three other regions therefore gave us seven sets of sediment carnbon data to compare.
Organic carbon burial rates
Organic carbon burial rates were calculated from samples of very small (1 m2) ponds at Druridge Bay, south-east Northumberland. Full methodological details of sampling protocol and quantification are given in are given in Taylor et al. (2019).
We used 12 ponds of precisely known age, excavated in November of 1994 and either 18 or 20 years old (i.e., sampled in 2012 or 2014) when we sampled them. The ponds had been created in a field with a clay rich soil, which resulted in a very distinct demarcation between the original bottom of the pond and any organic rich sediment that had accumulated since they had been dug. Ponds were chosen to include a variety of plant communities that had developed over time.
Three of the ponds had their substrate entirely excavated by digging a trench alongside the pond then taking out blocks of sediment by working in from the side. The remaining ponds were cored using the same coring method as for the carbon sediment survey, and the same laboratory methods. Because we knew the precise age of the ponds the density of carbon in the sediment could then be translated to a burial rate by dividing the density by the age of the pond when sampled. Note that additional sampling of newly constructed ponds in the same field showed that very little carbon accumulated in the first three years so rate estimates could be adjusted to allow for this lag.
Burial rates are expressed as g OC m−2 year−1.
Data analyses
Statistical analyses
We analysed the carbon stocks, mg C cm−3, to characterise differences amongst the seven pond types.
Differences were tested using generalized linear mixed models, (GLMM). Natural log transformations were applied to the carbon stock data to meet requirements of normality and homogeneity of variance. Pond type was incorporated as a fixed factor in the GLMM.
The depth of each slice in a core was included as a covariate, with a repeat measures structure linking individual slice data down the length of the core. Carbon stocks had not shown a significant relationship to depth when tested with regression as part of data exploration (adjusted R2 − 0.001, P = 0.6), but we retained depth as a covariable in the GLMM in case the more complex model structure revealed a pattern. Individual ponds were treated as random factors, allowing both slope and intercept to vary between ponds in the models. Pond water depth was not included in models, partly because all samples were from ponds shallow enough to wade into. Analysis of intra-pond variation across the water depth profiles of test ponds had shown only limited variation with depth (Gilbert et al., 2021). Models were tested by adding in these elements consecutively and comparing models using changes to AIC. Post-hoc pairwise comparisons between individual pond types were tested using Bonferroni comparisons. The relationship between carbon stocks and pond depth was characterised using linear least squares regression, using all the data in one global test for all ponds. All analyses were carried out using SPSS 22.
Results
Sediment carbon stocks
Organic carbon measured as carbon density, mg C cm−3, varied between the seven sets of ponds, ranging from a mean of 20.7 mg C cm−3 in Lizard ponds up to 74.4 mg C cm−3 at Thompson Common.
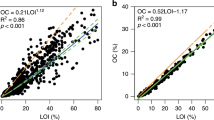
There was considerable variation in carbon density between some pond types, notably the Northumberland dune, Askham Bog and Thompson Common ponds containing significantly higher stocks per cm3. The quantity of carbon in the pond sediments is shown in Fig. 2, both as density (mg C cm−3) and also as %. Data for the Northumberland ponds are shown for each of the four land use types (arable, pasture, dune and wetland) separately and for each of the other three regions, Askham, Thompson Common and Lizard, making seven categories.
Pond sediment core carbon measures from the seven sets of samples; four from Northumberland, differentiated by land use (arable, sand dune, natural wetlands, pasture) and other three others from Askham Bog (Yorkshire), Lizard Peninsular (Devon) and Thompson Common (Norfolk). a Carbon measurements expressed as mg OC cm−3, b carbon measurement expressed as % of sediment
When measured as a % of the sediment the carbon varied markedly between some pond types, from a mean of 2.9% for arable field ponds up to 45.6% for the Askham ponds. Variation when using % carbon as a measure is because the measure does not account for sediment density. The sediment in the arable field ponds is dense agricultural soil so that even a small % is a more substantial absolute amount, whilst in the Askham ponds the sediment is looser, wet peat so that the % of carbon does not represent such a large absolute amount. We believe that the carbon density is a much more useful measure for estimating carbon stocks and the potential role of ponds for carbon capture and burial.
Unsurprisingly there remains some variation between ponds in each site or land use category. Carbon density was generally very variable with depth although the core profiles from the Lizard and Thompson Common generally show a decrease with depth. The lack of a significant relationship between carbon stock and depth surprised us so, despite this, we also quantified the stocks for depths down to 10 cm, 10–20 cm and 20 cm + for the four regions. Stocks did decline down the sediment core for Northumberland ponds (means 43.6, 39.9 and 29.5 mg C cm−3, respectively), barely for Askham or Thompson Common (means 68.7, 67.4 and 52.8 mg C cm−3 and 77.7, 77.7, 36.6 mg C cm−3, respectively, but note that for 20 cm + data were limited to 12 or five samples), whilst at the Lizard the shallow sediments showed a marked decline (25.1, 5.7 mg C cm−3, no sample > 20 cm).
We had anticipated very marked differences between the seven sets of ponds, in the case of the Northumberland land uses because ponds in the different landscapes supported very different animal and plant communities, and in the case of the other three regions because of the very different climate, biogeography and history of the sites. The GLMM outcomes did show significant differences between some of the seven groups (Table 1): the Lizard ponds held significantly lower carbon stocks than the Northumberland pasture, Northumberland dune, Askham and Norfolk sites. Askham and Norfolk carbon stocks were significantly higher than the Northumberland natural, Northumberland arable and Lizard sites. However, none of the four Northumberland pond types defined by land use differed significantly from one another and all seven groups showed some wide variations within groups, resulting in considerable overlap.
Quantifying carbon stored in a standard volume of temperate pond sediment
We were surprised that the carbon stocks from each pond type did not differ more consistently and strongly given the very different regions and land uses. Instead the extent of the overlap between ponds in the different types suggested the opportunity to combine the data from all 55 ponds to create a overall carbon stock for a volume of temperate pond sediment.
We used a volume of 100 cm2 and 20 cm deep; we chose this depth as a typical depth for pond sediments in our survey. Sediment depth in ponds is seldom reported but published figures suggest 20 cm is a useful threshold, for example mean ponds sediment depths: 11 cm (Nicolet et al., 2004; DeClerck et al., 2006; Tsai et al., 2011). We treated the carbon stock as the same throughout this depth because neither the GLMM or the exploratory regression showed a relationship with depth, essentially the high C% in upper levels of sediment tends to get evened out as bulk density increases with depth although this is a simplification because some pond types, Askham and the Lizard, suggest a trend of decreasing stocks with depth. Combining the carbon stock data from all 55 ponds gives a mean density and standard deviation of 46.9 mg cm−3 ± 28.24 mg cm−3 applicable down to 20 cm. Note that the large number of samples (the slices from each core, n = 931) results in a small 95% CI range ± 1.81, but this should not obscure the wide range with individual samples from a minimum of 1.13 to a maximum of 201.8 mg cm−3.
Therefore a block of sediment measuring 100 cm wide by 100 cm long by 20 cm deep has a volume of 200,000 cm3 and a mean total carbon stock of = 46.9 × 200,000 = 9,380,000 mg or 9.38 kg, (95% CI 9.01–9.74 kg).
An estimate of overall carbon stocks in ponds in Great Britain
To calculate the total carbon stock in British ponds we multiplied up our standard 9.38 kg C for a 1 m−2 by 20 cm deep sediment block by an estimate of the total area of pond habitat in Great Britain. Carbon data for habitats is commonly scaled up to ha−1, so our data give a mean of 94 tonnes C ha−1.
To estimate the overall area of ponds in Great Britain we used data from the Countryside Survey (Williams et al., 2010). The Countryside Survey is a survey a carried out on behalf of central government of land use in England, Scotland and Wales based on systematic field surveys of 591 representative 1 × 1 km squares across these countries, carried out every few years between 1978 and 2007. For the 2007 survey the pond work was done by the Centre for Ecology and Hydrology, with the their surveyors trained by Pond Conservation, now the Freshwater Habitats Trust, the UK’s lead organisation for pond research and conservation, ponds being defined by the FHT size criterion body of standing water between 25 m2 and 2 ha in area which usually holds water for at least four months of the year. The survey data estimated pond numbers in four size categories; 200–400, 400–2000, 2000–10,000 and 10,000–20,000 m−2. The median area of ponds in the four size ranges differentiated in the Countryside Survey are 140, 800, 3000 and 14,550 m−2, respectively. We estimated the total area of pond habitat in Great Britain by multiplying the estimated numbers of ponds in each size category by their respective median areas. Table 2 shows the results including for the 95% CI boundaries of pond numbers. To estimate total pond sediment carbon stocks in Great Britain we multiplied our global estimate of 9.38 kg OC in a 1 m2 × 20 cm deep block of sediment by the overall estimate of pond area from the Countryside Survey data (Table 2). This gives an estimate of organic carbon in pond sediments in Great Britain as 2.63 million tonnes, with 95% CI of 1.8 and 3.7 million tonnes. To extend the estimation of variation further we have combined the high and low 95% CI for carbon density with the high and low estimates of pond numbers, which gives a range of 1.4 up to 3.8 million tonnes (Table 2).
For comparison to the pond data presented here Table 3 gives data for carbon stocks in the soils and sediments of UK habitats from the recent review by Natural England (Gregg et al., 2021). The habitats are broad types such as woodland, scrub or main grassland types, those presented here chosen because soil depth data were given and depths were comparable to the depths of our pond cores.
Organic carbon burial rates
The results for OC burial rates (Taylor et al., 2019) from the small ponds gave a mean of 142 ± 19 g OC m−2 year−1, the equivalent of 1.42 t OC ha−1 year−1, (95% CI 1.39 to 1.45 t OC ha−1 year−1) with minima and maxima of 79–247 g OC m−2 year−1. Taylor et al. (2019) compared these rates to those given by Downing in his original discussion of carbon in ponds; they are much higher than Downing’s figures for boreal and temperate forest or grasslands, the ponds burying OC sixty to thirty times faster. However, more recent data for other habitats such as grassland, woodland and bogs (Gregg, 2021) show much more overlap with our burial rate estimates for ponds although our pond rates are higher than woodland and lakes (Table 3).
Discussion
The estimates of sediment carbon stocks from our data are the first detailed survey of a range of lowland, temperate, ponds. The data set is small, and the extrapolations up to Great Britain national level using Countryside Survey data must be treated with caution but the results give a first approximation figure where previously none existed; 2.625 million metric tons of organic carbon in ponds in Great Britain. The data suggest that, square metre for square metre, the sediment of small ponds holds more carbon, and buries additional OC, at least as rapidly as many other terrestrial habitats such as woodland and grassland. Our estimate is a first approximation: despite having purposefully chosen biogeographically varied localities to sample beyond the main Northumberland site there are many more pond types throughout Great Britain. Combining of data from all the ponds into a single measure of sediment carbon to apply generally may be untenable as more data are obtained. In particular, no upland ponds were included in our sampling.
Note also that carbon expressed as density, in this study as OC mg cm−3, gives a much more insightful measure than simply using the % C in the sediments. Density measurements adjust for the overall density of the sediment and soil. In our study the use of % OC measurement results in apparently much lower carbon in the arable field soils and much higher in the Askham Bog site. However, once data are expressed as density these apparent differences are markedly reduced. The relationship between depth and carbon stocks also remains uncertain. Taking our data altogether there was no significant relationship between carbon stocks and depth, although comparisons across the three broad depth ranges of < 10 cm 10–20 and 20 + across the four regions did show some decrease at the lowest depths. Taylor et al. (2019) demonstrated limited carbon accumulation in the first 1–3 years of the lives of newly dug ponds. Taking these two pieces of evidence together we believe that the burial rate and carbon-depth relationship requires further data, not least if ponds are created with the purpose of carbon sequestration.
Nature-based solutions to help mitigate climate change increasingly include habitat creation (Gregg et al., 2021; Stafford et al., 2021; Rewilding Britain, 2021), primarily tree planting and peatland restoration. In many places, ponds may be just as good an intervention. They are relatively easy to create (many of the alleged constraints are myths, debunked in recent years, Biggs et al., 1994), ponds can be fitted in amongst diverse land uses, they begin burying carbon rapidly within a year or two of creation (Taylor et al., 2019) and bring a wealth of other biodiversity benefits (Céréghino et al., 2014).
Ponds may occupy only a very small proportion of most temperate landscapes but have a disproportionately important role due to their high level of biogeochemical activity. For example, in Great Britain the Countryside Survey data suggests that ponds occupy roughly only 0.0012% of the land area compared to 6.0% for broadleaved woodland but woodland only buries about double the amount of OC per year compared to the ponds (Taylor et al., 2019). Whilst our figures are based on limited data, both for ponds and often the other habitats, the potential of ponds to be helpful nature-based solutions to mitigate climate change are apparent and ponds have recently taken their rightful place in reports disseminating contemporary knowledge on the importance of varied habitat types in the UK (Gregg et al., 2021; Rewilding Britain, 2021; Stafford et al., 2021).
The estimates for carbon in pond sediments broadly overlap those of other UK terrestrial habitats (Table 3) but the higher end of our estimated range exceeds all habitats other than bogs and swamps. However, we do not have enough evidence to be confident about what drivers create a particularly carbon rich pond sediment. There are a few studies that identify the possible importance of the precise plant species, overall diversity and functional types as significant factors (Mo et al., 2015; Sun et al., 2019; Taylor et al., 2019) but these drivers need much more research.
However, the benefits of this ecosystem service need to be seen in the context of ponds as a potentially significantly source of green-house gases. There is very good evidence, from diverse pond systems around the world, that ponds can be significant sources of greenhouse gases, in particular methane. Examples of significant methane emissions include artificial agricultural ponds in Queensland, Australia (Grinham et al., 2018), woodland vernal ponds in north-east USA (Kifner et al., 2018), urban ponds in Berlin and Sweden (Ortega et al., 2019; Peacock et al., 2019) and arctic thaw ponds (Abnizova et al., 2012; Kuhn et al., 2018; Burke et al., 2019) although Polishchuk et al. (2018) argue that the small overall area of very small thaw ponds limits their likely importance. Emissions of CO2 may also be significant, especially for temporary ponds when they dry (Obrador et al., 2018; Marcé et al., 2019). Greenhouse gas emissions are likely to increase as temperate ponds warm (Yvon-Durocher et al., 2017). The precise drivers of greenhouse gas emissions remain uncertain. In urban ponds allochthonous carbon and high nutrients seem likely to increase emissions (Van Bergen et al., 2019) whereas in German kettle holes the interplay of nutrients and precise length of drying phase drives variation (Reverey et al., 2016) whilst the potential role of plants remains unclear (Mo et al., 2015). Overall though, small water bodies are increasingly spotlighted as significant sources of both CH4 and CO2, for example Holgerson and Raymond (2016) and Peacock et al. (2021).
As we begin to better quantify carbon stocks, burial rates and greenhouse gas fluxes, two important gaps in our knowledge are now clear: (1) the drivers of carbon dynamics in ponds, in particular the role of productivity and of plants, and (2) the lack of studies linking sediment stocks, burial rates and gas fluxes. Given the contrasting messages of ponds as important carbon sinks based on sediment carbon measures but also significant greenhouse gas emitters in flux studies these challenges need attention if we are to maximise the use of ponds for carbon sequestration and storage.
Whilst the precise detail of ponds’ role in the carbon cycle remains patchily understood, the role of ponds in the carbon cycle has a longer history than our recent studies. An 1887 painting by the pre-Raphealite artist Ford Madox Brown in Manchester’s town hall shows the chemist John Dalton collecting marsh gas from a small pond: the painting is so exact that the plants are identifiable, for example Alisma plantago–aquatica, amongst Lemna and Carex species. Dalton was collecting “carbureted” gases (that is methane) from lakes and ponds in the early nineteenth century as part of his ground-breaking work into atomic theory to explain the reality of atoms and how they form compounds. Ponds may be small, and still sometimes find themselves lumped in with lakes in global reviews of carbon (for example Rosentreter et al., 2021) but their significance is now well-established. We need to better understand the links between carbon dynamics in ponds, across diverse landscapes and climates, and scale up the limited, local, studies into more generally applicable lessons at a continental scale. Whether ponds are a source or sink of green-house gases, these small water bodies are now centre stage in research.
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Abnizova, A., J. Siemens, M. Langer & J. Boike, 2012. Small ponds with major impact: the relevance of ponds and lakes in permafrost landscapes to carbon dioxide emissions. Global Biogeochemcial Cycles. https://doi.org/10.1029/2011GB004237.
Allende, L. & G. Mataloni, 2013. Short-term analysis of the phytoplankton structure and dynamics in two ponds with distinct trophic states from Cierva Point (maritime Antarctica). Polar Biology 36: 629–644.
Avila, A. C., C. Stenert, E. N. L. Rodrigues & L. Maltchik, 2017. Habitat structure determines spider diversity in highland ponds. Ecological Research 32: 359–367.
Bastien, N. R. P., S. Arthur & M. J. McLoughlin, 2012. Valuing amenity: public perceptions of sustainable drainage systems ponds. Water and Environment Journal 26: 19–29.
Berg, M. D., S. C. Popescu, B. P. Wilcox, J. P. Angerer, E. C. Rhodes, J. McAlister & W. E. Fox, 2016. Small farm ponds: overlooked features with important impacts on watershed sediment transport. Journal of the American Water Resources Association 52: 67–76.
Biggs, J., A. Corfield, D. Walker, P. Whitfield & P. Williams, 1994. New approaches to the Management of ponds. British Wildlife 5: 273–287.
Biggs, J., S. von Fumetti & M. Kelly-Quinn, 2017. The importance of small water bodies for biodiversity and ecosystems services: implications for policy makers. Hydrobiologia 793: 3–39.
Bilton, D. T., L. C. McAbendroth, P. Nicolet, A. Bedford, S. D. Rundle, A. Foggo & P. M. Ramsay, 2009. Ecology and conservation status of temporary and fluctuating ponds in two areas of southern England. Aquatic Conservation: Marine and Freshwater Ecosystems 19: 134–146.
Burke, S. A., M. Wik, A. Lang, A. R. Contosta, M. Place, P. M. Crill & R. K. Varner, 2019. Long-term measurements of methane ebullition from thaw ponds. JGR Biogeosciences 124: 2208–2221.
Calhoun, A. J. K., D. M. Mushet, K. P. Bell, D. Boix, J. A. Fitzsimons & F. Isselin-Nondedeu, 2017. Temporary wetlands: challenges and solutions to conserving a ‘disappearing’ ecosystem. Biological Conservation 211: 3–11.
Céréghino, R., J. Biggs, B. Oertli & S. Declerck, 2008. The ecology of European ponds: defining the characteristics of neglected freshwater habitat. Hydrobiologia 597: 1–6.
Céréghino, R., D. Boix, H.-M. Cauchie, K. Martens & B. Oertli, 2014. The ecological role of ponds a changing world. Hydrobiologia 723: 1–6.
Clay, P., 2015. The origin of relic cryogenic mounds at East Walton and Thompson Common, Norfolk, England. Proceedings of the Geologists’ Association 126: 522–535.
Cole, J. J., Y. T. Prairie, N. F. Caraco, W. H. McDowell, L. J. Travnik, R. G. Streigl, C. M. Duarte, P. Kortelainen, J. A. Downing, J. J. Middelburg & J. Melack, 2007. Plumbing the global carbon cycle: integrating inland waters into the terrestrial carbon budget. Ecosystems 10: 171–184.
Davies, B. R., J. Biggs, P. Williams, M. Whitfield, P. Nicolet, D. Sear, S. Bray & S. Maund, 2008. Comparative biodiversity of aquatic habitats in the European agricultural landscape. Agriculture, Ecosystems & Environment 125: 1–8.
DeClerck, S., T. De Bie, D. Ercken, H. Hampel, S. Schrijvers, J. Van Wichelen, V. Gillard, R. Mandiki, B. Loson, D. Bauwens, S. Keijers, W. Vyerman, B. Goddereris, L. De meester, L. Brendonck & K. Martens, 2006. Ecological characteristics of small farm ponds: associations with land use practices at multiple spatial scales. Biological Conservation 131: 523–532.
Downing, J. A., 2010. Emerging global role of small lakes and ponds: little things mean a lot. Limnetica 29: 9–24.
Downing, J. A., J. J. Cole, J. J. Middleburg, R. G. Striegl, C. M. Duarte, P. Kotelainene, Y. T. Praire & K. A. Laaube, 2008. Sediment organic carbon burial in agriculturally eutrophic impoundments over the last century. Global Biogeochemical Cycles 22: 1–10.
Epele, L. B., M. G. Grech, E. A. Williams-Subiza, C. Stenert, K. McLean, H. S. Greig, L. Maltchik, M. M. Pires, M. S. Birds, A. Boissezon, D. Boix, E. Demiere, P. E. García, S. Gascón, M. J. Jeffries, J. M. Kneitel, O. Loskutova, L. M. Manzo, G. Mataloni, M. C. Mlambo, B. Oertli, J. Sala, E. E. Scheibler, H. Wu, S. A. Wissinger & B. P. Batzer, 2022. Perils of life on the edge: climatic threats to global diversity patterns of wetland macroinvertebrates. Science of the Total Environment 820: 1–10.
Fitter, A. H., J. Browne, T. Dixon & J. J. Tucker, 1980. Ecological studies at Askham Bog Nature Reserve. 1. Inter-relations of vegetation and environment. Naturalist 105: 89–101.
Foster, G., 1993. Pingo fens, water beetles and site evaluation. Antenna 17: 184–195.
Frajer, J. & D. Fiedor, 2018. Discovering extinct water bodies in the landscape of Central Europe using toponymic GIS. Moravian Geographical Reports 26: 121–134.
Gilbert, P. J., D. A. Cooke, M. E. Deary, S. Taylor & M. J. Jeffries, 2016. Quantifying rapid spatial and temporal variations of CO2 fluxes from small, lowland freshwater ponds. Hydrobiologia 793: 83–93.
Gilbert, P. J., S. Taylor, D. A. Cooke, M. E. Deary & M. J. Jeffries, 2021. Quantifying organic carbon storage in temperate pond sediments. Journal of Environmental Management. https://doi.org/10.1016/j.jenvman.2020.111698.
Gregg, R., J. L. Elias, I. Alonso, I.E. Crosher, P. Muto & M.D. Morecroft, 2021. Carbon storage and sequestration by habitat: a review of the evidence (second edition) Natural England Research Report NERR094. Natural England, York.
Grinham, A., S. Albert, N. Deering, M. Dunbabin, D. Bastviken, B. Sherman, C. E. Lovelock & C. D. Evans, 2018. The importance of small artificial water bodies as sources of methane emissions in Queensland, Australia. Hydrology and Earth Systems Sciences 22: 5281–5298.
Hallett, T., T. Coulson, J. Pilkington, T. Clutton-Brock, J. Pemberton & B. Grenfell, 2004. Why large-scale climate indices EEM to predict ecological processes better than local weather. Nature 430: 71–75.
Heckenberger, M. J., J. C. Russell, J. R. Toney & M. J. Schmidt, 2007. The legacy of cultural landscapes in the Brazilian Amazon: implications for biodiversity. Philosophical Transactions of the Royal Society B 362: 197–208.
Higgins, S. L., F. Thomas, B. Goldsmith, S. J. Brooks, C. Hassall, J. Harlow, D. Stone, S. Völker & P. White, 2019. Urban freshwaters, biodiversity, and human health and well-being: Setting an interdisciplinary research agenda. Wiley Interdisciplinary Reviews: Water. https://doi.org/10.1002/wat2.1339.
Hill, M. J., D. B. Ryves, J. C. White & P. J. Wood, 2016. Macroinvertebrate diversity in urban and rural ponds: implications for freshwater biodiversity conservation. Biological Conservation 201: 50–59.
Hill, M. J., C. Hassall, B. Oertli, L. Fahrig, B. J. Robson, J. Biggs, M. J. Samways, N. Usio, N. Takamura, J. Krishnaswamy & P. J. Wood, 2017. New policy directions for global pond conservation. Conservation Letters. https://doi.org/10.1111/coml.12447.
Hill, M. J., H. M. Greaves, C. D. Sayer, C. Hassall, M. Milin, V. S. Milner, L. Marazzi, R. Hall, L. R. Harper, I. Thornhill, R. Walton, J. Biggs, N. Ewald, A. Law, N. Willby, J. C. White, R. A. Briers, M. J. Mathers, M. J. Jeffries & P. J. Wood, 2021. Pond ecology and conservation: research priorities and knowledge gaps. Ecosphere 12: e03853.
Hinden, H., B. Oertli, N. Menetrey, L. Sager & J.-B. Lachavanne, 2005. Alpine pond biodiversity: what are the related environmental variables? Aquatic Conservation: Marine and Freshwater Ecosystems 15: 613–624.
Holgerson, M. A. & P. A. Raymond, 2016. Large contribution to inland waters CO2 and CH4 emissions from very small ponds. Nature Geosciences. https://doi.org/10.1038/NGEO2654.
Hunter, M. L., V. Acuña, D. M. Bauer, K. P. Bell, A. J. K. Calhoun, M. R. Felipe-Lucia, J. A. Fitzsimmons, E. González, M. Kinnison, D. Lindemayer, C. J. Lundquist, R. A. Medellin, E. J. Nelson & P. Psoschold, 2016. Conserving small natural features with large ecological roles: a synthetic overview. Biological Conservation 211: 88–95.
Huq, N., 2017. Small scale freshwater ponds in rural Bangladesh: navigating roles and services. Indian Journal of Water 11: 73–85.
Jeffries, M. J., 2012. Ponds and the importance of their history: an audit of pond numbers, turnover and the relationship between the origins of ponds and their contemporary plant communities in south east Northumberland, UK. Hydrobiologia 689: 11–12.
Jeffries, M. J., 2016. Flood, drought and the inter annual variation to the number and size of ponds and small wetlands in an English lowland landscape over three years of weather extremes. Hydrobiologia 768: 255–272.
Jeffries, M.J., L. Epele, J.M. Studinski & C.F. Vad, 2016. Invertebrates in temporary wetland ponds of the temperate biomes. In Invertebrates in Freshwater Wetlands. An International Perspective on Their Ecology. Switzerland: Springer.
Kifner, L. H., A. J. K. Calhoun, S. A. Norton, K. E. Hoffmann & A. Amirbahman, 2018. Methane and carbon dioxide dynamics within four vernal pools in Maine, USE. Biogeochemistry 139: 275–291.
Kuhn, M. K., E. J. Lundin, R. Giesler & M. Johansson, 2018. Emissions from thaw ponds largely offset the carbon sink of northern permafrost wetlands. Nature Scientific Reports 8: 9535.
Lewis-Phillps, J., S. J. Brooks, C. D. Sater, I. R. Patmore, G. M. Hilton, A. Harrison & H. Robson, 2020. Ponds as insect chimneys: restoring overgrown farm ponds benefits birds through elevated productivity of emerging insects. Biological Conservation. https://doi.org/10.1016/j.biocon.2019.108253.
Manoj, K. & P. K. Padhy, 2015. Environmental perspectives of pond ecosystems: global issues, services ad Indian scenarios. Current World Environment. https://doi.org/10.12944/CWE.10.3.16.
Marcé, R., B. Obrador, L. Gomez-Gener, N. Catalán, M. Koschorreck, M. I. Arce, G. Singer & D. von Schiller, 2019. Emissions from dry land waters are a blind spot in the global carbon cycle. Earth-Science Reviews 188: 240–248.
Martínez-Sanz, C., C. S. S. Canzano, M. Fernández-Aláez & F. García-Criado, 2012. Relative contribution of small mountain ponds to regional richness of littoral macroinvertebrates and the implications for conservation. Aquatic Conservation 22: 155–164.
McClain, M. E., E. W. Boyer, C. L. Dent, S. E. Gergel, N. B. Grimm, P. M. Groffman, S. C. Hart, J. W. Harvey, C. A. Johnston, E. Mayorga, W. H. McDowell & G. Pinay, 2003. Biogeochemical hot spots and hot moments at the interface of terrestrial and aquatic ecosystems. Ecosystems 3: 301–312.
Mo, Y., Z.-H. Deng, J.-Q. Gao, Y.-X. Guo & F.-H. Yu, 2015. Does richness of emergent plants affect CO2 and CH4 emissions in experimental wetlands? Freshwater Biology 60: 1537–1544.
Natural England, n.d. https://www.gov.uk/government/publications/national-character-area-profiles-data-for-local-decision-making/national-character-area-profiles.
Nicolet, P., J. Biggs, G. Fox, M. J. Hodson, C. Reynolds, M. Whitfield & P. Williams, 2004. The wetland plant and macroinvertebrate assemblages of temporary ponds in England and Wales. Biological Conservation 120: 261–278.
Obrador, B., D. von Schiller, R. Marcé, L. Gómez-Gener, M. Koschorreck & C. Borrego, 2018. Dry habitats sustain high CO2 emissions from temporary ponds across seasons. Nature Scientific Reports 8: 3105.
Oertli, B., 2018. Freshwater biodiversity conservation: the role of artificial ponds in the 21st century. Aquatic Conservation: Marine and Freshwater Ecosystem 28: 264–269.
Oertli, B., R. Céréghino, A. Hull & R. Miracle, 2009. Pond conservation: from science to practice. Hydrobiologia 634: 1–9.
Ortega, S. H., C. R. González-Quijano, P. Casper, G. A. Singer & M. O. Gessner, 2019. Methane emissions from contrasting urban freshwaters: rate, drivers, and a whole-city footprint. Global Change Biology. https://doi.org/10.1111/gcb.14799.
Peacock, M., J. Audet, S. Jordan, J. Smeds & M. B. Wallin, 2019. Greenhouse gas emissions from urban ponds are driven by nutrient status and hydrology. Ecosphere 10: 1–10.
Peacock, M., J. Audet, D. Bastviken, S. Cook, C. D. Evans, A. Grinham, M. A. Holgerson, L. Högbom, A. E. Pickard & P. Zieliński, 2021. Small artificial waterbodies are widespread and persistent emitters of methane and carbon dioxide. Global Change Biology: https://doi.org/10.1111/gcb.15762.
Pitt, A. L., R. F. Baldwin, D. J. Lipscomb, B. L. Brown, J. E. Hawley, C. M. Allard-Keese & P. B. Leonard, 2012. The missing wetlands: using local knowledge to find cryptic ecosystems. Biodiversity and Conservation 21: 51–63.
Polishchuk, Y. M., A. N. Bogdanov, I. N. Muratov, V. Y. Polishchuk, A. Lim, R. M. Manasypov, L. S. Shirokova & O. S. Pokrovsky, 2018. Minor contribution of small thaw ponds to the pools of carbon and methane in the inland waters of the perma-frost-affected part of the Western Siberian Lowland. Environmental Research Letters 13: 045002.
Poudel, J. M., 2018. Pond becomes as lake: challenges posed by climate change in the Trans-Himalayan regions of Nepal. Journal of Forest and Livelihood 16: 87–102.
Reverey, F., H.-P. Grossart, K. Premke & G. Lischeid, 2016. Carbon and nutrient cycling in kettle hole sediments depending on hydrological dynamics; a review. Hydrobiologia 775: 1–20.
Rewilding Britain, 2021. Rewilding and Climate Breakdown. How Restoring Nature can help Decarbonise the UK, Rewilding Britain, Leeds:
Riley, W. D., E. C. E. Potter, J. Biggs, A. L. Collins, H. P. Jarvie, J. I. Jones, M. Kelly-Quin, S. J. Ormerod, D. A. Sear, R. L. Wilby, S. Broademadow, C. D. Brown, P. Chanin, G. H. Copp, I. G. Cowx, A. Grogan, D. D. Hornby, D. Huggett, M. G. Kelly, M. Naura, J. R. Newman & G. M. Siriwardena, 2018. Small water bodies in Great Britain and Ireland: ecosystem function, human-generated degradation, and options for restorative action. Science of the Total Environment 645: 1598–1616.
Rosentreter, J. A., A. V. Borges, B. R. Deemer, M. A. Holgerson, S. Liu, C. Song, J. Melack, P. A. Raymond, C. M. Duarte, G. H. Allen, D. Olefeldt, B. Poulter, T. I. Battin & B. D. Eyre, 2021. Half of global methane emissions come from highly variable aquatic ecosystem sources. Nature Geogscience 14: 225–230.
Scott, A., I. M. D. Maclean, A. Byfield, A. R. Pay & R. J. Wilson, 2012. Artificial disturbance promotes recovery of rare Mediterranean temporary pond plant species on the Lizard Peninsula, Cornwall, England. Conservation Evidence 9: 76–86.
Seekell, D. A. & M. L. Pace, 2011. Does the Pareto distribution adequately describe the size-distribution of lakes? Limnology and Oceanography 56: 350–356.
Seekell, D. A., M. L. Pace, J. L. Tranvik & C. Verpoorter, 2013. A fractal-based approach to lake size distribution. Geophysical Research Letters 40: 517–521.
Søndergard, M., E. Jeppesen & J. P. Jensen, 2005. Pond or lake: does it make any difference? Archive Für Hydrobiologie 162: 143–165.
Stafford, R., B. Chamberlain, L. Clavey, P.K. Gillingham, S., McKain, M.D. Morecroft, C. Morrison-Bell & O. Watts, (Eds.), 2021. Nature-based solutions for climate change in the UK: a report by the British Ecological Society, London.
Sun, H., Q. Xin, Z. Ma & S. Lan, 2019. effects of plant diversity on carbon dioxide emissions and carbon removal in laboratory scale constructed wetland. Environmental Science and Pollution Research 26: 5076–5082.
Taylor, S., P. J. Gilbert, D. A. Cooke, M. E. Deary & M. J. Jeffries, 2019. High carbon burial rates by small ponds in the landscape. Frontiers of Ecology and Environment 17: 25–31.
Torgersen, T. & B. Branco, 2008. Carbon and oxygen fluxes from a small pond to the atmosphere: temporoal variability and the CO2/O2 imbalance. Water Resources Research. https://doi.org/10.12944/CWE.10.3.16.
Travnik, L. J., J. J. Cole & Y. T. Prairie, 2018. The study of carbon in inland waters – from isolated ecosystems to players in the global carbon cycle. Limnology and Oceanography Letter 3: 41–48.
Tsai, J.-S., L. S. Venne, S. T. McMurry & L. M. Smith, 2011. Local and landscape influences on plant communities in playa wetlands. Journal of Applied Ecology 49: 174–181.
Usio, N., M. Nakagawa, T. Aoki, S. Higuchi, Y. Kadono, Y. M. Akasaka & N. Takamura, 2017. Effects of land use on trophic states and multi-taxonomic diversity in Japanese farm ponds. Agriculture, Ecosystems and Environment 247: 205–215.
Van Bergen, T. J. H. M., N. Barros, R. Mendonça, R. C. H. Aben, L. H. J. Althuizen, V. Huszar, L. P. M. Lamers, M. Lurling, F. Roland & K. Kosten, 2019. Seasonal and diel variation in greenhouse gas emissions from an urban pond and its major drivers. Limnology and Oceanography 64: 2129–2139.
Vickruck, J. L., L. R. Best, M. P. Gavin, J. H. Devries & P. Galpern, 2019. Pothole wetlands provide reservoir habitat for native bees in prairie croplands. Biological Conservation 232: 43–50.
Walmsley, A., 2008. The Norfolk ‘Pingo’ Mapping Project, Norfolk Wildlife Trust, Norwich:
Wik, M., R. K. Varner, K. W. Anthony, S. MacIntyre & D. Bastviken, 2016. Climate-sensitive northern lakes and ponds are critical components of methane release. Nature Geoscience 9: 99–105.
Williams, P., J. Biggs, A. Crowe, J. Murphy, P. Nicolet, A. Weatherby, M. Dunbar, 2010. Countryside Survey: Ponds Report from 2007. Technical Report No. 7/07 Pond Conservation and NERC/Centre for Ecology & Hydrology, Lancaster.
Yvon-Durocher, G., C. J. Hulatt, G. Woodward & M. Trimmer, 2017. Long-term warming amplifies shifts in the carbon cycle of experimental ponds. Nature Climate Change. https://doi.org/10.1038/NCLIMATE3229.
Acknowledgements
We are very grateful to the landowners for allowing access and sampling of their sites, in particular on the very special Askham, Thompson Common and Lizard reserves. P.G. was funded by a Northumbria University postgraduate studentship. Thank you to the SEFS special session organisers for allowing us to present this work.
Funding
P.G. was funded by a Northumbria University postgraduate studentship. The authors have no other direct or indirect competing interests in the research.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Fieldwork, material preparation and data collection were performed by PJG and ST, and analyses were performed by PJG, ST and MJ. The first draft of the manuscript was written by MJ based on his SEFS 2021 presentation and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have not disclosed any competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Guest editors: Mary Kelly-Quinn, Jeremy Biggs, J. Iwan Jones & William D. Riley / Small Waterbodies in the Landscape
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Jeffries, M.J., Gilbert, P.J., Taylor, S. et al. Organic carbon in British lowland ponds: estimating sediment stocks, possible practical benefits and significant unknowns. Hydrobiologia 850, 3225–3239 (2023). https://doi.org/10.1007/s10750-022-04972-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10750-022-04972-z