Abstract
Variation in jaw protrusion is critical to cichlid fish trophic diversification. For instance, jaw protrusion distance can influence suction, attack speed, as well as bite force, and jaw protrusion angle is associated with exploiting prey from different substrates. Interestingly, premaxillary ascending process length has been shown to influence the maximum distance some cichlid fishes protrude their oral jaws, but its relationship to jaw protrusion angle is unclear. Using phylogenetic comparative methods, morphometrics, and field measurements in Malawi cichlid species, we tested the relationship between the length of the premaxillary ascending process and two components of jaw protrusion. In Malawi, the premaxilla's ascending process length ranged from 6.9 to 15.2% with respect to standard length. Maximum jaw protrusion ranged from 1.4 to 9.1% of standard length and jaw protrusion angle varied from 17° to 76°. Although jaw protrusion angle was not associated with premaxillary ascending process length, phylogenetically adjusted correlations between the ascending process and maximum jaw protrusion distance were highly significant. Evolutionary change in the premaxilla is likely critical for determining the maximum distance, but not the angle, of Malawi cichlid jaw protrusion. Examinations of this type of potential phenotypic multi-functionality will continue to illuminate the mechanisms contributing to cichlid fish diversity.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The ability of cichlids and other fishes to protrude their oral jaws has likely contributed to their unparalleled diversity (Alexander, 1967; Lauder, 1982; Ferry-Graham & Lauder, 2001). Cichlids are well known for exhibiting specialized feeding modes, and many of their stunning array of trophic abilities are tied to their oral jaw protrusion abilities (Schaeffer & Rosen, 1961; Ferry-Graham & Lauder, 2001; Hulsey & García de León 2005; Hulsey et al. 2019). For example, jaw protrusion can increase the speed that a predator can approach evasive prey fish (Waltzek & Wainwright, 2003), adapt feeding movements to the position of benthic dwelling prey types (Motta, 1984), increase the distance over which a predator can suck difficult to obtain items into the mouth (Ferry-Graham et al., 2001; Wainwright et al., 2001 , 2015), and augment the hydrodynamic forces exerted on attached and evasive prey (Holzman et al., 2008). Although several mechanisms are considered to contribute to the ability to protrude the oral jaws, the length of the ascending process of the premaxilla, that forms the toothed bone of the upper oral jaw in cichlids, is likely critical to the maximum distance that the jaw can be protruded (Witte, 1983; Hulsey et al., 2010; Bellwood et al., 2015). The premaxilla's ascending process has been found to be relatively long in a number of cichlids and other groups of fishes that protrude their jaws extensively as it serves to guide the upper jaw out during protrusion and back into its resting position as the jaws are closed (Eaton, 1943; Alexander, 1967; Westneat & Wainwright, 1989; Westneat, 1991; Ferry-Graham et al., 2001; Waltzek & Wainwright, 2003; Hulsey et al., 2010). However, because of their unparalleled diversity, Malawi cichlids could show strikingly different patterns of functional morphology when compared to other fishes (Liem 1973; Liem & Osse, 1975; Hulsey et al. 2019). Moreover, other mechanisms, such as rotation of the lower jaw, cranial elevation, and the four-bar linkage system of the anterior jaws, are also important in the evolution of jaw protrusion in cichlids and other fishes (Lauder & Liem, 2004; Westneat, 1991; Hulsey & Wainwright, 2002; Waltzek & Wainwright, 2003; Hulsey & García de León, 2005; Martinez et al., 2018; Hulsey et al., 2019). It also remains unclear how the length of the ascending process influences other aspects of jaw protrusion such as the angle at which the oral jaw is protruded. To evaluate the importance of the ascending process to Malawi cichlid trophic evolution, we examined whether the length of this bone is evolutionarily associated with either their angle of jaw protrusion or their maximum jaw protrusion distance.
Jaw protrusion angle and maximum jaw protrusion distance appear to evolve independently in Lake Malawi cichlids, but are both influenced by some of the same craniofacial skeletal components (Hulsey et al., 2019). Jaw protrusion distance has also tended to receive much more attention than the angle of jaw protrusion (Wainwright et al., 2001; York et al., 2015; Roberts et al. 2021). However, protrusion angle could be as crucial during cichlid feeding as protrusion distance, because the orientation of a fish’s mouth is often associated with exploitation of trophic resources from different substrates (Motta, 1984; Rupp & Hulsey, 2014; Konow et al., 2017). For instance, terminal mouths protrude in the same direction as the fish’s body thereby enhancing the ability to rapidly close the distance to a prey item in open water (Waltzek & Wainwright, 2003). Conversely, benthic-feeding species that feed from algae-covered rocks or by excavating sandy substrates often have subterminal mouths (Motta, 1984; Genner & Turner 2005). Transitions between feeding benthically or feeding in the water column represent major trophic shifts that have occurred repeatedly in Lake Malawi cichlids (Hulsey et al., 2013). Therefore, there is likely substantial evolutionary replication even within the Lake Malawi cichlids to examine the functional importance of traits like the length of the ascending process that are increasingly being examined in ecomorphological studies of cichlid diversification (Hulsey et al., 2010; Lopez-Fernandez et al., 2013; Ronco et al., 2021). Several skeletal elements in the oral jaws also contribute to the evolution of both jaw protrusion traits (Hulsey et al., 2019). Therefore, despite their apparent independent functional co-evolution, both jaw protrusion distance and jaw protrusion angle might depend on and be predicted from the length of the premaxillary ascending process.
To test the importance of the premaxillary ascending process to functional aspects of jaw protrusion, it would be ideal to examine the co-evolution of these traits in a well-resolved phylogenetic context. Because of their relatively young age and the rampant incomplete lineage sorting among genetic loci, the relationships among Malawi cichlids have been difficult to disentangle (Albertson et al., 1999; Hulsey et al., 2007; Hulsey et al. 2018a, b). However, whole-genome sequences are now providing much more robust estimates of systematic relationships among Malawi cichlids (Malinsky et al., 2018; Masonick et al., 2022; Scherz et al., 2022). These rapidly emerging phylogenetic frameworks of Malawi cichlid relationships are now sufficiently resolved to permit the widespread use of phylogenetic comparative analyses to examine the co-evolution of morphological and functional traits.
In this study, we combined morphological measurements, field-based measurements of jaw protrusion, and a recently reconstructed whole-genome based phylogenetic framework for 37 species of Lake Malawi cichlids. We then tested the evolutionary relationship between the length of the premaxilla ascending process and both maximum jaw protrusion distance as well as jaw protrusion angle.
Materials and methods
Premaxillary ascending process morphology
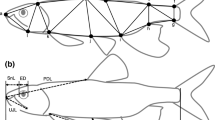
Specimens collected from the wild in Lake Malawi in 2010 from five sites (Table 1) were used to measure morphological traits. Fish were collected using SCUBA and barrier nets. The standard length (SL) and two functional aspects of jaw protrusion traits (below) of species were measured in the field on live specimens immediately following capture. The fish were then euthanized with an overdose of MS222 according to the University of Tennessee Animal Care and Use Committee (IUCAC protocol #1833), subsequently fixed in formalin, and then transferred to 70% ETOH for long-term storage and subsequent morphological measurements. The length of the ascending process of the premaxilla was measured from its most posterior process to the base of the tooth on the most anterior part of the premaxilla using dial calipers (Fig. 1a). The measurements were made with the calipers following the cutting of a small incision in the skin directly behind the most posterior point of the ascending process. To size-standardize the measurements for analyses, the ascending process of the premaxilla length was expressed as a percentage of SL (Table 2). To contextualize the extensive trophic variation in the cichlids examined (Ribbink et al., 1983; Konings, 1990), their general dietary habits as categorized primarily in Hulsey et al. (2007) are included (Table 2) to provide an ecological context for the examined morphology and jaw protrusion traits.
The length of the ascending process a of the premaxilla bone (shaded gray) was measured in 37 Lake Malawi cichlid species. Two functional aspects of jaw protrusion b maximum protrusion distance and jaw protrusion angle were measured on cichlids in the field using calipers and a protractor. The congeneric species Nimbochromis polystigma (Regan, 1922) and N. linni (Burgess & Axelrod, 1975) highlight the rapid evolution of variation in these jaw protrusion traits in the Malawi cichlids
Jaw protrusion
Immediately following collection of fish from Lake Malawi, we measured both maximum jaw protrusion distance and jaw protrusion angle on 2 to 30 recently euthanized individuals per species (Table 2). Maximum protrusion distance was determined using two measurements: 1. "jaw closed" and 2. "jaw open". Jaw closed was measured as the distance from the premaxilla's anterior tip to the most posterior point of the eye when the jaws were closed. Jaw open was determined as the distance from the tip of the premaxilla to the posterior point on the eye when the jaws were maximally extended. Special care was taken to measure to the tip point where the teeth emerged from the premaxilla especially with the species with hypertrophied lips to ensure the lips did not influence the measurements. We then subtracted jaw closed from jaw open and size-standardized these measurements by standard length (SL). This size-standardization allowed us to comparatively assess how maximum jaw protrusion might differ functionally among these species during prey capture. To measure jaw protrusion angle which was assumed to be independent of SL, we placed specimens upon water-proof graph paper. Then, we centered the pupil where two graph lines intersected and positioned the specimen so that one line ran through the body axis to the center of the caudal peduncle. The point that the front of the head intersected the line running through the body axis was then marked. We then rotated the lower jaws with forceps and marked the anterior tip of the premaxilla when the upper jaw was maximally protruded. Using a protractor (Fig. 1b), we determined the jaw protrusion angle in degrees (°) with respect to the body axis.
Phylogenetic comparative analyses
Correlations between the ascending process length and jaw protrusion variables were examined in a phylogenetic context. Since correlations among species values are not statistically independent because of the shared evolutionary history of species (Felsenstein, 1985), we calculated the phylogenetic independent contrasts 'pic' for these variables and examined their correlation in the 'ape' package (Paradis et al., 1984) in the R programming environment (R Core Team 2021). We performed these analyses on the highest maximum likelihood phylogeny taken from an analysis performed in IQ-TREE for 1,107,249 noncoding SNPs generated from resequenced whole-genomes (Masonick et al., 2022). For the comparative analyses, the branch lengths of this topology were first rendered ultrametric using non-parametric rate smoothing based on penalized likelihood (Sanderson 2002) using the function 'chronoPL' with a lamda smoothing parameter of 0.1 as implemented in the R program 'ape' (Paradis et al. 2004). This lambda value represents a trade-off between each branch having its own rate (lamda = 0.0), and increased lambda values where similar rate variation is assumed for all branches. We allowed the maximum age of the entire Lake Malawi clade to be set to the default of 1.0 and each descendant node to represent a fraction of this time. As the maximum age of the Lake Malawi radiation is assumed to be ~ 2.0 million years (Kocher and Danley 2001), inferred ages of nodes can be estimated by multiplying the fractional node age on the initial phylogeny by 2.0 (Fig. 2). To highlight the patterns of jaw protrusion evolution in Lake Malawi cichlids, the two jaw protrusion traits were mapped onto this initial phylogeny using maximum likelihood reconstruction of ancestral states performed using the 'fastAnc' function of R package 'phytools' (Revell 2012). Additionally, to account for phylogenetic uncertainty in the recovered topology and branch lengths for the independent contrast analyses, we also permuted relationships and branch lengths in the pruned topology 100 times using the function 'swapONE' implemented in the R package 'RRphylo' (Castiglione et al., 2018). Using a probability of switching node position as well as node age of 50%, we used these 100 randomizations to produce standard errors associated with the phylogenetically corrected correlation coefficients and their associated P-values obtained for the Lake Malawi cichlid species.
The relationships for the Lake Malawi cichlids used as a framework for comparative analyses (supplementary information 1) and pruned down to the study species from a phylogeny presented in Masonick et al. (2022). For the comparative analyses, this maximum likelihood phylogeny based on whole-genome resequencing was rendered ultrametric using penalized likelihood (Sanderson, 2002) and its branch lengths and topology permuted to simulate uncertainty in the phylogeny. In the upper left corner, Tyrannochromis nigriventer represents one of the species with the highest and Petrotilapia sp. represents a species with the lowest amount of jaw protrusion distance in the Malawi cichlids examined. At the phylogeny's tips separated by "/" and preceding the Latin name for each cichlid, we give the maximum jaw protrusion distance, jaw protrusion angle, and diet categories for each species (Table 2). To illustrate the general patterns of this trait evolution in the Malawi cichlids, ancestral states for maximum jaw protrusion distance (above each ancestral node) and jaw protrusion angle (below each ancestral node) were also reconstructed using maximum likelihood. The time axis in millions of years at the bottom of the phylogeny provides a rough temporal context for this diversification. This temporal scale was inferred simply from the smoothed branch lengths and the timeframe of ~ two million years over which the Malawi radiation is believed to have diversified (Danley & Kocher, 2001)
Results
For the 37 Malawi cichlid species examined, there was substantial diversity in length of the ascending process of the premaxilla (Table 2). The size-standardized length of the ascending process ranged from a low of 6.9% of SL in Labeotropheus trewavassae Fryer, 1956 to 15.2% of SL in Tyrnannochromis nigriventer Eccles, 1989. The average length for this critical skeletal element in the Malawi species examined was 9.8% of SL.
There was also substantial diversity in the two functional attributes of jaw protrusion examined in the Lake Malawi cichlids. For the 37 Malawi cichlid species examined, maximum jaw protrusion distance as a percentage of SL was 5.4% on average. The smallest amount of protrusion was recovered for the rock-dwelling fin biter Genyochromis mento (Trewavas, 1935) at only 1.4% of SL. The highest amount of jaw protrusion was recovered for the planktivore Copadichromis virginalis (Iles, 1960) (9.1%) and piscivore Tyranochromis nigriventer (8.1%). For all the species examined, mean jaw protrusion angle was measured as 43°. Protrusion angles indicative of more terminal mouths tended to be the smallest in planktivorous species like Copadichromis virginalis (17°), C. trimaculatus (Iles, 1960) (28°), and Mchenga eucinostomus (Regan, 1922) (28°) as well as piscivores like Tyranochromis nigriventer (24°) and Nimbochromis polystigma (27°). The species having the greatest angles of jaw protrusion, or most subterminal mouths, were Chilotilapia rhoadesii (Boulenger, 1908) (65°) as well as the species Tropheops microstoma (Trewavas, 1935) (67°), Labeotropheus trewavassae (72°), and L. artatorostris Pauers, 2017 (76°) who all three exploit algal resources in their rocky habitats.
The evolution of the premaxillary ascending process length was strongly correlated with the evolution of maximum jaw protrusion distance. The phylogenetic independent contrast correlation (picr) of these two variables was found to be 0.72 (P < 0.0001) on the best tree from Masonick et al., (2022). When we permuted relationships and branch lengths of this phylogeny, the ascending process length and maximum jaw protrusion distance were similarly and consistently strongly associated (mean picr = 0.74 ± 0.10 S.E.; mean P = 0.006 ± 0.06 S.E.). However, the contrast correlation of premaxillary ascending process length and jaw protrusion angle (Fig. 3b) was found to be not significant (picr = 0.15; P = 0.37) on the best tree. When we permuted relationships and branch lengths, the ascending process length and jaw protrusion angle also had no clear association (mean picr = 0.02 ± 0.27 S.E.; mean P = 0.43 ± 0.31 S.E.). In sum, the evolution of the premaxillary ascending process length appears to have little influence on jaw protrusion angle but readily predicts the evolution of maximum jaw protrusion distance.
A representative sample of the relationships between phylogenetic independent contrasts of premaxillary ascending process length as a percent of standard length (%SL) and size-standardized maximum jaw protrusion distance (a) as well as jaw protrusion angle (b). While the premaxillary ascending process length was not evolutionarily correlated with jaw protrusion angle across a range of permuted phylogenetic relationships (mean picr = 0.02 ± 0.27 S.E.; mean P = 0.43 ± 0.31 S.E.), phylogenetically corrected analyses found the length of the ascending process had a substantial correlation with maximum jaw protrusion distance (mean picr = 0.74 ± 0.10 S.E.; mean P = 0.006 ± 0.06 S.E.)
Discussion
The ascending process of the premaxilla has diversified substantially during the evolution of Malawi cichlids. Additionally, maximum jaw protrusion ranged from 1.4%–9.1% with respect to standard length and jaw protrusion angle ranged from 17° to 76° with respect to the long axis of the body (Table 2; Fig. 3). Although the diversity produced during the evolution of the premaxillary ascending process length in Malawi appears to have had little influence on jaw protrusion angle, change in the length of this craniofacial element readily predicts Malawi cichlid evolution of maximum jaw protrusion distance. This positive relationship mirrors what has been found in other teleost fishes (Eaton, 1943; Alexander, 1967; Westneat & Wainwright, 1989; Westneat, 1991; Ferry-Graham et al., 2001; Waltzek & Wainwright, 2003; Hulsey et al., 2010) while also reinforcing the lack of clarity in what morphological variation determines the angle of jaw protrusion in Malawi cichlids.
Nevertheless, Lake Malawi cichlids exhibit substantial diversity in both functional attributes of jaw protrusion that is even more impressive considering these traits have diverged in only approximately two million years (Fig. 2; Table 2). For instance, the smallest amount of protrusion was recovered for the rock-dwelling fin biter Genyochromis mento that protrudes its jaws only 1.4% of SL. This minimal jaw protrusion is extreme but is generally reflected in other closely related Malawi cichlids in genera like Labeotropheus and Petrotilapia that also likely do not need extensive jaw protrusion to exploit algal resources (Ribbink et al., 1983; Rupp & Hulsey 2014). In contrast, planktivores like Copadichromis virginalis and piscivores like Nimbochromis linni and Tyranochromis nigriventer tended to have extensive maximum jaw protrusion abilities. This might be favored to enhance suction feeding or overtaking prey during their predatory strikes (Wainwright et al., 2001; Waltzek & Wainwright, 2003; Hulsey & García De León, 2005; Holzman et al., 2008). Jaw protrusion angle also varied extensively in the Malawi cichlids. Protrusion angles indicative of more terminal mouths tended to be the smallest in planktivores like Copadichromis virginalis and Mchenga eucinostomus as well as piscivores like Nimbochromis polystigma. These relatively low jaw protrusion angles likely reflect their generally linear predatory attack path when overtaking rapidly escaping prey like fish and plankton (Ferry-Graham & Lauder, 2001; Wainwright et al., 2001). In contrast, species having the greatest jaw protrusion angles, or most subterminal mouths, were algivorous species like Tropheops microstoma and Labeotropheus species. This large angle of jaw protrusion likely helps them to maintain a parallel body axis to the rocky habitats they feed from and perhaps allows them to better maintain position or watch for competitors when exploiting algal resources (Rupp & Hulsey, 2014). The variation in both maximum protrusion distance and jaw protrusion angle likely reflect major ecomorphological axes of trophic diversification in Lake Malawi cichlids.
One of the primary goals of ecomorphology is to identify the types of morphometric characters that can be used to make consistent and clear inferences on the maximum abilities of an organism (Barel, 1982; Wainwright, 1996). Some other simple ecomorphological variables, such as oral gape width setting a maximum size of prey that a predatory fish can ingest or the cross-sectional area of the pharyngeal biting muscles determining the force a crushing predator can exert on a prey, have substantial explanatory power (Wainwright et al., 2005). Similarly, the length of the premaxillary ascending process appears to provide a highly predictive variable for determining the maximum distance that a cichlid can protrude its jaws (Fig. 3). Numerous other mechanisms such as rotation of the lower jaw, cranial elevation, and the four-bar linkage system of the anterior jaws are likely important in jaw protrusion of cichlids (Westneat, 1991; Waltzek & Wainwright, 2003; Hulsey & García de León, 2005; Holzman & Hulsey 2017; Hulsey et al., 2019). Nevertheless, the tight evolutionary relationship between length of the ascending process and the amount of jaw protrusion suggests the ascending process is likely critical to the evolution of cichlid trophic diversity (Witte, 1983; Hulsey et al., 2010).
Although we only recovered one clear evolutionary association between Malawi cichlid morphological variation and functional diversity in jaw protrusion, many of the traits that have diversified in Malawi cichlids are multi-functional (Holzman et al., 2012; Hulsey et al., 2019). This multi-functionality could be key to the incredible diversification of groups like Malawi cichlids (Liem, 1980; Wainwright et al., 2005). For instance, traits like cichlid eyes (Hulsey et al., 2007; Hofmann et al., 2009), body coloration (Urban et al., 2022), cranial linkages (Holzman & Hulsey, 2017; Hulsey et al., 2019), and teeth (Gorman & Hulsey, 2020; Karagic et al., 2020) are all likely to have more than a single function in Malawi cichlids. However, the ability of traits to evolve independently has also been suggested to be critical to cichlid diversification (Liem, 1973, 1980; Liem & Osse, 1975; Hulsey et al., 2006, 2019; Holzman et al., 2012). If we are to fully understand mechanistically why some groups like Malawi cichlids adaptively radiate while other groups do not, it will remain critical to further comparatively test how morphological variation translates into functional diversity.
References
Albertson, R. C., J. A. Markert, P. D. Danley & T. D. Kocher, 1999. Phylogeny of a rapidly evolving clade: the cichlid fishes of Lake Malawi, East Africa. Proceedings of the National Academy of Sciences 96: 5107–5110.
Alexander, R. . Mc. N., 1967. The functions and mechanisms of the protrusible upper jaws of some acanthopterygian fish. Journal of Zoology 151: 43–64.
Barel, C. D. N., 1982. Towards a constructional morphology of cichlid fishes (Teleostei, Perciformes). Netherlands Journal of Zoology 33: 357–424.
Bellwood, D. R., C. H. R. Goatley, O. Bellwood, D. J. Delbarre & M. Friedman, 2015. The rise of jaw protrusion in spiny-rayed fishes closes the gap on elusive prey. Current Biology 25: 2696–2700.
Castiglione, S., G. Tesone, M. Piccolo, M. Melchionna, A. Mondanaro, C. Serio, M. Di Febbraro & P. Raia, 2018. A new method for testing evolutionary rate variation and shifts in phenotypic evolution. Methods in Ecology and Evolution 9: 974–983.
Danley, P. D. & T. D. Kocher, 2001. Speciation in rapidly diverging systems, lessons from Lake Malawi. Molecular Ecology 10: 1075–1086.
Eaton, T. H., Jr., 1943. An adaptive series of protractile jaws in Cichlid fishes. Journal of Morphology 72: 183–190.
Felsenstein, J., 1985. Phylogenies and the comparative method. The American Naturalist 125: 1–15.
Ferry-Graham, L. A. & G. Lauder, 2001. Aquatic prey capture in Ray-finned fishes: A century of progress and new directions. Journal of Morphology 248: 99–119.
Ferry-Graham, L. A., P. Wainwright, C. D. Hulsey & D. Bellwood, 2001. Evolution and mechanics of long jaws in Butterflyfishes (Family Chaetodontidae). Journal of Morphology 248: 120–143.
Genner, M. J. & G. F. Turner, 2005. The mbuna cichlids of Lake Malawi: a model for rapid speciation and adaptive radiation. Fish and Fisheries 6: 1–34.
Gorman, C. E. & C. D. Hulsey, 2020. Non-trophic functional ecology of vertebrate teeth: a review. Integrative and Comparative Biology 60: 665–675.
Hofmann, C. M., K. E. O’Quin, N. J. Marshall, T. W. Cronin, O. Seehausen & K. L. Carleton, 2009. The eyes have it: regulatory and structural changes both underlie cichlid visual pigment diversity. PLOS Biology 7: e1000266.
Holzman, R. & C. D. Hulsey, 2017. Mechanical transgressive segregation and the rapid origin of trophic novelty. Scientific Reports 7: 40306.
Holzman, R., S. W. Day, R. S. Mehta & P. C. Wainwright, 2008. Jaw protrusion enhances forces exerted on prey by suction feeding fishes. Journal of the Royal Society Interface 5: 1445–1457.
Holzman, R., D. C. Collar, S. A. Price, C. D. Hulsey, R. C. Thomson & P. C. Wainwright, 2012. Biomechanical trade-offs bias rates of evolution in the feeding apparatus of fishes. Proceedings of the Royal Society B 279: 1287–1292.
Hulsey, C. D. & F. J. García De León, 2005. Cichlid jaw mechanics: linking morphology to feeding specialization. Functional Ecology 19: 487–494.
Hulsey, C. D. & P. C. Wainwright, 2002. Projecting mechanics into morphospace: disparity in the feeding system of labrid fishes. Proceedings of the Royal Society B 269: 317–326.
Hulsey, C. D., F. Garcia De Leon & R. Rodiles-Hernandez, 2006. Micro- and macroevolutionary decoupling of cichlid jaws: a test of Liem’s key innovation hypothesis. Evolution 60: 2096–2109.
Hulsey, C. D., M. C. Mims & J. T. Streelman, 2007. Do constructional constraints influence cichlid craniofacial diversification? Proceedings of the Royal Society B 274: 1867–1875.
Hulsey, C. D., P. Hollingsworth Jr. & R. Holzman, 2010. Coevolution of the premaxilla and jaw protrusion in cichlid fishes (Heroine: Cichlidae). Biological Journal of the Linnean Society 100: 619–629.
Hulsey, C. D., R. J., Roberts Y.-H. E., Loh M. F., Rupp & J. T. Streelman, 2013. Lake Malawi cichlid evolution along a benthic/limnetic axis. Ecology and Evolution 3(7): 2262–2272.
Hulsey, C. D., R. Holzman & A. Meyer, 2018a. Dissecting a potential spandrel of adaptive radiation: body depth and pectoral fin ecomorphology coevolve in Lake Malawi cichlid fishes. Ecology and Evolution 8: 11945–11953.
Hulsey, C. D., J. Zheng, R. Holzman, M. E. Alfaro, M. Olave & A. Meyer, 2018b. Phylogenomics of a putatively convergent novelty: did hypertrophied lips evolve once or repeatedly in Lake Malawi cichlid fishes? BMC Evolutionary Biology 18: 179.
Hulsey, C. D., M. E. Alfaro, J. Zheng, A. Meyer & R. Holzman, 2019. Pleiotropic jaw morphology links the evolution of mechanical modularity and functional feeding convergence in Lake Malawi cichlids. Proceedings of the Royal Society B 286: 20182358.
Karagic, N., R. F. Schneider, A. Meyer & C. D. Hulsey, 2020. A genomic cluster containing novel and conserved genes is associated with cichlid fish dental developmental convergence. Molecular Biology and Evolution 37: 3165–3174.
Konings, A., 1990. Konings’s Book of Cichlids and All the Other Fishes of Lake Malawi, T.F.H. Publications, Neptune City:
Konow, N., S. Price, R. Abom, D. Bellwood & P. Wainwright, 2017. Decoupled diversification dynamics of feeding morphology following a major functional innovation in marine butterflyfishes. Proceedings of the Royal Society B 284: 20170906.
Lauder, G. V., 1982. Patterns of evolution in the feeding Mechanism of actinopterygian fishes. American Zoologist 22: 275–285.
Lauder, G. V. & K. Liem, 2004. Prey capture by Luciocephalus pulcher: implications for models of jaw protrusion in teleost fishes. Environmental Biology of Fishes 6: 257–268.
Liem, K. F., 1973. Evolutionary strategies and morphological innovations: cichlid pharyngeal jaws. Systematic Biology 22: 425–441.
Liem, K. F., 1980. Adaptive significance of intra- and interspecific differences in the feeding repertoires of cichlid fishes. American Zoologist 20: 295–314.
Liem, K. F. & J. W. M. Osse, 1975. Biological versatility, evolution, and food resource exploitation in African cichlid fishes. American Zoologist 15: 427–454.
López-Fernández, H., J. H. Arbour, K. O. Winemiller & R. L. Honeycutt, 2013. Testing for ancient adaptive radiations in neotropical cichlid fishes. Evolution 67: 1321–1337.
Malinsky, M., H. Svardal, A. M. Tyers, E. A. Miska, M. J. Genner, G. F. Turner & R. Durbin, 2018. Whole-genome sequences of Malawi cichlids reveal multiple radiations interconnected by gene flow. Nature Ecology & Evolution 2: 1940–1955.
Martinez, C., M. McGee, S. Borstein & P. Wainwright, 2018. Feeding ecology underlies the evolution of cichlid jaw mobility. Evolution 72: 1645–1655.
Masonick, P., A. Meyer & C. D. Hulsey, 2022. Phylogenomic analyses show repeated evolution of hypertrophied lips among Lake Malawi cichlid fishes. Genome Biology and Evolution 14: evac051.
Motta, P. J., 1984. Mechanics and functions of jaw protrusion in teleost fishes: a review. Copeia 1984: 1–18.
Paradis, E., J. Claude & K. Strimmer, 2004. APE: analyses of phylogenetics and evolution in R language. Bioinformatics 20: 289–290.
R Core Team, 2021. R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, https://www.R-project.org/.
Revell, L. J., 2012. Phytools: an R package for phylogenetic comparative biology (and other things). Methods in Ecology and Evolution 3: 217–223.
Ribbink, A. J., B. A. Marsh, A. C. Marsh, A. C. Ribbink & B. J. Sharp, 1983. A preliminary survey of the cichlid fishes of rocky habitats in Lake Malawi. South African Journal of Zoology 18: 149–310.
Roberts, A. S., J. R. Hodge, P. Chakrabarty & P. C. Wainwright, 2021. Anatomical basis of diverse jaw protrusion directionality in ponyfishes (Family Leiognathidae). Journal of Morphology 282: 427–437.
Ronco, F., M. Matschiner, A. Böhne, A. Boila, H. H. Büscher, A. El Taher, A. Indermaur, M. Malinsky, V. Ricci, A. Kahmen, S. Jentoft & W. Salzburger, 2021. Drivers and dynamics of a massive adaptive radiation in cichlid fishes. Nature 589: 76–81.
Rupp, M. F. & C. D. Hulsey, 2014. Influence of substrate orientation on feeding kinematics and performance of algae-grazing Lake Malawi cichlid fishes. Journal of Experimental Biology 217: 3057–3066.
Sanderson, M. J., 2002. Estimating absolute rates of molecular evolution and divergence times: a penalized likelihood approach. Molecular Biology and Evolution 19: 101–109.
Schaeffer, B. & D. E. Rosen, 1961. Major adaptive levels in the evolution of the actinopterygian feeding mechanism. American Zoologist 1: 187–204.
Scherz, M. D., P. Masonick, A. Meyer & C. D. Hulsey, 2022. Between a rock and a hard polytomy: Phylogenomics of the rock-dwelling mbuna cichlids of Lake Malawi. Systematic Biology 15: syac006.
Urban, S., J. Gerwin, C. D. Hulsey, A. Meyer & C. F. Kratochwil, 2022. The repeated evolution of stripe patterns is correlated with body morphology in the adaptive radiations of East African cichlid fishes. Ecology and Evolution 12: e8568.
Wainwright, P. C., L. A. Ferry-Graham, T. B. Waltzek, A. M. Carroll, C. D. Hulsey & J. R. Grubich, 2001. Evaluating the use of ram and suction during prey capture by cichlid fishes. Journal of Experimental Biology 204: 3039–3051.
Wainwright, P. C., M. E. Alfaro, D. I. Bolnick & C. D. Hulsey, 2005. Many-to-One mapping of form to function: a general principle in organismal design? Integrative and Comparative Biology 45: 256–262.
Wainwright, P. C., M. D. McGee, S. J. Longo & L. P. Hernandez, 2015. Origins, innovations, and diversification of suction feeding in vertebrates. Integrative and Comparative Biology 55: 134–145.
Waltzek, T. & P. Wainwright, 2003. Functional morphology of extreme jaw protrusion in Neotropical cichlids. Journal of Morphology 257: 96–106.
Westneat, M., 1991. Linkage biomechanics and evolution of the unique feeding mechanism of Epibulus insidiator (Labridae: Teleostei). Journal of Experimental Biology 159: 165–184.
Westneat, M. W. & P. C. Wainwright, 1989. Feeding mechanism of Epibulus insidiator (Labridae; Teleostei): evolution of a novel functional system. Journal of Morphology 202: 129–150.
Witte, F., 1983. Consistency and functional significance of morphological differences between wild-caught and domestic Haplochromis squamipinnis (Pisces, Cichlidae). Netherlands Journal of Zoology 34: 596–612.
York, R. A., C. Patil, C. D. Hulsey, O. Anoruo, J. T. Streelman & R. D. Fernald, 2015. Evolution of bower building in Lake Malawi cichlid fish: phylogeny, morphology, and behavior. Frontiers in Ecology and Evolution 3: 18.
Acknowledgements
We thank Phillip Hollingsworth and Richard Zatha for help in the field. We also thank the Malawi Parks Department for providing us permits to collect cichlids in Lake Malawi.
Funding
Open Access funding provided by the IReL Consortium. The authors have not disclosed any funding.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no financial or non-financial interests that are directly or indirectly related to the work submitted for publication.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Guest editors: S. Koblmüller, R. C. Albertson, M. J. Genner, K. M. Sefc & T. Takahashi / Advances in Cichlid Research V: Behavior, Ecology and Evolutionary Biology
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Hulsey, C.D., Gorman, C.E. Coevolution of the premaxillary ascending process and jaw protrusion in Lake Malawi cichlid fishes. Hydrobiologia 850, 2231–2240 (2023). https://doi.org/10.1007/s10750-022-04949-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10750-022-04949-y