Abstract
The behaviour of returning Salmo salar (Linnaeus, 1758) approaching, and attempting to pass low-head weirs remains relatively unknown. A radio telemetry array was created at a low-head weir to enable the behaviour of S. salar (n = 120) to be observed as they approached and attempted to pass the barrier. The majority of fish successfully passed the barrier on their first or second attempt, some individuals required 11 attempts prior to successful passage occurring. Mean delay at the barrier per fish was 47.8 h (±SD 132.0 h), range 15 min to 31 days. Passage success on a fish’s initial attempt was significantly predicted by the amount of searching a fish undertook, fork length, and fat content. Fish were more likely to have a successful first passage attempt if it was smaller with a low fat content and exerted a greater effort in searching for a passage channel. Small-scale barriers cause delays and increased energy expenditure in migrating fish. Barriers may be creating an anthropogenic selection pressure for traits which enable passage success. The impact of a delay at a barrier and increased energy expenditure on reproduction and gonad development remains unknown but is likely to be negatively impacted by instream anthropogenic structures.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The loss and fragmentation of habitat truncate movement, reduce connectivity, and often precede the decline and extirpation of a species (Ceballos & Ehrlich, 2002; Baguette et al., 2013). In rivers, habitat connectivity is primarily longitudinal and in general confined to the river corridor. A single impoundment thus has the potential to isolate adjacent habitats completely for many species (Jager et al., 2001; Cote et al., 2009; Branco et al., 2012). In-river structures, both natural and artificial such as waterfalls and weirs can have major impacts on species that have multiple, life stage dependent, aquatic habitat requirements. Highly mobile anadromous and catadromous fish, which have a complex life-cycle, are among some of the species most affected. The Atlantic salmon (Salmo salar Linnaeus, 1758) is one species shown to be highly vulnerable to river corridor fragmentation (Baras et al., 1994; Lucas & Frear, 1997; Jager et al., 2001; O’Hanley & Tomberlin, 2005; Kemp et al., 2008).
The impacts of large-scale obstacles (>5 m hydraulic head height), particularly their effect on fish migrations, are well documented (Gowans et al., 2003; Antonio et al., 2007; Meixler et al., 2009; Branco et al., 2012). Considerable effort has been made to mitigate the effects of river obstacles through the development of fish passes, which aim to facilitate the upstream and downstream migration of individuals around or through obstacles (Larinier, 1998; Guiny et al., 2005; Bunt et al., 2012). The efficiency of such structures is however often questioned; flow conditions such as water velocity and depth within the pass itself are not always conducive to upstream passage of fish (Thorstad et al., 2008; Noonan et al., 2012; Cooke & Hinch, 2013). For example, the addition of fish screens at the 86-m-high Pitlochry Dam (Scotland) increased the proportion of fish ascending the dam from 45% of fish which attempted (Webb, 1990) up to 100% by guiding fish away from the turbine entrances (Gowans et al., 1999).
Fish pass facilities are generally built at large, high head impoundments. Low-head obstacles (defined here as <5 m hydraulic head height), in general, lack such passage structures, relying on the fish’s own ability to successfully ascend them. In Europe, there is a legislative framework requiring EU member states to ensure fish passage and are outlined within the EU Water Framework Directive (Directive 2000/60/EC), and EU Eel legislation (EC No. 1100/2007). It is estimated that within England and Wales there are some 25,000 in-river obstructions, of which 3,000 are significant and require mitigation in order to meet the ecological objectives set out in these directives (Environment Agency, 2009). There is a paucity of knowledge on the effects of low-head obstacles; it is likely that they may also present serious deleterious impacts for fish populations through habitat fragmentation (Lucas & Frear, 1997; Ovidio & Philippart, 2002; O’Connor et al., 2006). Determining the likelihood of fish passage at riverine obstacles is highly complex due to variable swimming and leaping capabilities of fish of different sizes and species, coupled with the heterogeneity of environmental variables associated with riverine systems (Ovidio & Philippart, 2002; Sigourney et al., 2015). Viewed in the terms of fish passage, any single obstacle may prevent migration, cause a temporary delay in migration or have no effect. The likelihood is that man-made obstacles will disrupt upstream migration, resulting in at least some delay in the upstream movement of migratory fish.
There is evidence that upstream migrating adult Atlantic salmon are sometimes reluctant to pass obstacles which present no obvious physical impediment to upstream movement (Gerlier & Roche, 1998; Ovidio & Philippart, 2002). An upstream migrating Atlantic salmon was unable to surmount an obstacle 1.4 m in height due to low water depth below the obstacle and insufficient water depth on the face of the weir (Ovidio & Philippart, 2002). Conversely Chanseau et al. (1999) indicated that Atlantic salmon were successful in ascending low obstacles <1.5 m in height within 24 h on the Pau River (France). In the same study, severe delays were encountered at high obstacles, >2.5 m in height with passage highly dependent on specific fish passage structures and downstream pool water depth. Low-head obstacles rely on variations in flow conditions (e.g. water depth and velocity) to facilitate passage (Kemp et al., 2008; Meixler et al., 2009; Silva et al., 2011). Low-head obstacle permeability is likely to change significantly with environmental conditions, particularly flow, with fish characteristics (such as species and body size) and environmental conditions combining to create a discrete period of time when passage may be successful (Kemp & O’Hanley, 2010).
The biological consequences of a delayed migration are unclear, logically however, increased movement and searching behaviours caused as a direct result of an encounter with an impassable (even if only temporarily) riverine obstacle is likely to result in increased energy expenditure. Fish attempting to ascend through the Baigts hydroelectric station (Gave de Pau River, France) were delayed up to 80 days despite the presence of a fish pass. Telemetry demonstrated that fish moved between the fish pass and a holding pool approximately 500-m downstream, expending energy in attempting to pass the barrier (Chanseau & Larinier, 1999). The increased energy expenditure associated with obstacle passage may translate into a subsequent cost on gonad production and spawning activity. In Atlantic salmon, energetic costs cannot be recovered as adult salmon cease feeding while in fresh water (Mills, 1989; Bardonnet & Baglinière, 2000). Ultimately, energy loss associated with obstacle navigation has the potential to reduce the overall fitness of the individual. A number of studies have shown successful migrants (i.e. individuals which reached spawning grounds) had lower approach and passage times at obstacles when compared with unsuccessful individuals (Chanseau et al., 1999; Naughton et al., 2005; Lundqvist et al., 2008; Makiguchi et al., 2011), suggesting potentially rapid obstacle passage reduces energetic costs in barrier passage resulting in greater success of reproduction.
Radio telemetry provides a technique to investigate the behaviour and migration pathways of fish in the wild, providing data on temporal and spatial scales that were previously unattainable. In the study reported here, a radio telemetry detection array was established to investigate the movements of wild Atlantic salmon as they approached and attempted to pass a low-head, complex (multiple potential passage channels), riverine obstacle during the upstream spawning migration. The aim of this study was to: (1) determine the behaviour of fish prior to attempts to ascend a river obstacle; (2) determine the behavioural response of fish when they are unable to ascend the obstacle; (3) determine the length of any potential delay at a low-head obstacle; and (4) determine the characteristics of fish that determine passage success.
Methods
Study site
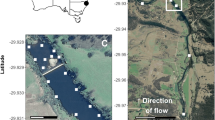
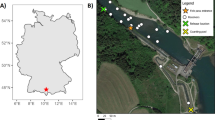
The Foyle system (55°00′N; 07°20′W) has a catchment area of 4450 km2 and forms part of the border between Northern Ireland and the Republic of Ireland (Fig. 1). The Foyle system is a designated European Union, Special Area of Conservation (SAC) for Atlantic salmon. The River Mourne, the largest tributary of the catchment, has a number of riverine obstacles along its length, the most downstream of which is located at Sion Mills (54°46.968N; 7°27.689W). As there is no spawning habitat downstream of the obstacle at Sion Mills, anadromous fish must pass this obstacle to access spawning grounds upstream. The obstacle at Sion Mills is a complex sloping weir which presents multiple potential channels of passage for migrating fish (Figs. 2, 3).
The Foyle catchment showing location on the border between Northern Ireland and the Republic of Ireland within the small inset. The large map outlines the river barrier location (Sion mills weir) and telemetry array along with the capture and release site for fish in 2012. Also highlighted is the capture and release site of fish in 2013
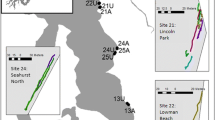
Cross section of the weir transects identified in Fig. 2. These cross sections represent the three possible channel options for fish ascending the main face of the weir. The width (meters) and slope (%) are outlined for each downstream portion of the weir face
The weir is 265 m wide (left bank to right bank) and is positioned at approximately 50° to the main flow of the river (Fig. 2). Its purpose is to deflect water into an old mill lade, which now generates hydropower. The outlet of the lade is completely inaccessible to fish due to the presence of an electric barrier. The weir has a sloping main face, presenting a swim obstacle to fish and, under certain conditions, at the foot of the barrier, a leap obstacle. The foot of the weir falls directly onto a bedrock and boulder substrate. The weir has become degraded and eroded (Figs. 2, 3) resulting in variation in the effective length of the weir for fish passage (the distance that a passing fish is required to negotiate) varies along its width (Fig. 3). Two fish passes are present; one a Denil pass on the right-hand bank and a Larinier pass in the centre of the weir (Fig. 2). Beside the Larinier pass, are two attraction channels designed to guide flow towards the foot of the pass, enabling fish to locate and ascend this route. Two deep channels have been carved in the bedrock leading to the entrance of each fish pass. These are designed to guide fish to suitable passage channels. Both fish passes are highly turbulent and, due to river bed scouring, the Larinier pass now requires a leap for fish to access it. Fish are able to cross the weir without using the fish passes.
The weir profile is best described by three transects which are representative of the wider weir structure at each location. Each transect was selected to most accurately reflect the wider weir face in the immediate area. The physical characteristics of each transect are presented in Fig. 3. In general, fish are required to swim up a sloping weir, the gradient of which varies (Fig. 3). In transect A, fish are also required to leap onto the weir face before attempting to ascend, a plunge pool is also present at this location.
Fish capture and tagging
Atlantic salmon were captured during the spawning migration of 2012 and 2013. In 2012, Atlantic salmon were collected with a fish trap installed within the upstream section of the Denil fish pass (Figs. 1, 2). The trap (3 × 2.5 × 2 m) was checked periodically (two or more times daily). Fish were removed from the trap by dip net and transferred to a holding box for examination. In 2013, obstacle-naïve fish were collected through draft (seine) netting, downstream of the weir, within the tidal part of the river (Fig. 1). Fish were netted during darkness and transferred directly to a holding box filled with fresh river water for inspection. Fish were rejected from the study if they indicated any signs of disease or physical damage. Prior to tagging, fish were immersed in an anaesthetic bath of clove oil (Ethanol: clove oil 10:1, 0.5 mg per litre). Once anaesthetised, a radio tag (Model: F1835, Advanced Telemetry Systems) was inserted via the oesophagus into the stomach. Fish were then held to recover in fresh water whilst fork length and depth of the fish were measured. Fat content was measured by using a fish fat meter (Distell, Model—FM 692). A panjet was used to mark each fish with alcian blue dye between the pectoral fins on the ventral surface of the fish to enable anglers easy identification and subsequent release of tagged individuals. Anglers were encouraged to release fish and take note of captures through information leaflets dispersed through the catchment. Fish were placed into a protective sling and weighed. In 2012, fish were then placed into a fish transport box containing aerated river water before being transferred to the release site downstream of the weir (Fig. 2). On release, fish were held by hand in slow flowing current and allowed to recover. In 2013, following weighing, fish were transferred to a holding pen submerged within the river in an area of gentle flow for recovery and to prevent recapture by subsequent netting attempts. Fish were released at the end of each netting session.
Telemetry array and fish tracking
A telemetry array was installed within the vicinity of the weir to enable the movements of tagged individuals to be assessed (Fig. 2). Three fixed automatic listening stations (R4520c/R4500, Advanced Telemetry Systems, inc. Minnesota, United States of America) were used to create eight detection zones detailed in Fig. 2. Coaxial cable was stripped to create aerials; the length of exposed core was modified to create varying detection ranges. Aerials were either exposed to air (wide detection, n = 4) or submerged in water (confined detection, n = 4). Wide detection antennas were mounted on concrete structures at the weir to provide some directionality in detection. In combination with variable gain receivers, this set-up enabled the establishment of precise detection zones (Fig. 3). Range testing was conducted throughout the study period to ensure that these detection areas were maintained. Receivers scanned on an individual aerial for 6 s before selecting the next aerial, upon detecting a fish scan time increased to 15 s to enable tag identification. Post-processing of the data enabled fish location to be determined through a stepwise process. If a fish was detected at multiple detection zones, it was deemed to be within the most confined zone of those detected. For example, if a fish was detected at Zone 2 and Zone 3, the fish was deemed to be in Zone 2 due to the confined detection area of that zone.
Wide detection zones were used to investigate broad behaviour patterns for fish approaching and leaving the obstacle vicinity. Upstream migrating fish would first be detected at a large detection zone 150–450 m downstream of the weir. This ‘downstream detection’ zone covered a deep pool which had been reported (pers comm) as a holding area for fish. A wide detection zone was installed between the weir and the downstream detection zone called the fall back detection zone (Fig. 2). The fall back detection zone was used to detect fish which were in the vicinity of the weir but not necessarily directly within confined detection zones at the weir face (Fig. 2. Zone 1–5). A large detection zone upstream, ‘upstream detection’ zone enabled identification of fish that had successfully passed the weir.
Detection zones were created at all channel passages where it was possible to place equipment (Fig. 2). Zone 1 identified when fish had ascended the initial baffled section of the Denil fish pass into a holding pool within the pass itself. Zone 2 detected fish at the entrance to the Denil pass. Zone 3 covered the right-hand bank, detecting fish as they approached the weir face; Zone 4 detected fish as they approached the left hand bank of the main weir face. There was a small overlap between zones 3 and 4. A combination of the signal strength and the number of tag detections was used to determine whether fish were located in Zone 3 or 4. Zone 5 identified fish at the entrance of the Larinier fish pass.
The telemetry array was operational throughout the study period in each year (May to the following January). Outside the obstacle array, from 8-km downstream to 14-km upstream, locations of tagged fish were recorded a minimum of 5 days per week by manual bankside tracking between 1st May and 1st December. Wider area searches across the catchment and tributaries were undertaken every two to 3 days to try and locate fish which had moved out with the intensive search area. In January 2013, a fly over with an aerial mounted on a helicopter was undertaken to search all major tributaries of the catchment.
Fish movement and behaviour
Fish behaviour was quantified through a number of metrics. ‘Total delay’ is defined as the time difference between the first detection of an individual at the weir face (i.e. in Zones 1–5, Fig. 2), and the time at which passage was deemed to have occurred, defined by detection at the upstream detection zone. For many fish, total delay will include multiple passage attempts. An ‘attempt’ is defined here as detection of the fish at the weir face aerials (Zone 1–5). A new ‘attempt’ was assigned when there was a gap in detections at weir face detection Zones (1–5) of greater than 15 min or if the fish was detected continuously on a downstream aerial. ‘Passage attempt time’ is the difference in time from the start of an attempt to the end of an attempt. Passage attempt time is assumed to represent the time spent searching at the weir face for successful passage. An attempt and passage attempt time is deemed to have ended when either a fish passes the barrier and is detected on the upstream detection zone (also a ‘successful passage’), or when fall-back occurs (‘unsuccessful passage’). ‘Fall back’ is deemed to have occurred by continuous detections in the downstream or fallback detection zones (Fig. 2), or where there is a gap in the data where the fish is no longer detected at weir face aerials.
‘Fallback’, in this study, is defined by a fish moving downstream between any individual passage attempt. The fallback ‘distance’ and ‘location’ were split into 3 categories: ‘Short range’ (<80 m from weir), fish remained close to the weir within the fallback detection zone but not detected within weir face zones (Zone 1:5). ‘Medium range;’ fish held between the fallback detection zone and the downstream detection zone (~130 m from weir). ‘Long range;’ fish moved downstream and held within a deep pool covered by the downstream detection zone (>225 m downstream from weir; Fig. 3) or further.
To determine if fish were attracted to specific areas of the weir, the proportion of time spent in each zone (Zones 1–5) during the entire attempt was calculated, and the zone with the highest proportion of time was assumed to be the channel of preference for that fish. χ 2 tests were used to determine if greater numbers of fish were attracted to specific sections of the weir. If no preference was observed there would be equal numbers of fish exhibiting a preference across each of the detection zones.
A number of non-parametric tests were conducted on behavioural traits. Shapiro-Wilk normality tests were used to test for normality in the data, log10 transformations failed to improve the spread of the data thus leading to the use of Wilcox rank sum tests on the following: (1) A difference in the total delay for each fish between years. (2) A difference in the passage attempt time between a successful or unsuccessful first passage attempt. (3) A difference in passage attempt time between first and second attempts.
Measures of behaviour were modelled to determine what factors enabled a rapid successful passage over the obstacle with a minimal delay. An initial mixed logistic regression model (Model 1) was developed to identify the variables determining passage success on an individual’s first passage attempt. The response variable was binary, either passage success occurred or it did not, the independent response variables are outlined in Table 1. A second model (Model 2) was developed to determine the independent variables influencing passage attempt time on an individual’s successful passage attempt. The response variable was the passage attempt time recorded when the fish successfully crossed the weir; the independent response variables are outlined in Table 1. Within each model, an interaction between mean search flow and mean search temperature as tested to account for the reduction in temperature associated with increased discharge. The predictor variables were selected based on a subjective approach whereby variables most likely to have a known biological mechanistic effect on the response were utilised as opposed to exhaustive searching. Due to low sample size and low a priori knowledge of factors effecting behaviour exhaustive searching may identify correlations but the relative importance of this unknown hence a subjective approach in model formulation was undertaken.
All analysis was conducted using R (R version 3.1.3 [2015-03-09]) statistical computing package (R Core Team, 2013).
Environmental data
River flow data for the rivers were provided in the form of discharge data at 15-min intervals (provided by the Department of Agriculture and Rural Development, Northern Ireland). The discharge at each passage attempt was taken as the mean discharge of all data records during the specific passage attempt. Temperature data are recorded remotely and provided by the Loughs Agency for every 15-min period. The temperature for each passage attempt was taken as the mean temperature of data records during the specific passage attempt.
Day and night values were calculated using the sunriset function in the maptools package developed by Bivand and Lewin-Koh (2016) within R (R Core Team, 2013). Light conditions were used within Chi-squared tests to determine if there was a preference for passage attempts either during daylight or at night.
Modelling approach
Fish behaviour within years was likely to be more similar than between years as a result of environmental variables and capture/release method; thus, a mixed modelling approach was taken with ‘year’ included as a random effect. Data exploration identified outliers which were removed and independent variables violating the assumption of non-collinearity were also removed.
Due to the complexities associated with the highly exploratory nature of this study, a priori information about predictor relevance is relatively unknown. The glmulti function in the glmulti package (Calcagno, 2013) enables the generation of all possible model formulas from a set of specified effects from which model selection is performed. Glmulti is a general wrapper for glm and related functions and generates all possible model formulas. The glmulti function (Calcagno & Mazancourt, 2010), was utilised in conjunction with the glmer (Model 1) and lmer (Model 2) functions within the lme4 package (Bates et al., 2015) with a wrapper to enable use of random effects (Calcagno & Mazancourt, 2010), uses a genetic algorithm to sample a large number of first order models (the terms within the model are a subset of the full model) and was used to allow selection of the model comprising the best set of independent variables with minimum Akaike Information Criterion (AIC). The best candidate models within two AIC units (competing models) were assessed based on Akaike weights which is considered as the weight of evidence in favour of model i being the actual best model. In addition, evidence ratios of the Akaike weights were used to determine strength of support for the best model, and the modelled sum of weights was used to estimate the relative importance of variables under consideration (Burnham & Anderson, 2002, Symonds & Moussalli, 2011). P values were obtained by likelihood ratio tests of the full model with the effect in question against the model without the effect in question. The model selected based on the best AIC sometimes included independent variables which were not significant. Final models were generated with non-significant variables being dropped as determined by likelihood ratio tests.
Results
Of the 132 fish tagged (mean ± SD for: fork length [L F] = 609.2 ± 41.65 mm, mass = 2.96 ± 0.51 kg, fat content = 9.52 ± 3.82%) in this two-year study (12 in 2012 and 120 in 2013), 51 fish (39%) were detected within the telemetry array and 40 (77%) of these fish were deemed to have had a successful passage attempt (9 in 2012, 31 in 2013). Of the 11 fish that were detected but failed to pass, one fish arrived at the obstacle but failed in ascending; 10 fish were detected in the stream reach immediately downstream of the weir, and however they were not detected at the weir itself. The ultimate fate of the 11 fish that did not pass the obstacle could not be determined, it is possible that they were destined for a neighbouring river system.
The following results are based on 36 salmon of the 40 which successfully ascended the weir. Four fish were removed from the analysis. Three of these fish were detected upstream by manual tracking; however their passage route at the weir could not be determined and were removed from any subsequent analysis. It is possible that these fish ascended the weir under flood conditions where routes not normally available for passage and not covered by the telemetry array, and were accessible for a brief period of time when high water conditions allowed; however, their exact behaviour cannot be determined. One fish was not detected at the weir but was routinely tracked to a location downstream of the weir (approx. 9 km), and was subsequently detected upstream of the study site via an aerial tracking survey (17 January 2014); it is likely this fish ascended the weir after the array ceased to operate.
Time to first detection at the weir from release was highly variable (mean ± SD = 48.7 ± 33.7 days); two fish reached the weir in under five hours after release; conversely the maximum time to detection at the weir was 130 days. Mean total delay at the weir per fish was 47.8 h (±SD 132.0 h, range 15 min to 31 days) with no significant difference in total delay between years (Wilcox–Rank–Sum, W = 138, P = 0.44).
The majority of fish were successful in passing the weir on either their first (46%) or second (43%) attempt. However, four fish required 3, 5 ,7 and 11 attempts respectively, to ascend the weir. Mean passage attempt time per fish was 561 ± 1707 SD minutes (median = 132 min, range 8 min to 10 days). Mean passage attempt time on a successful attempt was 755 ± 2370 SD minutes (median = 125 min, range 10 min to 10 days) but this was not significantly different (Wilcox–Rank–Sum, W = 79, P = 0.7) from first unsuccessful attempts (passage attempt time mean ± SD = 378 ± 611 min, range 8–2760 min). Mean passage attempt time for successful first attempts was 198 ± 213 SD minutes (range 23–867 min) but not significantly different (Wilcox–Rank–Sum, W = 86, P = 0.2) from fish which passed on their second passage attempt (mean = 1343.267 ± S.D. 3567 min, median = 240 min, range 10 min to 10 days). A greater number of passage attempts were initiated during daylight hours compared with darkness hours (χ 2 = 20.1, P = < 0.001); however, there was no significant difference between the number of successful passage attempts in either the day or night (χ 2 = 0.04, P = 0.8).
Behaviour at the weir
Analysis indicated significant channel preference during all passage attempts (χ 2 = 164.8, P < 0.001), successful passage attempts (χ 2 = 97.2, P < 0.001), and unsuccessful passage attempts (χ 2 = 97.2, P < 0.001). Out of all successful passage attempts, 33 of the 36 attempts occurred at Zone 4. For unsuccessful passage attempts, 31 of 37 occurred at Zone 4 with five individuals making attempts at Zone 3 and one individual at Zone 2.
Fish pass use
The total number of observations of fish on weir face aerials (Zone 1–5) was 22,460; of these, 1831 (8.2%) were at the entrance to the constructed fish pass channels (Zone 2 and Zone 5); however, 1665 (91%) of these detections came from a single individual, indicating only 166 detections (0.74%) came from other fish. Of the 45 fish detected at the weir, 20 had at least one detection in Zone 2 or Zone 5; however, 12 of these fish had less than 8 detections at the foot of fish pass channels. A significantly greater (χ 2 = 1050.7, P = < 0.001) number of detections occurred in Zone 2 than in Zone 5. Three fish (8%), of those making a successful passage attempt, were deemed to have utilised the fish pass as a successful passage route, all three fish passed through the Denil fish pass (Fig. 3, Zone 2 and Zone 1). No fish ascended through the Larinier fish pass. Fish tagged in 2012, which were initially caught within the pass did not re-ascend through this channel, instead re-ascending over the weir face.
Factors influencing passage speed and success
The binary response of a fish’s success or failure at ascending the obstacle on its first attempt was modelled using logistic regression (glmer) with explanatory variables (outlined in Table 1). From 2,100 models, the best model indicated by AIC scores was that which included; zone per unit time (χ 2 (1) = 4.99; P = 0.03), length (χ 2 (1) = 10.09, P = 0.002), and fat content (χ 2 (1) = 4.71, P = 0.03) (Fig. 4). A fish was more likely to have a successful first passage attempt if it was smaller (fork length) with a low fat content and exerted a greater effort in searching for a passage channel (Fig. 4). A number of competing models (11) were also identified within two AIC units of the best model. Further examination of Akaike weights ratios suggests that there is low support for the best model, the evidence ratio for the best model, versus the model two AIC units worse is only 2.67. The variable akaike weight for the three variables indicated that the best model was in excess of 0.9 and thus strong evidence that these variables are components of the actual ‘best’ model.
Individual effects of significant variables identified by model selection (model 1) on their influences on passage success. These plots were generated using the ‘effects’ package (Fox, 2003)
To examine the factors (Table 1) influencing the passage attempt time on successful obstacle passage, the passage attempt time on each successful passage attempt, for attempts one and two (due to highly unbalanced data across all attempts; only four fish had more than two attempts) is modelled on predictor variables (outlined in Table 1) with the addition of the passage attempt number (one or two). Following model validation, the dependent variable was log transformed log10(y). The model was re-run with the transformed data and the assumptions of homogeneity of variance and normality were confirmed. Out of 1,050 models, the model of lowest AIC was that which included only the ‘mean search flow’ (Table 1) as an independent variable (χ 2 (1) = 25.26, P = < 0.001). Passage attempt time increases with mean search flow during an attempt. Although this model was ranked best by AIC, a number of competing models (56) were also identified within two AIC units of the best model. Further examination of Akaike weights ratios suggests that there is low support for the best model; the evidence ratio for the best model versus the model two AIC units worse is only 2.7. The variable weight for mean search flow was in excess of 0.9 and thus strong evidence that this variable is a component of the ‘best’ model.
Fall-back
Following a failed passage attempt, the distance to which fish moved downstream was highly variable, one individual fell back downstream 3.4 km following an unsuccessful passage attempt, but did eventually ascend the weir. Another individual, despite being detected at the weir and registering a passage attempt, fell back downstream and was later recorded 45 km away in a neighbouring river system and did not ascend the weir.
Discussion
This is the first study to identify the behaviour of returning adult Atlantic salmon on approach to a low-head complex river obstacle over small spatial scales. It has highlighted their ability to surmount such a structure but also the variability in behaviour which is required to do so. Of the 41 tagged fish which were detected at the weir, 40 were successful in ascending the obstacle enabling them to reach suitable spawning grounds.
The variability in delay and also the number of attempts prior to successful passage may be linked with physiological characteristics of individuals. A significant predictor in an initial passage attempt was fish length, with larger fish less likely to be successful in their initial passage attempt. Similarly, Kristinsson et al. (2015) observed a small but significant positive relationship between delay at an obstacle and fish length. For high average thrust, fish need large caudal fins (Weihs, 1973), and as Webb (1973) suggests, a deep caudal fin is required to generate high acceleration. Conditions which prevent a fish utilising its caudal fin at maximum efficiency, such as in shallow water where part of the fin is exposed to air, ultimately reduces a fishes thrust. Hence, shallow water flowing over a sloping weir face (as presented by the barrier in this study) will reduce the ability of larger fish with deeper caudal fins to ascend them. There is evidence to suggest that alterations in selective regimes may occur within 6-15 generations in salmonids following implementation of hydroelectric dams or fish ladders (Haugen et al., 2008; Fraser et al., 2011). It has previously been reported that successful fish passage has a significant size-selective influence on upstream migrating Atlantic salmon with larger fish less likely to successfully ascend hydroelectric dams (Sigourney et al., 2015). The findings presented here support this position and suggest that selection may be occurring at much smaller riverine obstacles, and not those which are at the upper limits of salmon swimming ability.
Laboratory experiments have shown maximum swimming speeds vary substantially between physiological capabilities (Fisher & Hogan, 2007) and populations of the same species (Webb et al., 1984; Ralph et al., 2012). Thus, it may not be surprising that some individuals take longer to ascend the obstacle than others; this is particularly true when passage relies heavily on the swimming ability of an individual which is pre-determined by its physical characteristics and genetic make-up (Fisher & Hogan, 2007). Sockeye salmon have been shown to be predisposed at the beginning of their migration to their fate (success or failure), Genetic profiling indicated survivors expressed 88 genes at a higher level than mortalities, suggesting individuals die due to a variety of physiological reasons, whereas those which survive have a common physiology (Cooke et al., 2008). It can be hypothesised that successful passage may partly be a result of their genetic make-up, again providing evidence for the anthropogenic selection of salmon which are able to ascend barriers.
Kinnison et al. (2016) demonstrate that the cost of migration is not only at the expense of tissue energy reserve, but also a cost in ovarian investment expressed through reduced egg size. Greater delay exposes fish to predation/angling pressure and increased energetic expenditure. Mesa and Magie (2006) found that Chinook salmon Oncorhynchus tshawytscha migrating slowly between dams in the Columbia river basin utilised 5–8% more energy from muscle than faster migrants. Over an average of 29 days, fish lost between 6 and 17% of muscle energy density depending on their travel time (Mesa & Magie, 2006). A cumulative delay of such magnitude may not be uncommon along a river length with multiple low-head obstacles. Energy expenditure is also likely to increase with the number of passage attempts a fish makes to ascend an obstacle and the distance to which an individual may fall-back downstream following a failed attempt. In this study, when a fish failed in its passage attempt, the fish moved back downstream to suitable resting locations (fall-back), and though fall-back distance was not significant in determining a subsequent successful passage attempt, it does emphasise the cost of delay.
The fact that higher search rates (zone per unit time) at the weir face and a lower fish fat content were significant factors in predicting fish passage on an initial attempt, implies that fish with lower energy reserves cannot afford to be delayed at an obstacle and thus may increase energy expended in a single passage attempt. Obstacles directly increase energy consumption, thus fish with low energy reserves may need to reach spawning locations rapidly so as to rest and preserve remaining reserves for spawning. Although the effect of increased energy expenditure on reproductive success remains unknown, it would not be unreasonable to hypothesise that reduced energy reserves will ultimately have a negative impact on reproductive success (Thorstad et al., 2008).
Significant preference for passage at Zone 4 was shown throughout the study, yet this is potentially the most physically challenging passage route available. It is the shortest channel in length (upstream to downstream distance) of the weir (Fig. 3A); however, it requires a leap onto the weir face removing any potential momentum gain, followed by a swim up a steep gradient on the weir for 13 meters to gain passage. Following guidelines on available obstacle porosity measurements outlined by a water framework barrier classification tool (SNIFFER, 2010), such a channel would be deemed impassable to adult Atlantic salmon by the assessment protocol. Barrier assessment remains a challenge to fish biologists; however, it may be possible to identify if a barrier effects fish migration through changes in their migration behaviour. A greater number of attempts were initiated during daylight as opposed to during the night; however, there was no difference in light conditions between successful and unsuccessful attempts, suggesting that a greater success for attempts under dark conditions. In salmonids, the relationship between light intensity and passage attempts at obstacles is not clearly defined. At large complex obstacles, where fish are delayed and their migration thwarted, passage occurs primarily during daylight (Chanseau & Larinier, 1999; Chanseau et al., 1999; Gowans et al., 1999, 2003; Null & Niemela, 2011), whilst at less complex structures and natural by-pass channels passage generally occurs at night (Dunkley & Shearer, 1989; Chanseau et al., 1999). Light intensity preference for passage requirements appears to be site specific and related to the visual orientation needs at each given obstacle (Banks, 1969; Thorstad et al., 2008). The timing of passage (in either day or night) may be an early indicator of passage difficulty for salmon, with daytime passage potentially indicating higher levels of passage difficulty. Indeed there is likely to be an interaction between the light required to successfully ascend obstacles and the preference of turbid water or darkness as an anti-predator mechanism (Banks, 1969; Thorstad et al., 2008).
Surprisingly, fish in 2012 which were sampled from within the fish pass and subsequently released downstream did not attempt to re-ascend through the pass a second time, instead ascending across the weir face. It is possible that the initial passage attempt which ultimately resulted in failure due to capture and release downstream was a learning event which influenced subsequent passage attempts through a secondary route; the same phenomena were reported by Karpinnen et al. (2002). Studies involving the transfer of fish, in general, assume that the transportation does not affect subsequent migration behaviour.
For any given obstacle, there is a highly variable temporal window within which environmental variables such as flow and water temperature combine with fish characteristics to enable passage for an individual. This passage window fluctuates significantly from one individual to the next depending on their own physical features. There is ever growing evidence that riverine barriers are generating an anthropogenic selection pressure on anadromous salmonids. There is an urgent requirement to understand the costs and potential selection pressure associated with delay and passage success at an obstacle. The overarching impact of a delay remains unknown. Understanding the impact of a delay on the reproductive ability of an individual either through fewer eggs, smaller egg sizes, or eggs with lower nutrient availability is essential.
References
Agency, E., & Environment Agency, 2009. Response to DEFRA consultation: Modernisation of salmon and freshwater fisheries legislation; new regulatory order to address the passage of fish (for WFD and EU eel regulation).
Antonio, R. R., A. A. Agostinho, F. M. Pelicice, D. Bailly, E. K. Okada, J. Henrique & P. Dias, 2007. Blockage of migration routes by dam construction: can migratory fish find alternative routes? Neotropical Icthyology 5: 177–184.
Baguette, M., S. Blanchet, D. Legrand, V. M. Stevens & C. Turlure, 2013. Individual dispersal, landscape connectivity and ecological networks. Biological Reviews 88: 310–326.
Banks, J. W., 1969. A review of the literature on the upstream migration of adult salmonids. Journal of Fish Biology 1: 85–136.
Baras, E., H. Lambert & J. Philippart, 1994. A comprehensive assessment of the failure of Barbus barbus spawning migrations through a fish pass in the canalized River Meuse (Belgiurn). Aquatic Living Resources 7: 181–189.
Bardonnet, A. & J. Baglinière, 2000. Freshwater habitat of Atlantic salmon (Salmo salar). Canadian Journal of Fisheries and Aquatic Sciences 57: 497–506.
Bates, D., M. Maechler, B. Bolker, & S. Walker, 2015. Fitting linear mixed-effects models using lme4. Journal of Statistical Software 67: 1–48. doi:10.18637/jss.v067.i01.
Bivand, R., & N. Lewin-Koh, 2016. maptools: tools for reading and handling spatial objects. R package version 0.8-40. https://CRAN.R-project.org/package=maptools.
Branco, P., P. Segurado, J. M. Santos, P. Pinheiro & M. T. Ferreira, 2012. Does longitudinal connectivity loss affect the distribution of freshwater fish? Ecological Engineering 48: 70–78.
Bunt, C. M., T. Castro-Santos & A. Haro, 2012. Performance of fish passage structures at upstream barriers to migration. River Research and Applications 28: 457–478.
Burnham, K. P., & D. R. Anderson, 2002. Model selection and multimodel inference: a practical information-theoretic approach, 2nd edn. Springer, New York.
Calcagno, V., 2013. glmulti: model selection and multimodel inference made easy. R package version 1.0.7. https://CRAN.R-project.org/package=glmulti.
Calcagno, V. & C. Mazancourt, 2010. glmulti: an R Package for easy automated model selection with (generalized) linear models. Journal of Statistical Software 34: 1–29.
Ceballos, G. & P. R. Ehrlich, 2002. Mammal population losses and the extinction crisis. Science 296: 904–907.
Chanseau, M., & M. Larinier, 1999. The behaviour of returning adult Atlantic salmon (Salmo salar L.) in the vicinity of a hydroelectric plant on the Gave de Pau river (France) as determined by radiotelemetry In Moore, A., & I. Russell (eds), Advances in Fish Telemetry: 257–264.
Chanseau, M., O. Croze & M. Larinier, 1999. The impact of obstacles on the Pau River (France) on the upstream migration of returning adult Atlantic salmon (Salmo salar L.). Bulletin Francais De La Peche Et De La Pisciculture 353: 211–237.
Cooke, S. J. & S. G. Hinch, 2013. Improving the reliability of fishway attraction and passage efficiency estimates to inform fishway engineering, science, and practice. Ecological Engineering 58: 123–132.
Cooke, S. J., S. G. Hinch, A. P. Farrell, D. A. Patterson, K. Miller-Saunders, D. W. Welch, M. R. Donaldson, K. C. Hanson, G. T. Crossin, M. T. Mathes, A. G. Lotto, K. A. Hruska, I. C. Olsson, G. N. Wagner, R. Thomson, R. Hourston, K. K. English, S. Larsson, J. M. Shrimpton & G. Van der Kraak, 2008. Developing a mechanistic understanding of fish migrations by linking telemetry with physiology, behavior, genomics and experimental biology: an interdisciplinary case study on adult fraser river Sockeye Salmon. Fisheries 33: 321–339.
Cote, D., D. G. Kehler, C. Bourne & Y. F. Wiersma, 2009. A new measure of longitudinal connectivity for stream networks. Landscape Ecology 24: 101–113.
Dunkley, D. A. & W. M. M. Shearer, 1989. Swimming height of Atlantic salmon, Salmo salar L., crossing a Crump weir. Aquaculture and Fisheries Management 20: 193–198.
Fisher, R. & J. D. Hogan, 2007. Morphological predictors of swimming speed: a case study of pre-settlement juvenile coral reef fishes. Journal of Experimental Biology 210: 2436–2443.
Fox, John, 2003. Effect displays in R for generalised linear models. Journal of Statistical Software 8(15): 1–27. http://www.jstatsoft.org/v08/i15/
Fraser, D. J., L. K. Weir, L. Bernatchez, M. M. Hansen & E. B. Taylor, 2011. Extent and scale of local adaptation in salmonid fishes: review and meta-analysis. Heredity Nature Publishing Group 106: 404–420.
Gerlier, M. & P. Roche, 1998. A radio telemetry study of the migration of Atlantic salmon (Salmo salar L.) and sea trout (Salmo trutta trutta L.) in the upper Rhine. Hydrobiologia 371: 283–293.
Gowans, A. R. D., J. D. Armstrong & I. G. Priede, 1999. Movements of adult Atlantic salmon in relation to a hydroelectric dam and fish ladder. Journal of Fish Biology 44: 713–726.
Gowans, A. R. D., J. D. Armstrong, I. J. Priede & S. Mckelvey, 2003. Movements of Atlantic salmon migrating upstream through a fish-pass complex in Scotland. Ecology of Freshwater Fish 12: 177–189.
Guiny, E., D. A. Ervine & J. D. Armstrong, 2005. Hydraulic and biological aspects of fish passes for Atlantic salmon. Journal of Hydraulic Engineering 131: 542–553.
Haugen, T. O., P. Aass, N. C. Stenseth & L. A. Vøllestad, 2008. Changes in selection and evolutionary responses in migratory brown trout following the construction of a fish ladder. Evolutionary Applications 1: 319–335.
Jager, H. I., J. A. Chandler, K. B. Lepla & W. Van Winkle, 2001. A theoretical study of river fragmentation by dams and its effects on white sturgeon populations. Environmental Biology of Fishes 60: 347–361.
Karppinen, P., T. S. Mäkinen, J. Erkinaro, V. V. Kostin & R. V. Sadkovskij, 2002. Migratory and route-seeking behaviour of ascending Atlantic salmon in the regulated River Tuloma. Hydrobiologia 483: 23–30.
Kemp, P. S. & J. R. O’Hanley, 2010. Procedures for evaluating and prioritising the removal of fish passage barriers: a synthesis. Fisheries Management and Ecology 17: 297–322.
Kemp, P. S., I. J. Russon, B. Waterson, J. O. Hanley & G. R. Pess, 2008. Recommendations for a “coarse-resolution rapid-assessment” methodology to assess barriers to fish migration, and associated prioritization tools. International Centre for Ecohydraulic Research, Southampton.
Kinnison, M. T., M. J. Unwin, A. P. Hendry, & T. P. Quinn, 2016. Migratory Costs and the Evolution of Egg Size and Number in Introduced and Indigenous Salmon Populations, Vol. 55. Society for the Study of Evolution: 1656–1667.
Kristinsson, K. Ó., G. Gudbergsson & G. M. Gislason, 2015. Variable migration and delay in two stock components of an Atlantic salmon population. Environmental Biology of Fishes 98: 1513–1523.
Larinier, M., 1998. Upstream and downstream fish passage experience in France. Fish migration and fish bypasses. Fishing News Books Ltd, Farnham.
Lucas, M. C. & P. A. Frear, 1997. Effects of a flow-gauging weir on the migratory behaviour of adult barbel, a riverine cyprinid. Journal of Fish Biology 50: 382–396.
Lundqvist, H., P. Rivinoja, K. Leonardsson & S. McKinnell, 2008. Upstream passage problems for wild Atlantic salmon (Salmo salar L.) in a regulated river and its effect on the population. Hydrobiologia 602: 111–127.
Makiguchi, Y., Y. Konno, K. Konishi, K. Miyoshi, T. Sakashita, H. Nii, K. Nakao & H. Ueda, 2011. EMG telemetry studies on upstream migration of chum salmon in the Toyohira River, Hokkaido, Japan. Fish Physiology and Biochemistry 37: 273–284.
Meixler, M. S., M. B. Bain & M. T. Walter, 2009. Predicting barrier passage and habitat suitability for migratory fish species. Journal of Ecological Modelling 220: 2782–2791.
Mesa, M. G. & C. D. Magie, 2006. Evaluation of energy expenditure in adult spring Chinook salmon migrating upstream in the Columbia river basin: an assessment based on sequential proximate analysis. River Research and Applications 22: 1085–1095.
Mills, D., 1989. Ecology and management of Atlantic salmon. Chapman and Hall, London.
Naughton, G. P., C. C. Caudill, M. L. Keefer, T. C. Bjornn, L. C. Stuehrenberg & C. A. Peery, 2005. Late-season mortality during migration of radio-tagged adult sockeye salmon (Oncorhynchus nerka) in the Columbia River. Canadian Journal of Fisheries and Aquatic Sciences 62: 30–47.
Noonan, M. J., J. W. A. Grant & C. D. Jackson, 2012. A quantitative assessment of fish passage efficiency. Fish and Fisheries 13: 450–464.
Null, R., & K. Niemela, 2011. Monitoring and evaluation of the modified fish ladder and barrier weir at the Coleman National Fish Hatchery.
O’Connor, J. P., D. J. O’Mahony, J. M. O’Mahony & T. J. Glenane, 2006. Some impacts of low and medium head weirs on downstream fish movement in the Murray Darling Basin in southeastern Australia. Ecology of Freshwater Fish 15: 419–427.
O’Hanley, J. R. & D. Tomberlin, 2005. Optimizing the removal of small fish passage barriers. Environmental Modeling & Assessment 10: 85–98.
Ovidio, M. & J. Philippart, 2002. The impact of small physical obstacles on upstream movements of six species of fish. Hydrobiologia 483: 55–69.
R Core Team, 2013. R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/.
Ralph, A. L., B. I. Berli, P. Burkhardt-Holm & K. B. Tierney, 2012. Variability in swimming performance and underlying physiology in rainbow trout (Oncorhynchus mykiss) and brown trout (Salmo trutta). Comparative Biochemistry and Physiology Part A: Molecular & Integrative Physiology 163: 350–356.
Sigourney, D. B., J. D. Zydlewski, E. Hughes & O. Cox, 2015. Transport, Dam Passage, and Size Selection of Adult Atlantic Salmon in the Penobscot River, Maine. North American Journal of Fisheries Management 35: 1164–1176.
Silva, A. T., J. M. Santos, M. T. Ferreira, A. N. Pinheiro & Katopo, 2011. Effects of water velocity and turbulence on the behaviour of Iberian barbel (Luciobarbus bocagei, Steindachner 1864) in an experimental pool-type fishway. River Research and Applications 27: 360–373.
SNIFFER, 2010. Trialling of the methodology for quantifying the impacts of obstacles to fish passage.
Symonds, M. R. E., & A. Moussalli, 2011. A brief guide to model selection, multimodel inference and model averaging in behavioural ecology using Akaike's information criterion. Behavoural Ecology Sociolbiology 65: 13–21.
Thorstad, E. B., F. Økland, K. Aarestrup & T. G. Heggberget, 2008. Factors affecting the within-river spawning migration of Atlantic salmon, with emphasis on human impacts. Reviews in Fish Biology and Fisheries 18: 345–371.
Webb, J., 1990. The Behaviour of Adult Atlantic Salmon Ascending the Rivers Tay and Tummel to Pitlochry Dam. Scottish Fisheries Research Report Number 48, 1990. Department of Agriculture and Fisheries for Scotland in Association with the Atlantic Salmon Trust.
Webb, P. W., 1973. Effects of partial caudal-fin amputation on the kinematics and metabolic rate of underyearling sockeye salmon (Oncorhynchus nerka) at steady swimming speeds. Journal of Experimental Biology 59: 565–581.
Webb, P. W., P. T. Kostecki & E. D. Stevens, 1984. The effect of size and swimming speed on locomotor kinematics of rainbow trout. Journal of Experimental Biology 109: 77–95.
Weihs, D., 1973. The mechanism of rapid starting of slender fish. Biorheology 10: 343–350.
Acknowledgements
Funding was provided by INTERREG IVA (Grant No. Project 2859 'IBIS').
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling editor: Michael Power
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Newton, M., Dodd, J.A., Barry, J. et al. The impact of a small-scale riverine obstacle on the upstream migration of Atlantic Salmon. Hydrobiologia 806, 251–264 (2018). https://doi.org/10.1007/s10750-017-3364-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10750-017-3364-3