Abstract
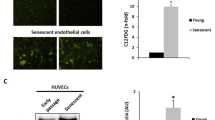
Studies have shown that miR-217 can induce cell senescence, but its mechanism of action in vascular endothelial cell senescence is less reported. Therefore, this study aimed to investigate how miR-217 plays a role in endothelial cell senescence. Human umbilical vein endothelial cells (HUVECs) were used to replicate the aging model, and the population doubling levels (PDLs) during cell passage were counted. Senescence-associated β-galactosidase (SA-β-gal) staining, Real-time quantitative PCR (RT-qPCR), MTT assay, Transwell, and tube formation were used to detect the effects of miR-217 on young and senescent HUVECs. Targetscan7.2 and luciferase assay predicted and verified the relationship between miR-217 and the target gene, and the expression of silent information regulator 1 (SIRT1) and p53 was detected by RT-qPCR and western blot. In addition, SA-β-gal staining detected the effects of miR-217 inhibitor and SIRT1 on senescent HUVECs. MiR-217 was upregulated in senescent endothelial cells. Overexpression of miR-217 promoted the increase of SA-β-gal positive cells, and inhibited proliferation, migration and angiogenesis during endothelial cell growth. Furthermore, SIRT1 was a target gene of miR-217. Simultaneous silencing of SIRT1 reversed the effect of miR-217 inhibitor on the reduction of SA-β-gal positive-staining cells. Our data suggest that overexpression of miR-217 promoted vascular endothelial cell senescence by targeting the SIRT1/p53 signaling pathway, which may provide a new basis for studying the mechanism of action in vascular endothelial cell senescence.





Similar content being viewed by others
Availability of data and materials
The analysed data sets generated during the study are available from the corresponding author on reasonable request.
References
Baar MP et al (2017) Targeted apoptosis of senescent cells restores tissue homeostasis in response to chemotoxicity and aging. Cell 169:132–147, e116. https://doi.org/10.1016/j.cell.2017.02.031
Behrouzfar K, Alaee M, Nourbakhsh M, Gholinejad Z, Golestani A (2017) Extracellular NAMPT/visfatin causes p53 deacetylation via NAD production and SIRT1 activation in breast cancer cells. Cell Biochem Funct 35:327–333. https://doi.org/10.1002/cbf.3279
Dimmeler S, Nicotera P (2013) MicroRNAs in age-related diseases. EMBO Mol Med 5:180–190. https://doi.org/10.1002/emmm.201201986
Doebele C et al (2010) Members of the microRNA-17-92 cluster exhibit a cell-intrinsic antiangiogenic function in endothelial cells. Blood 115:4944–4950. https://doi.org/10.1182/blood-2010-01-264812
Fan C et al (2019) Ginsenoside Rb1 attenuates high glucose-induced oxidative injury via the NAD-PARP-SIRT axis in rat retinal capillary endothelial cells. Int J Mol Sci 20:4936. https://doi.org/10.3390/ijms20194936
Han D et al (2017) Reduced silent information regulator 1 signaling exacerbates sepsis-induced myocardial injury and mitigates the protective effect of a liver X receptor agonist. Free Radic Biol Med 113:291–303. https://doi.org/10.1016/j.freeradbiomed.2017.10.005
Heo JI, Kim KI, Woo SK, Kim JS, Choi KJ, Lee HJ, Kim KS (2019) Stromal cell-derived factor 1 protects brain vascular endothelial cells from radiation-induced brain damage. Cells 8:1230. https://doi.org/10.3390/cells8101230
Jeyapalan Z, Deng Z, Shatseva T, Fang L, He C, Yang BB (2011) Expression of CD44 3′-untranslated region regulates endogenous microRNA functions in tumorigenesis and angiogenesis. Nucleic Acids Res 39:3026–3041. https://doi.org/10.1093/nar/gkq1003
Kamali K, Korjan ES, Eftekhar E, Malekzadeh K, Soufi FG (2016) The role of miR-146a on NF-kappaB expression level in human umbilical vein endothelial cells under hyperglycemic condition. Bratislavske lekarske listy 117:376–380
Karamysheva AF (2008) Mechanisms of angiogenesis. Biochemistry (Biokhimiia) 73:751–762
Kato M et al (2009) TGF-beta activates Akt kinase through a microRNA-dependent amplifying circuit targeting PTEN. Nat Cell Biol 11:881–889. https://doi.org/10.1038/ncb1897
Kumar S, Vijayan M, Bhatti JS, Reddy PH (2017) MicroRNAs as peripheral biomarkers in aging and age-related diseases. Prog Mol Biol Transl Sci 146:47–94. https://doi.org/10.1016/bs.pmbts.2016.12.013
Lee BY et al (2006) Senescence-associated beta-galactosidase is lysosomal beta-galactosidase. Aging Cell 5:187–195. https://doi.org/10.1111/j.1474-9726.2006.00199.x
Leong DP, Joseph PG, McKee M, Anand SS, Teo KK, Schwalm JD, Yusuf S (2017) Reducing the global burden of cardiovascular disease, part 2: prevention and treatment of cardiovascular disease. Circ Res 121:695–710. https://doi.org/10.1161/circresaha.117.311849
Li W et al (2015) Silent information regulator 1 (SIRT1) promotes the migration and proliferation of endothelial progenitor cells through the PI3K/Akt/eNOS signaling pathway. Int J Clin Exp Pathol 8:2274–2287
Lin Y, Cheng K, Wang T, Xie Q, Chen M, Chen Q, Wen Q (2017) miR-217 inhibits proliferation, migration, and invasion via targeting AKT3 in thyroid cancer. Biomed Pharmacother (Biomedecine and Pharmacotherapie) 95:1718–1724. https://doi.org/10.1016/j.biopha.2017.09.074
Magenta A et al (2011) miR-200c is upregulated by oxidative stress and induces endothelial cell apoptosis and senescence via ZEB1 inhibition. Cell Death Differ 18:1628–1639. https://doi.org/10.1038/cdd.2011.42
Menghini R et al (2009) MicroRNA 217 modulates endothelial cell senescence via silent information regulator 1. Circulation 120:1524–1532. https://doi.org/10.1161/circulationaha.109.864629
Mensà E et al (2020) Small extracellular vesicles deliver miR-21 and miR-217 as pro-senescence effectors to endothelial cells. J Extracell Vesicles 9:1725285. https://doi.org/10.1080/20013078.2020.1725285
Neault M, Couteau F, Bonneau E, De Guire V, Mallette FA (2017) Molecular regulation of cellular senescence by MicroRNAs: implications in cancer and age-related diseases. Int Rev Cell Mol Biol 334:27–98. https://doi.org/10.1016/bs.ircmb.2017.04.001
Piechota M, Sunderland P, Wysocka A, Nalberczak M, Sliwinska MA, Radwanska K, Sikora E (2016) Is senescence-associated beta-galactosidase a marker of neuronal senescence? Oncotarget 7:81099–81109. https://doi.org/10.18632/oncotarget.12752
Potente M, Gerhardt H, Carmeliet P (2011) Basic and therapeutic aspects of angiogenesis. Cell 146:873–887. https://doi.org/10.1016/j.cell.2011.08.039
Prattichizzo F et al (2016) Endothelial cell senescence and inflammaging: microRNAs as biomarkers and innovative therapeutic tools. Curr Drug Targets 17:388–397
Rau CS et al (2014) Lipopolysaccharide-induced microRNA-146a targets CARD10 and regulates angiogenesis in human umbilical vein endothelial cells. Toxicol Sci 140:315–326. https://doi.org/10.1093/toxsci/kfu097
Regina C et al (2016) Vascular ageing and endothelial cell senescence: molecular mechanisms of physiology and diseases. Mech Ageing Dev 159:14–21. https://doi.org/10.1016/j.mad.2016.05.003
Rio DC, Ares M Jr, Hannon GJ, Nilsen TW (2010) Purification of RNA using TRIzol (TRI reagent). Cold Spring Harbor Protoc 2010:pdb.prot5439. https://doi.org/10.1101/pdb.prot5439
Sassi Y et al (2017) Cardiac myocyte miR-29 promotes pathological remodeling of the heart by activating Wnt signaling. Nat Commun. 8:1614. https://doi.org/10.1038/s41467-017-01737-4
Schulte C, Karakas M, Zeller T (2017) microRNAs in cardiovascular disease—clinical application. Clin Chem Lab Med 55:687–704. https://doi.org/10.1515/cclm-2016-0576
Sturtzel C (2017) Endothelial cells. Adv Exp Med Biol 1003:71–91. https://doi.org/10.1007/978-3-319-57613-8_4
Tabuchi T, Satoh M, Itoh T, Nakamura M (2012) MicroRNA-34a regulates the longevity-associated protein SIRT1 in coronary artery disease: effect of statins on SIRT1 and microRNA-34a expression. Clin Sci (London, England : 1979) 123:161–171. https://doi.org/10.1042/cs20110563
Thiara B (2015) Cardiovascular disease. Nurs Standard (Royal College of Nursing (Great Britain): 1987) 29:60. https://doi.org/10.7748/ns.29.33.60.s44
Tian XL, Li Y (2014) Endothelial cell senescence and age-related vascular diseases. J Genet Genomics (Yi chuan xue bao) 41:485–495. https://doi.org/10.1016/j.jgg.2014.08.001
Tome M, Sepulveda JC, Delgado M, Andrades JA, Campisi J, Gonzalez MA, Bernad A (2014) miR-335 correlates with senescence/aging in human mesenchymal stem cells and inhibits their therapeutic actions through inhibition of AP-1 activity. Stem Cells (Dayton, Ohio) 32:2229–2244. https://doi.org/10.1002/stem.1699
Tufekci KU, Oner MG, Meuwissen RL, Genc S (2014) The role of microRNAs in human diseases. Methods Mol Biol (Clifton, NJ) 1107:33–50. https://doi.org/10.1007/978-1-62703-748-8_3
Wang Y, Boerma M, Zhou D (2016) Ionizing radiation-induced endothelial cell senescence and cardiovascular diseases. Radiat Res 186:153–161. https://doi.org/10.1667/rr14445.1
Xu X, Jiao X, Song N, Luo W, Liang M, Ding X, Teng J (2017) Role of miR21 on vascular endothelial cells in the protective effect of renal delayed ischemic preconditioning. Mol Med Rep 16:2627–2635. https://doi.org/10.3892/mmr.2017.6870
Yang J, Zhang HF, Qin CF (2017) MicroRNA-217 functions as a prognosis predictor and inhibits pancreatic cancer cell proliferation and invasion via targeting E2F3. Eur Rev Med Pharmacol Sci 21:4050–4057
Yang S, Mi X, Chen Y, Feng C, Hou Z, Hui R, Zhang W (2018) MicroRNA-216a induces endothelial senescence and inflammation via Smad3/IkappaBalpha pathway. J Cell Mol Med 22:2739–2749. https://doi.org/10.1111/jcmm.13567
Yuan Y, Cruzat VF, Newsholme P, Cheng J, Chen Y, Lu Y (2016) Regulation of SIRT1 in aging: Roles in mitochondrial function and biogenesis. Mech Ageing Dev 155:10–21. https://doi.org/10.1016/j.mad.2016.02.003
Zeng L et al (2009) Silent information regulator, Sirtuin 1, and age-related diseases. Geriatr Gerontol Int 9:7–15. https://doi.org/10.1111/j.1447-0594.2008.00504.x
Zhang M et al (2017) miR-217 suppresses proliferation, migration, and invasion promoting apoptosis via targeting MTDH in hepatocellular carcinoma. Oncol Rep 37:1772–1778. https://doi.org/10.3892/or.2017.5401
Zhou M, Hu M, He S, Li B, Liu C, Min J, Hong L (2018) Effects of RSC96 Schwann cell-derived exosomes on proliferation, senescence, and apoptosis of dorsal root ganglion cells in vitro. Med Sci Monit 24:7841–7849. https://doi.org/10.12659/msm.909509
Zhu HY et al (2016) Up-regulation of FGFBP1 signaling contributes to miR-146a-induced angiogenesis in human umbilical vein endothelial cells. Sci Rep 6:25272. https://doi.org/10.1038/srep25272
Zhu L, Yang S, Wang J (2019) miR-217 inhibits the migration and invasion of HeLa cells through modulating MAPK1. Int J Mol Med 44:1824–1832. https://doi.org/10.3892/ijmm.2019.4328
Funding
This work was supported by the Self-financing project of Hebei Science and Technology Plan [182777148].
Author information
Authors and Affiliations
Contributions
Substantial contributions to conception and design: ZW. Data acquisition, data analysis and interpretation: DS, NZ, TY, HT. Drafting the article or critically revising it for important intellectual content: ZW. Final approval of the version to be published: All authors. Agreement to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of the work are appropriately investigated and resolved: All authors.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflicts of interests.
Research involved in human and animal rights
No human and animals are involved in this research.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Wang, Z., Shi, D., Zhang, N. et al. MiR-217 promotes endothelial cell senescence through the SIRT1/p53 signaling pathway. J Mol Histol 52, 257–267 (2021). https://doi.org/10.1007/s10735-020-09945-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10735-020-09945-x