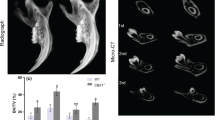
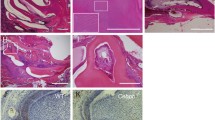
Abstract
Dentin is a major component of teeth that protects dental pulp and maintains tooth health. Bone morphogenetic protein (BMP) signaling is required for the formation of dentin. Mice lacking a BMP type I receptor, activin A receptor type 1 (ACVR1), in the neural crest display a deformed mandible. Acvr1 is known to be expressed in the dental mesenchyme. However, little is known about how BMP signaling mediated by ACVR1 regulates dentinogenesis. To explore the role of ACVR1 in dentin formation in molars and incisors in mice, Acvr1 was conditionally disrupted in Osterix-expressing cells (designated as cKO). We found that loss of Acvr1 in the dental mesenchyme led to dentin dysplasia in molars and osteodentin formation in incisors. Specifically, the cKO mice exhibited remarkable tooth phenotypes characterized by thinner dentin and thicker predentin, as well as compromised differentiation of odontoblasts in molars. We also found osteodentin formation in the coronal part of the cKO mandibular incisors, which was associated with a reduction in the expression of odontogenic gene Dsp and an increase in the expression of osteogenic gene Bsp, leading to an alteration of cell fate from odontoblasts to osteoblasts. In addition, the expressions of WNT antagonists, Dkk1 and Sost, were downregulated and B-catenin was up-regulated in the cKO incisors, while the expression levels were not changed in the cKO molars, compared with the corresponding controls. Our results indicate the distinct and critical roles of ACVR1 between incisors and molars, which is associated with alterations in the WNT signaling related molecules. This study demonstrates for the first time the physiological roles of ACVR1 during dentinogenesis.








Similar content being viewed by others
References
Ahn YH, Kim TH, Choi H, Bae CH, Yang YM, Baek JA, Lee JC Cho ES (2015) Disruption of Tgfbr2 in odontoblasts leads to aberrant pulp calcification. J Dent Res 94(6):828–835. https://doi.org/10.1177/0022034515577427
Amri N, Djole SX, Petit S, Babajko S, Coudert AE, Castaneda B, Simon S, Berdal A (2016) Distorted patterns of dentinogenesis and eruption in Msx2 null mutants: involvement of sost/sclerostin. Am J Pathol 186(10):2577–2587. https://doi.org/10.1016/j.ajpath.2016.06.013
Boushell LW, Nagaoka H, Nagaoka H, Yamauchi M (2011) Increased matrix metalloproteinase-2 and bone sialoprotein response to human coronal caries. Caries Res 45(5):453–459. https://doi.org/10.1159/000330601
Chen J, Shapiro HS, Sodek J (1992) Development expression of bone sialoprotein mRNA in rat mineralized connective tissues. J Bone Miner Res 7(8):987–997. https://doi.org/10.1002/jbmr.5650070816
Chen S, Gluhak-Heinrich J, Wang YH, Wu YM, Chuang HH, Chen L, Yuan GH, Dong J, Gay I, MacDougall M (2009) Runx2, osx, and dspp in tooth development. J Dent Res 88(10):904–909. https://doi.org/10.1177/0022034509342873
Chen G, Deng C, Li YP (2012) TGF-beta and BMP signaling in osteoblast differentiation and bone formation. Int J Biol Sci 8(2):272–288. https://doi.org/10.7150/ijbs.2929
de Souza LE, Malta TM, Haddad KS, Covas DT (2016) Mesenchymal stem cells and pericytes: to what extent are they related? Stem Cells Dev 25(24):1843–1852. https://doi.org/10.1089/scd.2016.0109
Dong X, Shen B, Ruan N, Guan Z, Zhang Y, Chen Y, Hu X (2014) Expression patterns of genes critical for BMP signaling pathway in developing human primary tooth germs. Histochem Cell Biol 142(6):657–665. https://doi.org/10.1007/s00418-014-1241-y
Dudas M, Sridurongrit S, Nagy A, Okazaki K, Kaartinen V (2004) Craniofacial defects in mice lacking BMP type I receptor Alk2 in neural crest cells. Mech Dev 121(2):173–182. https://doi.org/10.1016/j.mod.2003.12.003
Fan L, Deng S, Sui X, Liu M, Cheng S, Wang Y, Gao Y, Chu CH, Zhang Q (2018) Constitutive activation of beta-catenin in ameloblasts leads to incisor enamel hypomineralization. J Mol Histol 49(5):499–507. https://doi.org/10.1007/s10735-018-9788-x
Feng J, Jing J, Li J, Zhao H, Punj V, Zhang T, Xu J, Chai Y (2017) BMP signaling orchestrates a transcriptional network to control the fate of mesenchymal stem cells in mice. Development 144(14):2560–2569. https://doi.org/10.1242/dev.150136
Foster BL, Ao M, Willoughby C, Soenjaya Y, Holm E, Lukashova L, Tran AB, Wimer HF, Zerfas PM, Nociti Jr FH, Kantovitz KR, Somerman MJ (2015) Mineralization defects in cementum and craniofacial bone from loss of bone sialoprotein. Bone 78:150–164. https://doi.org/10.1016/j.bone.2015.05.007
Graf D, Malik Z, Hayano S, Mishina Y (2016) Common mechanisms in development and disease: BMP signaling in craniofacial development. Cytokine Growth Factor Rev 27:129–139. https://doi.org/10.1016/j.cytogfr.2015.11.004
Grafe I, Alexander S, Peterson JR, Snider TN, Levi B, Lee B, Mishina Y (2017) TGF-beta family signaling in mesenchymal differentiation. Cold Spring Harb Perspect Biol. https://doi.org/10.1101/cshperspect.a022202
Hunter DJ, Bardet C, Mouraret S, Liu B, Singh G, Sadoine J, Helms JA (2015) Wnt acts as a prosurvival signal to enhance dentin regeneration. J Bone Miner Res 30(7):1150–1159. https://doi.org/10.1002/jbmr.2444
Ishimoto K, Hayano S, Yanagita T, Kurosaka H, Kawanabe N, Itoh S, Ono M, Kuboki T, Kamioka H, Yamashiro T (2015) Topical application of lithium chloride on the pulp induces dentin regeneration. PLoS ONE 10(3):e0121938. https://doi.org/10.1371/journal.pone.0121938
Jani PH, Gibson MP, Liu C, Zhang H, Wang X, Lu Y, Qin C (2016) Transgenic expression of Dspp partially rescued the long bone defects of Dmp1-null mice. Matrix Biol 52–54:95–112. https://doi.org/10.1016/j.matbio.2015.12.001
Kaartinen V, Nagy A (2001) Removal of the floxed neo gene from a conditional knockout allele by the adenoviral Cre recombinase in vivo. Genesis 31(3):126–129
Kamiya N, Ye L, Kobayashi T, Lucas DJ, Mochida Y, Yamauchi M, Kronenberg HM, Feng JQ, Mishina Y (2008) Disruption of BMP signaling in osteoblasts through type IA receptor (BMPRIA) increases bone mass. J Bone Miner Res 23(12):2007–2017. https://doi.org/10.1359/jbmr.080809
Kamiya N, Kobayashi T, Mochida Y, Yu PB, Yamauchi M, Kronenberg HM, Mishina Y (2010) Wnt inhibitors Dkk1 and Sost are downstream targets of BMP signaling through the type IA receptor (BMPRIA) in osteoblasts. J Bone Miner Res 25(2):200–210. https://doi.org/10.1359/jbmr.090806
Kamiya N, Kaartinen VM, Mishina Y (2011) Loss-of-function of ACVR1 in osteoblasts increases bone mass and activates canonical Wnt signaling through suppression of Wnt inhibitors SOST and DKK1. Biochem Biophys Res Commun 414(2):326–330. https://doi.org/10.1016/j.bbrc.2011.09.060
Katsura KA, Horst JA, Chandra D, Le TQ, Nakano Y, Zhang Y, Horst OV, Zhu L, Le MH, DenBesten PK (2014) WDR72 models of structure and function: a stage-specific regulator of enamel mineralization. Matrix Biol 38:48–58. https://doi.org/10.1016/j.matbio.2014.06.005
Kawashima N, Okiji T (2016) Odontoblasts: specialized hard-tissue-forming cells in the dentin–pulp complex. Congenit Anom (Kyoto) 56(4):144–153. https://doi.org/10.1111/cga.12169
Kim TH, Bae CH, Lee JY, Lee JC, Ko SO, Chai Y, Cho ES (2015) Temporo-spatial requirement of Smad4 in dentin formation. Biochem Biophys Res Commun 459(4):706–712. https://doi.org/10.1016/j.bbrc.2015.03.014
Kuang-Hsien Hu J, Mushegyan V, Klein OD (2014) On the cutting edge of organ renewal: identification, regulation, and evolution of incisor stem cells. Genesis 52(2):79–92. https://doi.org/10.1002/dvg.22732
Laugel-Haushalter V, Paschaki M, Thibault-Carpentier C, Dembele D, Dolle P, Bloch-Zupan A (2013) Molars and incisors: show your microarray IDs. BMC Res Notes 6:113. https://doi.org/10.1186/1756-0500-6-113
Lee TY, Lee DS, Kim HM, Ko JS, Gronostajski RM, Cho MI, Son HH, Park JC (2009a) Disruption of Nfic causes dissociation of odontoblasts by interfering with the formation of intercellular junctions and aberrant odontoblast differentiation. J Histochem Cytochem 57(5):469–476. https://doi.org/10.1369/jhc.2009.952622
Lee DS, Park JT, Kim HM, Ko JS, Son HH, Gronostajski RM, Cho MI, Choung PH, Park JC (2009b) Nuclear factor I-C is essential for odontogenic cell proliferation and odontoblast differentiation during tooth root development. J Biol Chem 284(25):17293–17303. https://doi.org/10.1074/jbc.M109.009084
Lerner UH, Ohlsson C (2015) The WNT system: background and its role in bone. J Intern Med 277(6):630–649. https://doi.org/10.1111/joim.12368
Li J, Huang X, Xu X, Mayo J, Bringas P Jr, Jiang R, Wang S, Chai Y (2011a) SMAD4-mediated WNT signaling controls the fate of cranial neural crest cells during tooth morphogenesis. Development 138(10):1977–1989. https://doi.org/10.1242/dev.061341
Li L, Lin M, Wang Y, Cserjesi P, Chen Z, Chen Y (2011b) BmprIa is required in mesenchymal tissue and has limited redundant function with BmprIb in tooth and palate development. Dev Biol 349(2):451–461. https://doi.org/10.1016/j.ydbio.2010.10.023
Li L, Wang Y, Lin M, Yuan G, Yang G, Zheng Y, Chen Y (2013) Augmented BMPRIA-mediated BMP signaling in cranial neural crest lineage leads to cleft palate formation and delayed tooth differentiation. PLoS ONE 8(6):e66107. https://doi.org/10.1371/journal.pone.0066107
Li J, Feng J, Liu Y, Ho TV, Grimes W, Ho HA, Park S, Wang S, Chai Y (2015) BMP-SHH signaling network controls epithelial stem cell fate via regulation of its niche in the developing tooth. Dev Cell 33(2):125–135. https://doi.org/10.1016/j.devcel.2015.02.021
Li J, Parada C, Chai Y (2017) Cellular and molecular mechanisms of tooth root development. Development 144(3):374–384. https://doi.org/10.1242/dev.137216
Lin S, Svoboda KK, Feng JQ, Jiang X (2016) The biological function of type I receptors of bone morphogenetic protein in bone. Bone Res 4:16005. https://doi.org/10.1038/boneres.2016.5
Liu W, Sun X, Braut A, Mishina Y, Behringer RR, Mina M, Martin JF (2005) Distinct functions for Bmp signaling in lip and palate fusion in mice. Development 132(6):1453–1461. https://doi.org/10.1242/dev.01676
Liu L, Chen W, Li L, Xu F, Jiang B (2017) Inhibition of chondroitin sulfate glycosaminoglycans incorporation affected odontoblast differentiation in cultured embryonic mouse molars. J Mol Histol 48(5–6):337–345. https://doi.org/10.1007/s10735-017-9732-5
Lungova V, Radlanski RJ, Tucker AS, Renz H, Misek I, Matalova E (2011) Tooth-bone morphogenesis during postnatal stages of mouse first molar development. J Anat 218(6):699–716. https://doi.org/10.1111/j.1469-7580.2011.01367.x
Macias-Silva M, Hoodless PA, Tang SJ, Buchwald M, Wrana JL (1998) Specific activation of Smad1 signaling pathways by the BMP7 type I receptor, ALK2. J Biol Chem 273(40):25628–25636
Majidinia M, Yousefi B (2017) The roles of signaling pathways in bone repair and regeneration. J Cell Physiol. https://doi.org/10.1002/jcp.26042
Malik Z, Alexiou M, Hallgrimsson B, Economides AN, Luder HU, Graf D (2018) Bone morphogenetic protein 2 coordinates early tooth mineralization. J Dent Res 97(7):835–843. https://doi.org/10.1177/0022034518758044
Meschi N, Hilkens P, Lambrichts I, Van den Eynde K, Mavridou A, Strijbos O, De Ketelaere M, Van Gorp G, Lambrechts P (2016) Regenerative endodontic procedure of an infected immature permanent human tooth: an immunohistological study. Clin Oral Investig 20(4):807–814. https://doi.org/10.1007/s00784-015-1555-8
Mishina Y, Crombie R, Bradley A, Behringer RR (1999) Multiple roles for activin-like kinase-2 signaling during mouse embryogenesis. Dev Biol 213(2):314–326. https://doi.org/10.1006/dbio.1999.9378
Nose-Ishibashi K, Watahiki J, Yamada K, Maekawa M, Watanabe A, Yamamoto G, Enomoto A, Matsuba Y, Nampo T, Taguchi T, Ichikawa Y, Maki K (2014) Soft-diet feeding after weaning affects behavior in mice: potential increase in vulnerability to mental disorders. Neuroscience 263:257–268. https://doi.org/10.1016/j.neuroscience.2013.12.065
Nudelman F, Lausch AJ, Sommerdijk NA, Sone ED (2013) In vitro models of collagen biomineralization. J Struct Biol 183(2):258–269. https://doi.org/10.1016/j.jsb.2013.04.003
Orvis GD, Jamin SP, Kwan KM, Mishina Y, Kaartinen VM, Huang S, Roberts AB, Umans L, Huylebroeck D, Zwijsen A, Wang D, Behringer RR (2008) Functional redundancy of TGF-beta family type I receptors and receptor-Smads in mediating anti-Mullerian hormone-induced Mullerian duct regression in the mouse. Biol Reprod 78(6):994–1001. https://doi.org/10.1095/biolreprod.107.066605
Qin C, Brunn JC, Cadena E, Ridall A, Tsujigiwa H, Nagatsuka H, Nagai N, Butler WT (2002) The expression of dentin sialophosphoprotein gene in bone. J Dent Res 81(6):392–394. https://doi.org/10.1177/154405910208100607
Rahman MS, Akhtar N, Jamil HM, Banik RS, Asaduzzaman SM (2015) TGF-beta/BMP signaling and other molecular events: regulation of osteoblastogenesis and bone formation. Bone Res 3:15005. https://doi.org/10.1038/boneres.2015.5
Rakian A, Yang WC, Gluhak-Heinrich J, Cui Y, Harris MA, Villarreal D, Feng JQ, MacDougall M, Harris SE (2013) Bone morphogenetic protein-2 gene controls tooth root development in coordination with formation of the periodontium. Int J Oral Sci 5(2):75–84. https://doi.org/10.1038/ijos.2013.41
Rodda SJ, McMahon AP (2006) Distinct roles for Hedgehog and canonical Wnt signaling in specification, differentiation and maintenance of osteoblast progenitors. Development 133(16):3231–3244. https://doi.org/10.1242/dev.02480
Saito K, Fukumoto E, Yamada A, Yuasa K, Yoshizaki K, Iwamoto T, Saito M, Nakamura T, Fukumoto S (2015) Interaction between fibronectin and beta1 integrin is essential for tooth development. PLoS ONE 10(4):e0121667. https://doi.org/10.1371/journal.pone.0121667
Schiaffino S, Reggiani C (1996) Molecular diversity of myofibrillar proteins: gene regulation and functional significance. Physiol Rev 76(2):371–423. https://doi.org/10.1152/physrev.1996.76.2.371
Schwarz F, Mihatovic I, Golubovic V, Becker J (2013) Dentointegration of a titanium implant: a case report. Oral Maxillofac Surg 17(3):235–241. https://doi.org/10.1007/s10006-012-0378-x
Sfakianou A, Emmanouil DE, Tosios KI, Sklavounou A (2016) Peripheral tumor with osteodentin and cementum-like material in an infant: odontogenic hamartoma or odontoma? J Dent Child (Chic) 83(1):38–41
Shi S, de Gorter DJ, Hoogaars WM, Ac’t Hoen P, ten Dijke P (2013) Overactive bone morphogenetic protein signaling in heterotopic ossification and Duchenne muscular dystrophy. Cell Mol Life Sci 70(3):407–423. https://doi.org/10.1007/s00018-012-1054-x
Staines KA, MacRae VE, Farquharson C (2012) The importance of the SIBLING family of proteins on skeletal mineralisation and bone remodelling. J Endocrinol 214(3):241–255. https://doi.org/10.1530/JOE-12-0143
Sun Y, Lu Y, Chen L, Gao T, D’Souza R, Feng JQ, Qin C (2011) DMP1 processing is essential to dentin and jaw formation. J Dent Res 90(5):619–624. https://doi.org/10.1177/0022034510397839
Suzuki H, Amizuka N, Kii I, Kawano Y, Nozawa-Inoue K, Suzuki A, Yoshie H, Kudo A, Maeda T (2004) Immunohistochemical localization of periostin in tooth and its surrounding tissues in mouse mandibles during development. Anat Rec A Discov Mol Cell Evol Biol 281(2):1264–1275. https://doi.org/10.1002/ar.a.20080
Suzuki S, Haruyama N, Nishimura F, Kulkarni AB (2012) Dentin sialophosphoprotein and dentin matrix protein-1: two highly phosphorylated proteins in mineralized tissues. Arch Oral Biol 57(9):1165–1175. https://doi.org/10.1016/j.archoralbio.2012.03.005
Taniguchi K, Roberts LR, Aderca IN, Dong X, Qian C, Murphy LM, Nagorney DM, Burgart LJ, Roche PC, Smith DI, Ross JA, Liu W (2002) Mutational spectrum of beta-catenin, AXIN1, and AXIN2 in hepatocellular carcinomas and hepatoblastomas. Oncogene 21(31):4863–4871. https://doi.org/10.1038/sj.onc.1205591
Toyono T, Nakashima M, Kuhara S, Akamine A (1997) Expression of TGF-beta superfamily receptors in dental pulp. J Dent Res 76(9):1555–1560. https://doi.org/10.1177/00220345970760090701
Vogel P, Read RW, Hansen GM, Powell DR, Kantaputra PN, Zambrowicz B, Brommage R (2016) Dentin dysplasia in notum knockout mice. Vet Pathol 53(4):853–862. https://doi.org/10.1177/0300985815626778
Wang X, Wang J, Liu Y, Yuan B, Ruest LB, Feng JQ, Qin C (2015) The specific role of FAM20C in dentinogenesis. J Dent Res 94(2):330–336. https://doi.org/10.1177/0022034514563334
Xavier GM, Patist AL, Healy C, Pagrut A, Carreno G, Sharpe PT, Martinez-Barbera JP, Thavaraj S, Cobourne MT, Andoniadou CL (2015) Activated WNT signaling in postnatal SOX2-positive dental stem cells can drive odontoma formation. Sci Rep 5:14479. https://doi.org/10.1038/srep14479
Xu H, Snider TN, Wimer HF, Yamada SS, Yang T, Holmbeck K, Foster BL (2016) Multiple essential MT1-MMP functions in tooth root formation, dentinogenesis, and tooth eruption. Matrix Biol 52–54:266–283. https://doi.org/10.1016/j.matbio.2016.01.002
Yamakoshi Y, Richardson AS, Nunez SM, Yamakoshi F, Milkovich RN, Hu JC, Bartlett JD, Simmer JP (2011) Enamel proteins and proteases in Mmp20 and Klk4 null and double-null mice. Eur J Oral Sci 119(Suppl 1):206–216. https://doi.org/10.1111/j.1600-0722.2011.00866.x
Yang Z, Hai B, Qin L, Ti X, Shangguan L, Zhao Y, Wiggins L, Liu F (2013) Cessation of epithelial Bmp signaling switches the differentiation of crown epithelia to the root lineage in a beta-catenin-dependent manner. Mol Cell Biol 33(23):4732–4744. https://doi.org/10.1128/MCB.00456-13
Yi SE, Daluiski A, Pederson R, Rosen V, Lyons KM (2000) The type I BMP receptor BMPRIB is required for chondrogenesis in the mouse limb. Development 127(3):621–630
Yun CY, Choi H, You YJ, Yang JY, Baek JA, Cho ES (2016) Requirement of Smad4-mediated signaling in odontoblast differentiation and dentin matrix formation. Anat Cell Biol 49(3):199–205. https://doi.org/10.5115/acb.2016.49.3.199
Zhang H, Jani P, Liang T, Lu Y, Qin C (2017) Inactivation of bone morphogenetic protein 1 (Bmp1) and tolloid-like 1 (Tll1) in cells expressing type I collagen leads to dental and periodontal defects in mice. J Mol Histol 48(2):83–98. https://doi.org/10.1007/s10735-016-9708-x
Acknowledgements
The authors thank Dr. Vesa Kaartinen for the Acvr1-floxed mice. This work was supported by grants from the National Key Research and Development Program of China (2016YFC1102800), the Natural Science Foundation of China (81320108011, 81500820, 81600890, 81600843, 81600879 and 81600823), China Postdoctoral Science Foundation (2017M621219 and 2018T110258), the Fundamental Research Funds for the Central Universities, and JLU Science and Technology Innovative Research Team 2017TD-11. YM is supported by a grant from the National Institutes of Health (R01DE020843).
Author information
Authors and Affiliations
Contributions
Xue Zhang and Ce Shi contributed to the experimental design, experiment performance, and paper writing; Xue Zhang, Ce Shi, Qilin Liu, Huan Zhao and Xinqing Hao prepared the mouse samples and performed the genotyping; Yue Hu and Cangwei Liu did the HE/IHC stainings; Guangxing Yan analyzed the IHC data; Daowei Li performed the micro-CT scanning and analysis; Xue Zhang, Ce Shi, Qilin Liu, Yuji Mishina and Hongchen Sun prepared and revised the manuscript. All authors have read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflicts of interest
There are no conflicts of interest.
Ethics approval
Animal use in this study was approved by the Institutional Animal Care and Use Committee of Jilin University, Changchun, People’s Republic of China. The animal experiments were performed in accordance with the requirements of the Experimental Animal Ethics and Welfare guidelines (Permit Number: 20160205).
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zhang, X., Shi, C., Zhao, H. et al. Distinctive role of ACVR1 in dentin formation: requirement for dentin thickness in molars and prevention of osteodentin formation in incisors of mice. J Mol Hist 50, 43–61 (2019). https://doi.org/10.1007/s10735-018-9806-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10735-018-9806-z