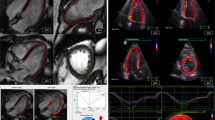
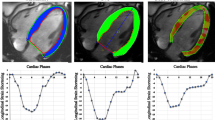
Abstract
Feature-tracking cardiac magnetic resonance (FT-CMR), with the ability to quantify myocardial deformation, has a unique role in the evaluation of subclinical myocardial abnormalities. This review aimed to evaluate the clinical use of cardiac FT-CMR-based myocardial strain in patients with various systemic diseases with cardiac involvement, such as hypertension, diabetes, cancer-therapy-related toxicities, amyloidosis, systemic scleroderma, myopathies, rheumatoid arthritis, thalassemia major, and coronavirus disease 2019 (COVID-19). We concluded that FT-CMR-derived strain can improve the accuracy of risk stratification and predict cardiac outcomes in patients with systemic diseases prior to symptomatic cardiac dysfunction. Furthermore, FT-CMR is particularly useful for patients with diseases or conditions which are associated with subtle myocardial dysfunction that may not be accurately detected with traditional methods. Compared to patients with cardiovascular diseases, patients with systemic diseases are less likely to undergo regular cardiovascular imaging to detect cardiac defects, whereas cardiac involvement in these patients can lead to major adverse outcomes; hence, the importance of cardiac imaging modalities might be underestimated in this group of patients. In this review, we gathered currently available data on the newly introduced role of FT-CMR in the diagnosis and prognosis of various systemic conditions. Further research is needed to define reference values and establish the role of this sensitive imaging modality, as a robust marker in predicting outcomes across a wide spectrum of patients.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Cardiovascular magnetic resonance (CMR) enables the accurate, noninvasive, and comprehensive characterization of cardiac tissue structure and function [1]. Feature tracking CMR (FT-CMR) imaging is a novel post-processing technique used to assess global and regional strains as parameters of myocardial deformation which can be encoded from routinely-acquired CMR cine images [2]. As a gage of local and overall ventricular function, strain is defined as local or global myocardial shortening, thickening, and lengthening. Longitudinal (LS) and circumferential strain (CS) are indicated by negative numbers representing the myocardial shortening in their respective orientation, while radial strain (RS) is reported as a positive number reflecting the myocardial thickening [3].
The advantage of using strain parameters over ejection fraction (EF) is that EF is not significantly reduced only after irreparable impairments at late stages of cardiac involvements and cannot demonstrate regional deformations. In ischemic heart diseases, FT-CMR integration as a diagnostic and prognostic modality has been extensively studied. This is the case for some of the non-ischemic heart diseases, such as myocarditis and cardiomyopathies as well [4].
Systemic diseases, as diseases that impact the body entirely instead of a specific organ or tissue, are occasionally accompanying cardiac involvements that can lead to adverse outcomes. The early diagnosis of these cardiac involvements can therefore improve their clinical outcomes. Cardiac inspections using sensitive imaging modalities can investigate the effect of different therapeutic agents administered in these patients on the cardiac tissue, as well. Current heart failure guidelines suggest CMR in infiltrative diseases, Fabry disease (FD), and myocarditis in order to investigate the myocardial tissue lesions [5]. However, still less is known on the effectiveness of FT-CMR implementation in the management of patients with systemic diseases who are prone to developing cardiac impairments as a result of their disease. In this review, we aimed to comprehensively review the available literature on the role of FT-CMR in systemic diseases, where its performance in cardiovascular diagnostics and outcome evaluation, was studied.
Hypertension
CMR-encoded parameters have been studied to evaluate cardiovascular risk, especially in high-risk individuals with underlying comorbidities such as hypertension and diabetes. In a study investigating the heart structural and functional differences between healthy and hypertensive participants, the feature-tracking-derived LS and CS were both significantly lower in the latter despite the controlled blood pressure in both groups of participants [6]. However, LS and CS were unable to depict a significant difference between hypertensive patients with or without coronary artery disease (CAD). LS and CS were also significantly lower in another study comparing a group of patients with both hypertension and diabetes to only hypertension and the control groups [7].
FT-CMR can also be implemented to evaluate the cardiac functional response to anti-hypertensive medications in a shorter period after receiving the treatment compared to echocardiography. For instance, a cohort study demonstrated a significant reduction in radial strain (RS) after 18 weeks of treatment. Not only this period was shorter than the earliest structural and functional changes detected by echocardiography (24 weeks), but FT-CMR was also able to detect significant RS (short axis) and CS changes, in agreement with reduced LVEDV and LV radial thickness as a consequent of reduced LVH [8]. Global LS (GLS) along with CMR-obtained lateral mitral annular plane systolic excursion (MAPSE) have been identified as independent predictors of mortality in patients with hypertension [9].
Detecting and treating left ventricular hypertrophy (LVH) in hypertension is critical for lowering cardiovascular risk. In a Spanish study, nearly one-quarter of hypertensive patients who visited outpatient clinics had LVH [10]. This number adds up to more than 30% in countries like India or Nigeria [11, 12]. Given the clinical significance of LVH in hypertensive patients, this constitutes a significant proportion of the population of hypertensive patients. Myocardial strain can trace cardiac tissue damage early and can be conveniently utilized for preventive strategies. FT-CMR in hypertensive patients may detect LA reservoir and conduit dysfunction early, even prior to the emergence of LVH [13, 14]. As a result, it appears that parameters of LA function, particularly conduit and reservoir function, are more powerful predictors of LA impairment in hypertension than LA structural indicators. Moreover, even before the drop in EF, GLS, global RS (GRS), and global CS (GCS) have been shown to be significantly worse and independently associated with mean arterial pressure and LV mass index in hypertensive patients with LVH [15,16,17]. Mirmojarabian et al. also introduced TRAFF2 as a marker of GLS, myocardial mass, and diameter prior to irreversible fibrosis [15].
Strain parameters as indicators of LV function can also be implemented to evaluate the cardiac response to anti-hypertensive medications in a shorter period of time after receiving the treatment compared to echocardiography. Furthermore, they are claimed to be subclinical markers of LVH, a critical condition associated with a high cardiovascular risk, in hypertension. Myocardial strain can trace cardiac tissue damage early and may detect LA dysfunction before LVH development and irreversible damage.
Diabetes
In type 2 diabetes mellitus, (DM) patients with subclinical right ventricle (RV) EF reduction, FT-CMR strains can quantify right ventricular deformation, with GLPS being the most accurate indicator of RV dysfunction [18], regardless of accompanying hypertension [19]. GLS has also been demonstrated to be the most affected strain by DM in heart failure patients with reduced EF (HFrEF) [20]. The reduction in the left atrium (LA) global strains has been also reported in diabetic patients compared to the controls, even before the appearance of the LV strain impairments. LA strain defects could be a result of LA remodeling and attenuated compliance following chronic inflammation and fibrosis. In a group of patients with both hypertension and diabetes, the LA strain, compared to the diabetes group, was significantly improved. This finding was either attributed to the compensatory effect of hypertension itself or the anti-hypertensive drugs [19]. Interestingly, higher BMI is reported to independently affect LA strains of reservoir and conduit [21]. This demonstrates the higher cardiovascular risk of cardiac dysfunction in type 2 DM patients with obesity.
In addition, LA GCS, as a parameter of systolic subepicardial contractability, was negatively correlated with microalbuminuria in type 2 diabetic patients [19]. Similar to hypertensive patients, LA reservoir and conduit strains, along with GLS, were reported to be early markers of major adverse cardiovascular events (MACE) in diabetic patients undergoing acute myocardial infarction (AMI), prior to a significant reduction in EF [22]. Increased HbA1c levels were also related to lower global circumferential and longitudinal peak diastolic strain rate (PDSR) in pre-diabetic and diabetic patients. Global circumferential and global longitudinal PDSR have been suggested to differentiate patients with diabetes from pre-diabetic patients, and the latter from the healthy controls [23].
As a chronic disease accompanying constant elevated inflammation, diabetes has been demonstrated to be associated with some major changes in functional cardiac parameters early in LA and eventually LV and RV. FT-CMR can be used as a helpful tool to stratify the cardiovascular risk or to monitor the treatment-related risk of adverse cardiac remodeling [24]. CMR-encoded strains along with late gadolinium enhancement (LGE) have also been used as a novel screening tool for the detection of silent MI in type 2 diabetes, outperforming ECG criteria in terms of accuracy [25].
Cancer therapy-related cardiotoxicity
Attempts to minimize cancer-therapy-related cardiac dysfunction (CTRCD) are focused on the early detection and treatment of LV dysfunction. Evidence implies that imaging modalities, such as novel echocardiography techniques or CMR, might be more efficient for the early diagnosis of CTRCD. Mostly because subclinical myocardial dysfunction can barely be indicated by LVEF reduction. Moreover, recovery of CTRCD necessitates early diagnosis and its possibility progressively declines after the establishment of myocardial damage and LV dysfunction [26]. Echocardiography might provide additional tools to assess cardio-toxic effects beyond left ventricular ejection fraction [27]. However, CMR-assessed strain as a rapid and more reproducible method of LV function monitoring can facilitate and improve the accuracy in the detection of cardiotoxicities in cancer patients receiving chemotherapies, namely anthracyclines, and trastuzumab, in a dose-dependent and non-dose-dependent manner, respectively [28]. Utilization as a routine part of cancer patients’ or survivors’ monitoring is under investigation, and only used in cases of discrepancies [27]. One of the most investigated CMR strain applications is in the early detection of trastuzumab-related cardiotoxicities in breast cancer.
Of 42 patients with human epidermal growth factor receptor 2 (HER2 +) breast cancer, 10 patients experienced trastuzumab-mediated cardiomyopathy within 2 years of follow-up. Although cardiac biomarkers’ levels such as troponin T, C-reactive protein, and brain natriuretic peptide were intact in patients experiencing cardiotoxicity, lateral S’ obtained by tissue velocity, and peak GLS and RS measured by FT-CMR were significantly changed within 3 months. LVEF decreased only after 6 months of follow-up in all patients with cardiomyopathy [29]. Although the results of this study recommend tissue velocity and CMR strain as subclinical markers of cardiotoxicity development, the need for further studies with more enrolled subjects and a longer period of follow-up were still unmet. In a later study, 125 patients with HER2 + early breast cancer receiving a sequential regimen of anthracycline/trastuzumab, and the association of two-dimensional echocardiography (2DE) and CMR-derived parameters with anticancer-related cardiotoxicities was evaluated. The patients were monitored before and during the treatment. During the follow-up period 28% of patients by CMR and 22% according to 2DE evaluation, developed cardiotoxicities. The odds ratio (OR) for cardiotoxicity development was the highest for 2DE-GLS (133% odds for a 15% reduction in 2DE-GLS), then for FT-CMR GCS (87%), and tagged-CMR GLS and GCS (47% and 50%, respectively), suggesting the 2DE-GLS as the optimal parameter and the superiority of 2DE-GLS compared to FT-CMR for early diagnosis of cardiotoxicity [30]. However, given the higher reproducibility and its ability to detect subclinical changes more accurately, FT-CMR is suggested to be used as a confirmatory modality in the diagnosis of CTRCD.
Ong et al. reported the correlation of temporal changes in LVEF with 6 months and 12 months of therapy with trastuzumab and FT-CMR-encoded GLS (r = −0.60, P < 0.001) and GCS (r = −0.75, P < 0.001). The changes in GLS and GCS were subsequent to the reduction in LVEF within 6 and 12 months after the initiation of the treatment. Six months after termination of the therapy, no significant change in LVEF, as well as GLS and GCS, was detected. The study was the first one to investigate temporal changes in FT-CMR-encoded parameters. The small size of the cohort, absence of patients with cardiovascular risk factors, and severe cardiotoxicities were the limitations of the study [31]. It seems that systolic strain rates by FT-CMR are likely more relevant than diastolic strain rates for tracking subclinical trastuzumab-related myocardial impairment, because, in a study on trastuzumab-treated cancer patients diastolic strain rates remained stable for 18 months following therapy beginning [32]. However, global diastolic strain rates measured with FT-CMR were found to be diminished late after anthracycline treatment as indicators of systolic dysfunction in a dose-dependent manner [33].
Amyloidosis
Amyloidosis is a heterogeneous systemic condition that results from the extracellular accumulation of insoluble protein in several organs [34]. Intra-myocardial amyloid infiltration leading to cardiac amyloidosis usually has a progressive course and is one of the primary reasons for mortality in affected patients [35]. The gold standard method to diagnose cardiac amyloidosis is the histopathological evaluation on an endo-myocardial biopsy, which is an invasive method and in some cases, impractical. Hence, CMR as a noninvasive imaging modality with adequate accuracy to depict functional and structural cardiac changes could be an optimal option for the diagnosis of cardiac amyloidosis, particularly with LGE imaging [36]. The application of FT in the diagnosis and risk association of cardiac amyloidosis has been studied as well.
To evaluate FT-CMR utilization to detect changes in myocardial amyloidosis, Pandey et al. studied 28 patients with a history of myocardial amyloidosis and 35 controls. They observed substantial reductions in longitudinal, circumferential, and radial stresses in cardiac amyloidosis patients and demonstrated that FT-CMR-encoded strains, for instance, RS, can differentiate patients with cardiac amyloidosis with high sensitivity (82.5%) and specificity (82.9%). The peak radial and circumferential strains are particularly affected at the basal layer due to the noteworthy changes in regional and global strain characteristics caused by myocardial amyloidosis [37]. Accordingly, the radial and circumferential strain ratio of apex to base can be utilized to differentiate cardiac amyloidosis from hypertrophic cardiomyopathy with a similar diagnostic accuracy compared to LGE [38].
In another study, Oda et al. evaluated 61 patients with systemic amyloidosis of whom 48 were LGE-positive and 13 were LGE-negative. They found out that the peak CS was significantly higher in the LGE-positive than in the LGE-negative patients (−13.3 ± 1.4% vs. −9.5 ± 2.3, P < 0.01). In contrast, the LGE-positive patients’ variability in the peak CS time was considerably higher than that of the LGE-negative patients’ (46.1 ± 24.5 vs. 21.2 ± 20.1 ms, P < 0.01). The diagnostic model incorporating CS variables has a sensitivity of 93.8%, a specificity of 76.9%, and an accuracy of 90.2% for detecting LGE-positive amyloidosis. Finally, they indicated that myocardial CMR strain was boosted to identify amyloidosis with LGE positivity without the use of a contrast agent. For the early identification of myocardial amyloid deposition, the peak CS and variation in the peak CS time may be more sensitive than LGE imaging, correlating with the severity of cardiac amyloid deposition in amyloidosis [39]. Another study from China assessed the feasibility of the FT-CMR algorithm to evaluate LV myocardial deformation in amyloid light-chain cardiac amyloidosis (AL-CA). Patients with AL-CA had significantly reduced global peak strain (PS) (longitudinal, circumferential, and radial) (P 0.05), while individuals with normal LVEF exhibited preserved longitudinal PS at the apical and significantly diminished longitudinal PS at the mid and basal segments [40]. Global strains were also reported to be predictors of all-cause mortality when integrated with a staging system based on cardiac biomarker levels, such as cardiac troponin T and N-terminal pro-brain natriuretic peptide [41]. As LA remodeling and dysfunction are widespread cardiac tissue changes in cardiac amyloidosis, LA reservoir strain has also been reported to be correlated with mortality in these patients [42].
FT-CMR could detect aberrant LV myocardial deformation in patients with cardiac amyloidosis, even in those with intact LVEF or negative LGE with acceptable diagnostic accuracy. Myocardial CMR strain can be applied to differentiate LGE-positive amyloidosis without the necessity for contrast medium injection. Moreover, significant reductions in GLS can differentiate CA patients in comparison to patients with other causes of LVH [43]. FT strains have been also investigated as markers of prognosis. Studies with a longer follow-up period can determine the prognostic role of FT-CMR-obtained strain in cardiac amyloidosis.
Rheumatic diseases
Rheumatoid arthritis (RA)
Increased inflammation in rheumatoid arthritis is linked with an increased cardiovascular risk [44]. Special initiatives should therefore be performed to detect and monitor myocardial deformities and functional defects in these patients. In addition to the diagnosis of subclinical cardiac functional defects, FT-CMR can be utilized to inspect the effect of different treatments on subclinical cardiac dysfunction in rheumatoid arthritis [45].
In subjects with no clinically manifested cardiac damage, rheumatoid arthritis is associated with an impaired LV systolic function according to the FT-CMR assessed LV strains, namely peak GLS and GCS in patients with no symptomatic cardiac disease. The interventricular septum and left ventricular lateral wall segments had significantly lower strains in rheumatoid arthritis patients compared to controls, demonstrating segmental strain impairments as well as global impairments. Interestingly, the administration of steroids might have protective cardiac effects in rheumatoid arthritis patients, as the peak GCS was significantly higher in patients using steroids than in patients who do not [46] biological disease-modifying anti-rheumatic drugs (DMARDs) have been reported to ameliorate the asymptomatic cardiac dysfunction, manifested by a significantly higher GCS in patients receiving such agents [45].
Due to the accuracy of FT-CMR in identifying the early cardiac manifestations of rheumatic diseases, its application has rarely been investigated in other chronic rheumatic diseases, such as Sjögren’s disease, as well [47]. Further studies could be of benefit to provide a comprehensive view of the strain changes and determine the clinical application of FT-CMR, especially in chronic conditions such as systemic lupus erythematosus which can lead to variable and heterogeneous manifestations, cardiac manifestations included [48].
Systemic scleroderma (SSc)
Systemic scleroderma (SSc) is a rare connective tissue disease characterized by skin and internal organ fibrosis. The heart is frequently involved in SSc. Although most of the affected patients are initially asymptomatic, myocardial deformations such as fibrosis and ventricular dysfunction can be detected in these patients, before the clinical manifestations appear. CMR-assessed parameters of diffuse tissue fibrosis (i.e., native T1 and extra-cellular volume) seem to be more capable in early detection of subclinical myocardial involvement in SSc patients than strain parameters or even indicators of focal fibrosis (i.e., LGE) [49, 50]. FT-CMR-obtained strains, as parameters of subclinical cardiac dysfunction, along with other parameters of cardiac remodeling can be implemented to identify serum markers of cardiac involvement, such as galectin 3, growth differentiation factor 15 (GDF-15), and N-terminal pro-brain natriuretic peptide, in SSc patients [51].
Cardiac involvement is identified as the leading cause of death in patients with SSc [52], of which pulmonary arterial hypertension (PAH) is a leading cause with a very poor prognosis [53]. Echocardiography is widely used in the clinic to screen and detect SSc patients for cardiac complications [54]. PAH is defined by a mean pulmonary artery pressure > 25 mm Hg at rest. Although right heart catheterization has remained the standard approach for PH diagnosis, there is still an unmet need for noninvasive methods to recognize subclinical patients, and also investigate the presence of left heart disease in this group of patients [55]. Novel imaging methods such as CMR can play an essential role in detecting subclinical cardiac involvements, but their clinical value in this group of patients is still under scrutiny [56].
To evaluate the clinical relevance of FT-CMR in detecting the cardiac implications of SS, 44 SSc patients with no cardiac symptoms, a median of 3.6 years after their diagnosis, were compared with a group of 21 age- and gender-matched controls. SSc patients had a higher LVEF (62 ± 6% vs 59 ± 5%, P = 0.01), and heart rate than controls. 40 patients underwent LGE assessment, of which 8% showed evidence of subendocardial infarction and 18% had RV insertion fibrosis. LV longitudinal strain was significantly worse in patients with insertion fibrosis (−18.0 ± 2.3) and infarction (−19.6 ± 3.3) compared to LGE-patients (−20.3 ± 3.2, P = 0.04). Other than RV longitudinal strain (SS: −27.0 ± 4.0% vs control: −28.2 ± 2.0, P = 0.01), SSc patients demonstrated no difference in radial, circumferential, or longitudinal indices in comparison with healthy controls [57]. Taken together, the results of this study suggest that FT-CMR, namely LV LS can be used in detecting cardiac involvements such as infarction and fibrosis in SSc patients in the absence of a reduction in EF.
LV and RV free wall GLS can non-invasively be utilized to differentiate SSc patients with PAH from other SSc patients with promising accuracy (area under the curve (AUC): 0.73 and 0.86 respectively) [58]. However, a later study claimed that not RV LS, but right atrium conduit strain is a reassuring prognostic marker of mortality in SSc patients and is significantly correlated with the pulmonary arterial pressure and diffusing capacity of the lungs for carbon monoxide [59]. LA reservoir strain has been also reported to be an independent determinant of all-cause mortality and New York Heart Association (NYHA) class II-IV heart failure symptoms [60].
There are discrepancies in the literature on whether different strain values encoded by FT-CMR can identify subclinical cardiac involvement in SSc patients [49, 58, 61]. This could be a result of different strain parameters assessment or different recruited populations of patients. Data support the promising role of FT-CMR, especially atrial strains, in determining the prognosis in this challenging and high-risk group of patients. Consequently, FT-CMR can be rendered as a reliable, noninvasive method to assess the risk of mortality or PAH development in SSc patients so that they can receive adequate care. Moreover, FT-CMR can be utilized to monitor the improvements in cardiac function after the administration of therapeutic regimens [62].
Myopathies
Duchenne muscular dystrophy
Patients with Duchenne muscular dystrophy (DMD) require regular imaging to monitor heart function and the likelihood of DMD-associated cardiomyopathy (DMDAC). Poor image windows may restrict echocardiography’s diagnostic value. When the optimal techniques of LV function evaluation by echocardiography, namely two-dimensional fractional shortening and 5/6 area-length LVEF are not assessable or when the detection of more subtle myocardial alterations is required, CMR should be regarded as the optimal technique of LV function measurement [63, 64]. FT-CMR may detect early changes in the myocardial tissue without a need for gadolinium contrast. Furthermore, compared to the healthy subjects, FT-CMR could detect cardiac abnormalities in areas where speckle-tracking echocardiography did not [65].
Hor et al. enrolled 77 DMD patients and 16 age-matched controls and assessed their CMR strain determined by HARP analysis. They demonstrated a declination trend in LV CS calculated by HARP in DMD patients by advancing age and reduction in LVEF [66]. Hor et al. conducted a further study with a larger population of patients (N = 191) and control subjects (N = 42). They sought to compare the accuracy of FT-CMR LV CS with CS derived from HARP-tagged image analysis, as a gold standard method. The LV CS determined by two methods demonstrated no significant difference within all 5 groups of DMD patients with different ages and EF, and within the control group and all DMD patients. They concluded that FT-CMR-encoded strain can be used as a rapid, accurate, and reproducible method of cardiac risk stratification in DMD patients [67]. CMR strain parameters can also be helpful to identify patients with DMDAC before its clinical development. In a study, 30 patients with DMDAC were diagnosed within a period of 3 years and were compared to 30 DMD patients without cardiomyopathy. The results suggest that GRS and GCS were both lower in the DMDAC group, with GCS having the best predictive value for identifying the patients who developed DMDAC within 3 years before symptomatic cardiac dysfunction [68]. GCS was also able to identify DMD patients with even preserved LV EF and the absence of LGE from controls, whereas GLS and GRS were able to do so in patients with positive LGE [69].
FT-derived strains can detect early myocardial abnormalities even without using a contrast agent or, at the very least, minimize the frequency of exposure to gadolinium-based contrast agents in DMD patients. Additionally, this modality requires reduced medical costs and shorter scan duration. Above that, CMR overcomes echocardiography’s normal imaging limitations regarding diagnostic accuracy in DMD patients. These strain measures could also be employed as sensitive biomarkers for the early initiation of DMDAC.
Fabry disease
FD is a progressive disease that affects multiple organs, specifically the kidneys, heart, and brain. The disease manifests primarily in hemizygous males, but heterozygous females may also develop disease-related complications, albeit with a delayed onset. Its cardiac manifestation mostly includes left ventricular hypertrophy [70].
CMR-obtained values can be used to evaluate cardiomyopathies in FD as well. Among 18 patients with FD, patients with LV hypertrophy had significantly higher CS compared to other patients (−13.5 \(\pm 5.0\%\) vs −18.7 \(\pm\) 2.7%, P = 0.042). The small sample size of the study limits its statistical power [71]. Base-to-apex CS gradient has also been reported to help identify FD patients before the development of LVH and LGE [72]. LA reservoir strain and LV GLS can also be implemented to detect early myocardial remodeling before symptomatic LVH [73,74,75].
Idiopathic inflammatory myopathy
Patients with idiopathic inflammatory myopathy (IIM) are at a high risk of developing mortal cardiac dysfunction [76]. Accordingly, detecting cardiac involvement in these patients can be of considerable benefit and is therefore necessary if patients at risk are to be identified prior to the appearance of cardiac symptoms. Global strain parameters make it plausible to detect high-risk IIM patients before the incidence of cardiac events, namely LVEF reduction, or tissue remodeling and fibrosis. LV and RV GLS and LA strains of reservoir and conduct could differentiate between IIM patients with or without LGE [77, 78]. In a follow-up period of about 8 months, besides the LVEF, all the global strain values were reduced significantly [77]. Consequently, this depicts the relatively rapid deterioration of myocardial deformation in IIM which further emphasizes the need for consistent screening for cardiac complications.
Thalassemia
Thalassemia is characterized by decreased or missing production of one of the globin chains in hemoglobin protein. Functional impairments in the myocardial tissue might be induced as a result of increased iron accumulation in the myocardial tissue even before alterations in the EF. Strain levels may manifest the deficiencies and help in the early recognition of the myocardial dysfunction to improve survival and prevent additional problems. Biventricular and RV GLS could distinguish thalassemia major patients in regards to the presence of myocardial iron overload and intermediately to highly probable pulmonary hypertension [79, 80]. CMR obtained T2 mapping is the gold standard parameter to assess the myocardial iron overload. Although there is inconsistency on which FT-CMR strains are best correlated with T2, RVGLS [79, 81], LV GLS and LV GCS [79, 82], and LV GRS [83, 84] have been reported to best correlate with T2 as a parameter of iron overload. These findings suggest that FT-CMR strains can accompany T2 assessment and provide an additional functional myocardial evaluation of thalassemia patients. Hence, strain parameters of cardiac deformation cannot replace T2 mapping, but they allow a more accurate estimation of cardiac function than parameters like LVEF, as they were reported to have a stronger correlation with T2 [82]. This applies to the prognostic role of strains in predicting the outcome, as well. Although the RV GLS could be a fair predictor of adverse cardiac events and mortality with 52.94% and 71.43% sensitivity and 90% specificity, RVEF provided the best prognostic accuracy in a median of 36 months of follow-up [85]. A longer follow-up period could albeit provide a more precise estimate of the prognostic accuracy of strains, as indicators of subclinical defects, compared to the EF, as it may take longer for events to happen in patients with subclinical deformations.
COVID-19
SARS-CoV-2 infection can lead to a wide range of systemic symptoms similar to those found in inflammatory or autoimmune diseases [86]. Cardiac involvement and an existent myocardial inflammation were reported to be detectable in 78% and 60% of patients who recovered from COVID-19, respectively [87]. Hence, there were concerns about cardiac involvement as a result of elongated coronavirus disease 2019 (COVID-19) systemic inflammation with myocarditis-like signs and symptoms or post-vaccination myocarditis. Most of the investigations were focused on CMR-assessed parameters of fibrosis and edema, demonstrating the emergent fibrotic lesions that mostly resolve in mid-term follow-up [88, 89]. Strains as indicators of the dynamics of the myocardium have been investigated as well. FT strains were only significantly altered in limited groups of patients. For instance, only in patients undergoing CMR less than 8 weeks after their COVID-19 infection compared to those who underwent 8 or more weeks after infection [90], or in LGE-positive patients [91, 92].
Altogether, it seems that strains are not identified as helpful monitoring tools in all COVID-19 patients with cardiac involvements, as the strain deformations seem to resolve in a relatively short period compared to fibrotic or edematic lesions. However, in LGE-positive patients with more extensive myocardial damage, the strains can detect impairments in the myocardial dynamics which might help with the understanding of the pathophysiology of the damage or management decisions.
Conclusion
The clinical application of FT-CMR can get wide-range not only in cardiology practice but also in the management of systemic diseases with susceptibility to cardiac involvements. In summary, FT-CMR has been used in studies to diagnose the myocardium’s subclinical and asymptomatic structural damage, identify high-risk patients, assess the cardiac impact of novel therapeutic agents, and elaborate on the pathophysiology of the cardiac manifestations in systemic diseases. More investigations are required to identify reference strain values, while these preliminary findings show that FT-CMR can be used to detect early cardiac manifestations of systemic diseases (Table 1). Moreover, strain-based parameters have been proposed as potential predictors of prognosis in patients with systemic diseases and are believed to be associated with the risk of future cardiovascular events (Table 2). The practicality of FT-CMR in decision-making will determine its definitive debut in clinical practice guidelines for systemic diseases. It has been widely accepted in the reviewed literature that the significant benefit of FT-CMR is its ability to detect early functional defects before symptomatic dysfunction or irreversible tissue damage in the heart with an outstanding spatial resolution. However, the precision and extent of the information provided would not be determinators for everyday clinical use. The demonstration that the additional time and cost put into the strain assessment from cine CMR is worth the additionally provided information compared to conventional modalities is necessary and needs further investigations.
Availability of data and materials
There is no additional data related to this manuscript.
Abbreviations
- CMR:
-
Cardiac magnetic resonance
- COVID-19:
-
Coronavirus disease 2019
- CS:
-
Circumferential strain
- DM:
-
Diabetes mellitus
- DMD:
-
Duchenne muscular dystrophy
- DMD-AC:
-
Duchenne muscular dystrophy-associated cardiomyopathy
- EF:
-
Ejection fraction
- FD:
-
Fabry disease
- FT:
-
Feature tracking
- GCS:
-
Global circumferential strain
- GLS:
-
Global longitudinal strain
- GRS:
-
Global radial strain
- LA:
-
Left Atrium
- LGE:
-
Late gadolinium enhancement
- LS:
-
Longitudinal strain
- LV:
-
Left ventricle
- NYHA:
-
New York Heart Association
- PAH:
-
Pulmonary arterial hypertension
- RA:
-
Right atrium
- RS:
-
Radial strain
- RV:
-
Right ventricle
- SSc:
-
Systemic scleroderma
References
Valbuena-Lopez S, Hinojar R, Puntmann VO (2016) Cardiovascular magnetic resonance in cardiology practice: a concise guide to image acquisition and clinical interpretation. Rev Esp Cardiol 69(2):202–210
Rahman Zu, Sethi P, Murtaza G, Virk HuH, Rai AB, Mahmod M et al (2017) Feature tracking cardiac magnetic resonance imaging: a review of a novel non-invasive cardiac imaging technique. World J Cardiol 9:312–9
Muser D, Castro SA, Santangeli P, Nucifora G (2018) Clinical applications of feature-tracking cardiac magnetic resonance imaging. World J Cardiol 10(11):210
Scatteia A, Baritussio A, Bucciarelli-Ducci C (2017) Strain imaging using cardiac magnetic resonance. Heart Fail Rev 22(4):465–476
Behnoush AH, Khalaji A, Naderi N, Ashraf H, von Haehling S (2022) ACC/AHA/HFSA 2022 and ESC 2021 guidelines on heart failure comparison. ESC Heart Failure n/a(n/a)
Homsi R, Kuetting D, Sprinkart A, Steinfeld N, Meier-Schroers M, Luetkens J et al (2017) Interrelations of epicardial fat volume, left ventricular T1-relaxation times and myocardial strain in hypertensive patients: a cardiac magnetic resonance study. J Thorac Imaging 32(3):169–175
Kropidlowski C, Meier-Schroers M, Kuetting D, Sprinkart A, Schild H, Thomas D et al (2020) CMR based measurement of aortic stiffness, epicardial fat, left ventricular myocardial strain and fibrosis in hypertensive patients. IJC Heart & Vasculature 27:100477
Jordan AN, Fulford J, Gooding K, Anning C, Wilkes L, Ball C et al (2021) Morphological and functional cardiac consequences of rapid hypertension treatment: a cohort study. J Cardiovasc Magn Reson 23(1):122
Romano S, Judd RM, Kim RJ, Kim HW, Heitner JF, Shah DJ et al (2019) Prognostic implications of mitral annular plane systolic excursion in patients with hypertension and a clinical indication for cardiac magnetic resonance imaging: a multicenter study. JACC Cardiovasc Imaging 12(9):1769–1779
Barrios V, Escobar C, Calderón A, Ribas L, MartÍ D, AsÍn E (2008) Prevalence of left ventricular hypertrophy detected by Cornell voltage-duration product in a hypertensive population. Blood Press 17(2):110–115
Murtala AN, Ojji DB, Hayatu U, Isezuo SA (2022) Prevalence of echocardiographic left ventricular hypertrophy among hypertensives in a tertiary health institution in Nigeria. Ann Med Res Pract
Yadav B, Kushwaha P (2022) Prevalence and determinants of left ventricular hypertrophy among elderly hypertensive patients attending tertiary health care center in West Bengal, India. Ann Geriatr Educ Med Sci 9:1–6
Li L, Chen X, Yin G, Yan W, Cui C, Cheng H et al (2020) Early detection of left atrial dysfunction assessed by CMR feature tracking in hypertensive patients. Eur Radiol 30(2):702–711
Song Y, Li L, Chen X, Shao X, Lu M, Cheng J et al (2022) Early left ventricular diastolic dysfunction and abnormal left ventricular-left atrial coupling in asymptomatic patients with hypertension: a cardiovascular magnetic resonance feature tracking study. J Thorac Imaging 37(1):26–33
Mirmojarabian SA, Lammentausta E, Liukkonen E, Ahvenjärvi L, Junttila J, Nieminen MT et al (2022) Myocardium assessment by relaxation along fictitious field, extracellular volume, feature tracking, and myocardial strain in hypertensive patients with left ventricular hypertrophy. Int J Biomed Imaging 2022:9198691
Neisius U, Myerson L, Fahmy AS, Nakamori S, El-Rewaidy H, Joshi G et al (2019) Cardiovascular magnetic resonance feature tracking strain analysis for discrimination between hypertensive heart disease and hypertrophic cardiomyopathy. PLoS One 14(8):e0221061
Liu H, Wang J, Pan Y, Ge Y, Guo Z, Zhao S (2020) Early and quantitative assessment of myocardial deformation in essential hypertension patients by using cardiovascular magnetic resonance feature tracking. Sci Rep 10(1):3582
Hu BY, Wang J, Yang ZG, Ren Y, Jiang L, Xie LJ et al (2019) Cardiac magnetic resonance feature tracking for quantifying right ventricular deformation in type 2 diabetes mellitus patients. Sci Rep 9(1):11148
Shao G, Cao Y, Cui Y, Han X, Liu J, Li Y et al (2020) Early detection of left atrial and bi-ventricular myocardial strain abnormalities by MRI feature tracking in normotensive or hypertensive T2DM patients with preserved LV function. BMC Cardiovasc Disord 20(1):196
Zhang G, Shi K, Yan WF, Li XM, Li Y, Guo YK et al (2022) Effects of diabetes mellitus on left ventricular function and remodeling in hypertensive patients with heart failure with reduced ejection fraction: assessment with 3.0 T MRI feature tracking. Cardiovasc Diabetol 21(1):69
Shen MT, Guo YK, Liu X, Ren Y, Jiang L, Xie LJ et al (2022) Impact of BMI on left atrial strain and abnormal atrioventricular interaction in patients with type 2 diabetes mellitus: a cardiac magnetic resonance feature tracking study. J Magn Reson Imaging 55(5):1461–1475
Backhaus SJ, Kowallick JT, Stiermaier T, Lange T, Navarra JL, Koschalka A et al (2020) Cardiac magnetic resonance myocardial feature tracking for optimized risk assessment after acute myocardial infarction in patients with type 2 diabetes. Diabetes 69(7):1540–1548
Zhou S, Zhang Z, Zhang Z, Gao Y, Li G, Lou M et al (2022) Evaluation of left ventricular systolic and diastolic function in subjects with prediabetes and diabetes using cardiovascular magnetic resonance-feature tracking. Acta Diabetol 59(4):491–499
Sandhu P, Ong JP, Garg V, Altaha M, Bello O, Singal SR et al (2021) The effects of saxagliptin on cardiac structure and function using cardiac MRI (SCARF). Acta Diabetol 58(5):633–641
Swoboda PP, McDiarmid AK, Erhayiem B, Haaf P, Kidambi A, Fent GJ et al (2016) A Novel and practical screening tool for the detection of silent myocardial infarction in patients with type 2 diabetes. J Clin Endocrinol Metab 101(9):3316–3323
Cardinale D, Colombo A, Bacchiani G, Tedeschi I, Meroni CA, Veglia F et al (2015) Early detection of anthracycline cardiotoxicity and improvement with heart failure therapy. Circulation 131(22):1981–1988
López-Fernández T, Thavendiranathan P (2017) Emerging cardiac imaging modalities for the early detection of cardiotoxicity due to anticancer therapies. Revista Española de Cardiología (English Edition) 70(6):487–495
Plana JC, Galderisi M, Barac A, Ewer MS, Ky B, Scherrer-Crosbie M et al (2014) Expert consensus for multimodality imaging evaluation of adult patients during and after cancer therapy: a report from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. J Am Soc Echocardiogr 27(9):911–939
Fallah-Rad N, Walker JR, Wassef A, Lytwyn M, Bohonis S, Fang T et al (2011) The utility of cardiac biomarkers, tissue velocity and strain imaging, and cardiac magnetic resonance imaging in predicting early left ventricular dysfunction in patients with human epidermal growth factor receptor ii–positive breast cancer treated with A. J Am Coll Cardiol 57(22):2263–2270
Houbois CP, Nolan M, Somerset E, Shalmon T, Esmaeilzadeh M, Lamacie MM et al (2021) Serial cardiovascular magnetic resonance strain measurements to identify cardiotoxicity in breast cancer: comparison with echocardiography. JACC: Cardiovasc Imaging 14(5):962–974
Ong G, Brezden-Masley C, Dhir V, Deva DP, Chan KKW, Chow C-M et al (2018) Myocardial strain imaging by cardiac magnetic resonance for detection of subclinical myocardial dysfunction in breast cancer patients receiving trastuzumab and chemotherapy. Int J Cardiol 261:228–233
Gong IY, Ong G, Brezden-Masley C, Dhir V, Deva DP, Chan KKW et al (2019) Early diastolic strain rate measurements by cardiac MRI in breast cancer patients treated with trastuzumab: a longitudinal study. Int J Cardiovasc Imaging 35(4):653–662
Barbosa MF, Fusco DR, Gaiolla RD, Werys K, Tanni SE, Fernandes RA et al (2021) Characterization of subclinical diastolic dysfunction by cardiac magnetic resonance feature-tracking in adult survivors of non-Hodgkin lymphoma treated with anthracyclines. BMC Cardiovasc Disord 21(1):170
Merlini G, Bellotti V (2003) Molecular mechanisms of amyloidosis. N Engl J Med 349(6):583–596
Banypersad SM, Moon JC, Whelan C, Hawkins PN, Wechalekar AD (2012) Updates in cardiac amyloidosis: a review. J Am Heart Assoc 1(2):e000364
vanden Driesen RI, Slaughter RE, Strugnell WE (2006) MR findings in cardiac amyloidosis. Am J Roentgenol 186(6):1682–5
Pandey T, Alapati S, Wadhwa V, Edupuganti MM, Gurram P, Lensing S et al (2017) Evaluation of myocardial strain in patients with amyloidosis using cardiac magnetic resonance feature tracking. Curr Probl Diagn Radiol 46(4):288–294
Jung HN, Kim SM, Lee JH, Kim Y, Lee S-C, Jeon E-S et al (2019) Comparison of tissue tracking assessment by cardiovascular magnetic resonance for cardiac amyloidosis and hypertrophic cardiomyopathy. Acta Radiol 61(7):885–893
Oda S, Utsunomiya D, Nakaura T, Yuki H, Kidoh M, Morita K et al (2017) Identification and assessment of cardiac amyloidosis by myocardial strain analysis of cardiac magnetic resonance imaging. Circ J 81(7):1014–1021
Li R, Yang Z-G, Xu H-Y, Shi K, Liu X, Diao K-Y et al (2017) Myocardial deformation in cardiac amyloid light-chain amyloidosis: assessed with 3T cardiovascular magnetic resonance feature tracking. Sci Rep 7(1):1–9
Illman JE, Arunachalam SP, Arani A, Chang IC-Y, Glockner JF, Dispenzieri A et al (2018) MRI feature tracking strain is prognostic for all-cause mortality in AL amyloidosis. Amyloid 25(2):101–8
Tan Z, Yang Y, Wu X, Li S, Li L, Zhong L et al (2022) Left atrial remodeling and the prognostic value of feature tracking derived left atrial strain in patients with light-chain amyloidosis: a cardiovascular magnetic resonance study. Int J Cardiovasc Imaging 38:1519–1532
Williams LK, Forero JF, Popovic ZB, Phelan D, Delgado D, Rakowski H et al (2017) Patterns of CMR measured longitudinal strain and its association with late gadolinium enhancement in patients with cardiac amyloidosis and its mimics. J Cardiovasc Magn Reson 19(1):61
Choy EHS, Ganeshalingam K, Semb AG, Szekanecz Z, Nurmohamed MT (2014) Cardiovascular risk in rheumatoid arthritis: recent advances in the understanding of the pivotal role of inflammation, risk predictors and the impact of treatment. Rheumatology (Oxford) 53:2143–2154
Yokoe I, Kobayashi H, Kobayashi Y, Nishiwaki A, Sugiyama K, Nagasawa Y et al (2020) Impact of biological treatment on left ventricular dysfunction determined by global circumferential, longitudinal and radial strain values using cardiac magnetic resonance imaging in patients with rheumatoid arthritis. Int J Rheum Dis 23(10):1363–1371
Tański W, Gać P, Chachaj A, Mazur G, Poręba R, Szuba A (2021) Left ventricular myocardial strain assessed by cardiac magnetic resonance feature tracking in patients with rheumatoid arthritis. Insights Imaging 12(1):5
Yokoe I, Kobayashi H, Nishiwaki A, Nagasawa Y, Kitamura N, Haraoka M et al (2021) Asymptomatic myocardial dysfunction was revealed by feature tracking cardiac magnetic resonance imaging in patients with primary Sjögren’s syndrome. Int J Rheum Dis 24(12):1482–1490
Luo S, Dou WQ, Schoepf UJ, Varga-Szemes A, Pridgen WT, Zhang LJ (2022) Cardiovascular magnetic resonance imaging in myocardial involvement of systemic lupus erythematosus. Trends Cardiovasc Med S1050-1738(22)00025-1
Gotschy A, Jordan S, Stoeck CT, von Deuster C, Peer T, Gastl M et al (2023) Diffuse myocardial fibrosis precedes subclinical functional myocardial impairment and provides prognostic information in systemic sclerosis. Eur Heart J Cardiovasc Imaging 24(3):373–382
Lee DC, Hinchcliff ME, Sarnari R, Stark MM, Lee J, Koloms K et al (2018) Diffuse cardiac fibrosis quantification in early systemic sclerosis by magnetic resonance imaging and correlation with skin fibrosis. J Scleroderma Relat Disord 3(2):159–169
Hromádka M, Seidlerová J, Suchý D, Rajdl D, Lhotský J, Ludvík J et al (2017) Myocardial fibrosis detected by magnetic resonance in systemic sclerosis patients - relationship with biochemical and echocardiography parameters. Int J Cardiol 249:448–453
Elhai M, Meune C, Boubaya M, Avouac J, Hachulla E, Balbir-Gurman A et al (2017) Mapping and predicting mortality from systemic sclerosis. Ann Rheum Dis 76(11):1897–1905
Steen VD, Medsger TA (2007) Changes in causes of death in systemic sclerosis, 1972–2002. Ann Rheum Dis 66(7):940–944
Tennøe AH, Murbræch K, Andreassen JC, Fretheim H, Garen T, Gude E et al (2018) Left ventricular diastolic dysfunction predicts mortality in patients with systemic sclerosis. J Am Coll Cardiol 72(15):1804–1813
Hoeper MM, Bogaard HJ, Condliffe R, Frantz R, Khanna D, Kurzyna M (eds) et al (2013) Definitions and diagnosis of pulmonary hypertension. 62(25 Suppl):D42–50
Smolenska Z, Barraclough R, Dorniak K, Szarmach A, Zdrojewski Z (2019) Cardiac involvement in systemic sclerosis: diagnostic tools and evaluation methods. Cardiol Rev 27(2):73–79
Bratis K, Lindholm A, Hesselstrand R, Arheden H, Karabela G, Stavropoulos E et al (2019) CMR feature tracking in cardiac asymptomatic systemic sclerosis: clinical implications. PLoS ONE 14(8):e0221021
Lindholm A, Hesselstrand R, Rådegran G, Arheden H, Ostenfeld E (2019) Decreased biventricular longitudinal strain in patients with systemic sclerosis is mainly caused by pulmonary hypertension and not by systemic sclerosis per se. Clin Physiol Funct Imaging 39(3):215–225
Vos JL, Butcher SC, Fortuni F, Galloo X, Rodwell L, Vonk MC et al (2022) The prognostic value of right atrial and right ventricular functional parameters in systemic sclerosis. Front Cardiovasc Med 9:845359
Butcher SC, Vos JL, Fortuni F, Galloo X, Liem SIE, Bax JJ et al (2023) Evaluation of left cardiac chamber function with cardiac magnetic resonance and association with outcome in patients with systemic sclerosis. Rheumatol (Oxford) 62(SI):SI20–SI31
Kobayashi Y, Kobayashi H, J TG, Yokoe I, Hirano M, Nakajima Y et al (2016) Detection of left ventricular regional dysfunction and myocardial abnormalities using complementary cardiac magnetic resonance imaging in patients with systemic sclerosis without cardiac symptoms: a pilot study. Intern Med 55(3):237–43
Sato T, Ambale-Venkatesh B, Lima JAC, Zimmerman SL, Tedford RJ, Fujii T et al (2018) The impact of ambrisentan and tadalafil upfront combination therapy on cardiac function in scleroderma associated pulmonary arterial hypertension patients: cardiac magnetic resonance feature tracking study. Pulm Circ 8(1):2045893217748307
Soslow JH, Xu M, Slaughter JC, Stanley M, Crum K, Markham LW et al (2016) Evaluation of echocardiographic measures of left ventricular function in patients with duchenne muscular dystrophy: assessment of reproducibility and comparison to cardiac magnetic resonance imaging. J Am Soc Echocardiogr: Official Publ Am Soc Echocardiogr 29(10):983–991
Buddhe S, Lewin M, Olson A, Ferguson M, Soriano BD (2016) Comparison of left ventricular function assessment between echocardiography and MRI in Duchenne muscular dystrophy. Pediatr Radiol 46(10):1399–1408
Siegel B, Olivieri LJ, Gordish-Dressman H, Spurney CF (2018) Myocardial strain using cardiac MR feature tracking and speckle tracking echocardiography in duchenne muscular dystrophy patients. Pediatr Cardiol 39:478–483
Hor KN, Wansapura J, Markham LW, Mazur W, Cripe LH, Fleck R et al (2009) Circumferential strain analysis identifies strata of cardiomyopathy in Duchenne muscular dystrophy: a cardiac magnetic resonance tagging study. J Am Coll Cardiol 53(14):1204–1210
Hor KN, Gottliebson WM, Carson C, Wash E, Cnota J, Fleck R et al (2010) Comparison of magnetic resonance feature tracking for strain calculation with harmonic phase imaging analysis. JACC: Cardiovasc Imaging 3(2):144–51
Siddiqui S, Alsaied T, Henson SE, Gandhi J, Patel P, Khoury P et al (2020) Left ventricular magnetic resonance imaging strain predicts the onset of duchenne muscular dystrophy–associated cardiomyopathy. Circulation: Cardiovasc Imaging 13(11):e011526
Panovský R, Pešl M, Máchal J, Holeček T, Feitová V, Juříková L et al (2021) Quantitative assessment of left ventricular longitudinal function and myocardial deformation in Duchenne muscular dystrophy patients. Orphanet J Rare Dis 16(1):57
Hagège AA (2010) Cardiac manifestations of Anderson-Fabry disease and efficacy of enzyme replacement therapy. La Revue de medecine interne 31(Suppl 2):S238–S242
Wilson HC, Ambach S, Madueme PC, Khoury PR, Hopkin RJ, Jefferies JL (2018) Comparison of native T1, strain, and traditional measures of cardiovascular structure and function by cardiac magnetic resonance imaging in patients with Anderson-Fabry disease. Am J Cardiol 122(6):1074–1078
Mathur S, Dreisbach JG, Karur GR, Iwanochko RM, Morel CF, Wasim S et al (2019) Loss of base-to-apex circumferential strain gradient assessed by cardiovascular magnetic resonance in Fabry disease: relationship to T1 mapping, late gadolinium enhancement and hypertrophy. J Cardiovasc Magn Reson 21(1):45
Halfmann MC, Altmann S, Schoepf UJ, Reichardt C, Hennermann JB, Kreitner KF et al (2022) Left atrial strain correlates with severity of cardiac involvement in Anderson-Fabry disease. Eur Radiol
Zhao L, Zhang C, Tian J, Saiedi M, Ma C, Li N et al (2021) Quantification of myocardial deformation in patients with Fabry disease by cardiovascular magnetic resonance feature tracking imaging. Cardiovasc Diagn Ther 11(1):91–101
Halfmann MC, Benz S, Schoepf UJ, Hennermann JB, Kreitner KF, Lollert A et al (2022b) Biventricular strain assessment indicates progressive impairment of myocardial contractility in phenotypically negative patients with Fabry’s disease. Eur J Radiol 155:110471
Schwartz T, Diederichsen LP, Lundberg IE, Sjaastad I, Sanner H (2016) Cardiac involvement in adult and juvenile idiopathic inflammatory myopathies. RMD Open 2(2):e000291
Kersten J, Güleroglu AM, Rosenbohm A, Buckert D, Ludolph AC, Hackenbroch C et al (2021) Myocardial involvement and deformation abnormalities in idiopathic inflammatory myopathy assessed by CMR feature tracking. Int J Cardiovasc Imaging 37(2):597–603
Liu W, Zhu Y, Feng C, Zhu X, Xu Y, Shi H (2023) Early cardiac involvement detected by cardiac magnetic resonance feature tracking in idiopathic inflammatory myopathy with preserved ejection fraction. Int J Cardiovasc Imaging 39(1):183–194
Rezaeian N, Mohtasham MA, Khaleel AJ, Parnianfard N, Kasani K, Golshan R (2020) Comparison of global strain values of myocardium in beta-thalassemia major patients with iron load using specific feature tracking in cardiac magnetic resonance imaging. Int J Cardiovasc Imaging 36(7):1343–1349
Rezaeian N, Asadian S, Parsaee M, Toloueitabar Y, Hemmati Komasi MM, Shayan L et al (2021) The predictive role of cardiac magnetic resonance imaging in determining thalassemia patients with intermediately to highly probable pulmonary hypertension. Echocardiography 38(10):1769–1777
Das KM, Baskaki UMA, Pulinchani A, Ali HM, Almanssori TM, Gorkom KV et al (2022) Significance of cardiac magnetic resonance feature tracking of the right ventricle in predicting subclinical dysfunction in patients with thalassemia major. Diagnostics (Basel) 12(8)
Alis D, Asmakutlu O, Topel C, Sahin AA, Karaarslan E (2022) Association between left ventricular strain and cardiac iron load in beta-thalassaemia major: a cardiac magnetic resonance study. Acta Cardiol 77(1):71–80
Ojha V, Ganga KP, Seth T, Roy A, Naik N, Jagia P et al (2021) Role of CMR feature-tracking derived left ventricular strain in predicting myocardial iron overload and assessing myocardial contractile dysfunction in patients with thalassemia major. Eur Radiol 31(8):6184–6192
Asadian S, Rezaeian N, Hosseini L, Toloueitabar Y, Komasi MMH, Shayan L (2021) How does iron deposition modify the myocardium? A feature-tracking cardiac magnetic resonance study. Int J Cardiovasc Imaging 37(11):3269–3277
Asadian S, Hosseini L, Rezaeian N (2022) The importance of functional and feature-tracking cardiac mri parameters in prediction of adverse cardiac events and cardiac mortality in thalassemia patients. Acad Radiol 29(Suppl 4):S91–S99
Ramos-Casals M, Brito-Zerón P, Mariette X (2021) Systemic and organ-specific immune-related manifestations of COVID-19. Nat Rev Rheumatol 17(6):315–332
Puntmann VO, Carerj ML, Wieters I, Fahim M, Arendt C, Hoffmann J et al (2020) Outcomes of cardiovascular magnetic resonance imaging in patients recently recovered from coronavirus disease 2019 (COVID-19). JAMA Cardiol 5(11):1265–1273
Fronza M, Thavendiranathan P, Chan V, Karur GR, Udell JA, Wald RM et al (2022) Myocardial injury pattern at MRI in COVID-19 vaccine-associated myocarditis. Radiology 304(3):553–562
Shiyovich A, Witberg G, Aviv Y, Eisen A, Orvin K, Wiessman M et al (2022) Myocarditis following COVID-19 vaccination: magnetic resonance imaging study. Eur Heart J Cardiovasc Imaging 23(8):1075–1082
Urmeneta Ulloa J, Martínez de Vega V, Salvador Montañés O, Álvarez Vázquez A, Sánchez-Enrique C, Hernández Jiménez S et al (2021) Cardiac magnetic resonance in recovering COVID-19 patients. Feature tracking and mapping analysis to detect persistent myocardial involvement. Int J Cardiol Heart Vasc 36:100854
Gröschel J, Bhoyroo Y, Blaszczyk E, Trauzeddel RF, Viezzer D, Saad H et al (2022) Different impacts on the heart after COVID-19 infection and vaccination: insights from cardiovascular magnetic resonance. Front Cardiovasc Med 9:916922
Wang H, Li R, Zhou Z, Jiang H, Yan Z, Tao X et al (2021) Cardiac involvement in COVID-19 patients: mid-term follow up by cardiovascular magnetic resonance. J Cardiovasc Magn Reson 23(1):14
Author information
Authors and Affiliations
Contributions
Parmida Sadat Pezeshki, Seyyed Mojtaba Ghorashi, Hamidreza Pouraliakbar, and Golnaz Houshmand drafted the manuscript. Mojdeh Ganjparvar, Kiara rezaei-kalantari, Amir Fazeli, and Negar Omidi critically revised and edited the manuscript for important intellectual content. All authors reviewed the manuscript and approved the version to be published.
Corresponding author
Ethics declarations
Ethical approval
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Pezeshki, P.S., Ghorashi, S.M., Houshmand, G. et al. Feature tracking cardiac magnetic resonance imaging to assess cardiac manifestations of systemic diseases. Heart Fail Rev 28, 1189–1199 (2023). https://doi.org/10.1007/s10741-023-10321-6
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10741-023-10321-6