Abstract
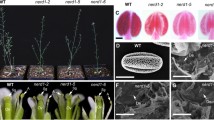
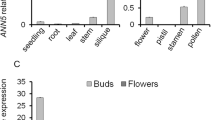
Anther dehiscence is a crucial step for pollen grains release and male fertility. Filaments, which transport water, nutrients and hormones to the anthers, are important for anther dehiscence. In this study, we characterized the Arabidopsis MICROTUBULE ORGANIZATION 1 (MOR1) gene that involves in the filament functions and plays important roles in anther dehiscence. The Arabidopsis microtubule organization 1–1 (mor1-1) mutant exhibited an anther indehiscence phenotype at 24 °C. Such the defect did not occur at a lower temperature (19 °C). Further analysis indicated that both the cortical microtubule (CMT) organization and plasma membrane homeostasis were drastically impaired and disturbed in mor1-1 filament cells under the growth conditions of 24 °C. Transmission electron microscopy and FM4-64 up-take assays showed that endocytosis process in the mor1-1 filament cells were disrupted at 24 °C. Furthermore, the cortical-associated RFP tagged clathrin light chain foci were reduced in the mor1-1 filament cells. These results suggested that the MOR1 mediated CMT organization is important for clathrin-mediated endocytosis in the filament cells, and critical for anther dehiscence in thermosensitivity.







Similar content being viewed by others
References
Acosta IF, Przybyl M (2019) Jasmonate signaling during Arabidopsis stamen maturation. Plant Cell Physiol 60(12):2648–2659. https://doi.org/10.1093/pcp/pcz201
Al-Bassam J, Larsen NA, Hyman AA, Harrison SC (2007) Crystal structure of a TOG domain: conserved features of XMAP215/Dis1-family TOG domains and implications for tubulin binding. Structure 15(3):355–362. https://doi.org/10.1016/j.str.2007.01.012
Bechtold N, Pelletier G (1998) In planta Agrobacterium mediated transformation of adult Arabidopsis thaliana plants by vacuum infiltration. In: Martinez-Zapater JM, Salinas J (eds) Arabidopsis protocols. Humana Press, Totowa, pp 259–266. https://doi.org/10.1385/0-89603-391-0:259
Bolte S, Talbot C, Boutte Y, Catrice O, Read ND, Satiat-Jeunemaitre B (2004) FM-dyes as experimental probes for dissecting vesicle trafficking in living plant cells. J Microsc 214(2):159–173. https://doi.org/10.1111/j.0022-2720.2004.01348.x
Collings DA, Gebbie LK, Howles PA, Hurley UA, Birch RJ, Cork AH, Hocart CH, Arioli T, Williamson RE (2008) Arabidopsis dynamin-like protein DRP1A: a null mutant with widespread defects in endocytosis, cellulose synthesis, cytokinesis, and cell expansion. J Exp Bot 59(2):361–376. https://doi.org/10.1093/jxb/erm324
Eleftheriou EP, Baskin TI, Hepler PK (2005) Aberrant cell plate formation in the Arabidopsis thaliana microtubule organization 1 mutant. Plant Cell Physiol 46(4):671–675. https://doi.org/10.1093/pcp/pci068
Fan L, Hao H, Xue Y, Zhang L, Song K, Ding Z, Botella MA, Wang H, Lin J (2013) Dynamic analysis of Arabidopsis AP2 sigma subunit reveals a key role in clathrin-mediated endocytosis and plant development. Development 140(18):3826–3837. https://doi.org/10.1242/dev.095711
Fan L, Li R, Pan J, Ding Z, Lin J (2015) Endocytosis and its regulation in plants. Trends Plant Sci 20(6):388–397. https://doi.org/10.1016/j.tplants.2015.03.014
Fowke L, Dibbayawan T, Schwartz O, Harper J, Overall R (1999) Combined immunofluorescence and field emission scanning electron microscope study of plasma membrane-associated organelles in highly vacuolated suspensor cells of white spruce somatic embryos. Cell Biol Int 23(6):389–397. https://doi.org/10.1006/cbir.1999.0368
Ge YX, Angenent GC, Dahlhaus E, Franken J, Peters J, Wullens GJ, Creemers-Molenaar J (2001) Partial silencing of the NEC1 gene results in early opening of anthers in Petunia hybrida. Mol Genet Genomics 265(3):414–423. https://doi.org/10.1007/s004380100449
Hamada T (2014) Microtubule organization and microtubule-associated proteins in plant cells. Int Rev Cell Mol Biol 312:1–52. https://doi.org/10.1016/B978-0-12-800178-3.00001-4
Hussey PJ, Hawkins TJ, Igarashi H, Kaloriti D, Smertenko A (2002) The plant cytoskeleton: recent advances in the study of the plant microtubule-associated proteins MAP-65, MAP-190 and the Xenopus MAP215-like protein, MOR1. Plant Mol Biol 50(6):915–924
Ishiguro S, Kawai-Oda A, Ueda J, Nishida I, Okada K (2001) The DEFECTIVE IN ANTHER DEHISCENCE1 gene encodes a novel phospholipase A1 catalyzing the initial step of jasmonic acid biosynthesis, which synchronizes pollen maturation, anther dehiscence, and flower opening in Arabidopsis. Plant Cell 13(10):2191–2209
Kawamura E, Wasteneys GO (2008) MOR1, the Arabidopsis thaliana homologue of Xenopus MAP215, promotes rapid growth and shrinkage, and suppresses the pausing of microtubules in vivo. J Cell Sci 121(24):4114–4123. https://doi.org/10.1242/jcs.039065
Keijzer CJ (1987) The processes of anther dehiscence and pollen dispersal. I. The opening mechanism of longitudinally dehiscing anthers. New Phytol 105(3):487–498. https://doi.org/10.1111/j.1469-8137.1987.tb00886.x
Konopka CA, Backues SK, Bednarek SY (2008) Dynamics of Arabidopsis dynamin-related protein 1C and a clathrin light chain at the plasma membrane. Plant Cell 20(5):1363–1380. https://doi.org/10.1105/tpc.108.059428
Lechner B, Rashbrooke MC, Collings DA, Eng RC, Kawamura E, Whittington AT, Wasteneys GO (2012) The N-terminal TOG domain of Arabidopsis MOR1 modulates affinity for microtubule polymers. J Cell Sci 125(Pt 20):4812–4821. https://doi.org/10.1242/jcs.107045
Ledbetter MC, Porter KR (1964) Morphology of microtubules of plant cell. Science 144(3620):872–874. https://doi.org/10.1126/science.144.3620.872
Li Q, Zheng J, Li S, Huang G, Skilling SJ, Wang L, Li L, Li M, Yuan L, Liu P (2017) Transporter-mediated nuclear entry of jasmonoyl-isoleucine is essential for jasmonate signaling. Mol Plant 10(5):695–708. https://doi.org/10.1016/j.molp.2017.01.010
Li S, Cao L, Chen X, Liu Y, Persson S, Hu J, Chen M, Chen Z, Zhang D, Yuan Z (2021) A synthetic biosensor for mapping dynamic responses and spatio-temporal distribution of jasmonate in rice. Plant Biotechnol J. https://doi.org/10.1111/pbi.13718
Ma H (2005) Molecular genetic analyses of microsporogenesis and microgametogenesis in flowering plants. Annu Rev Plant Biol 56:393–434. https://doi.org/10.1146/annurev.arplant.55.031903.141717
Paredez AR, Somerville CR, Ehrhardt DW (2006) Visualization of cellulose synthase demonstrates functional association with microtubules. Science 312(5779):1491–1495. https://doi.org/10.1126/science.1126551
Sanders PM, Bui AQ, Weterings K, McIntire KN, Hsu Y-C, Lee PY, Truong MT, Beals TP, Goldberg RB (1999) Anther developmental defects in Arabidopsis thaliana male-sterile mutants. Sex Plant Reprod 11(6):297–322. https://doi.org/10.1007/s004970050158
Sanders PM, Bui AQ, Le BH, Goldberg RB (2005) Differentiation and degeneration of cells that play a major role in tobacco anther dehiscence. Sex Plant Reprod 17(5):219–241. https://doi.org/10.1007/s00497-004-0231-y
Scott RJ, Spielman M, Dickinson HG (2004) Stamen structure and function. Plant Cell 16(Suppl):S46-60. https://doi.org/10.1105/tpc.017012
Sedbrook JC, Kaloriti D (2008) Microtubules, MAPs and plant directional cell expansion. Trends Plant Sci 13(6):303–310. https://doi.org/10.1016/j.tplants.2008.04.002
Shen J, Wang X, Jiang L (2020) Exocytosis, endocytosis and membrane recycling in plant cells. eLS. Wiley, Hoboken, pp 490–499. https://doi.org/10.1002/9780470015902.a0029216
Smyth DR, Bowman JL, Meyerowitz EM (1990) Early flower development in Arabidopsis. Plant Cell 2(8):755–767. https://doi.org/10.1105/tpc.2.8.755
Tan X, Wei J, Li B, Wang M, Bao Y (2017) AtVps11 is essential for vacuole biogenesis in embryo and participates in pollen tube growth in Arabidopsis. Biochem Biophys Res Commun 491(3):794–799. https://doi.org/10.1016/j.bbrc.2017.07.059
Twell D, Park SK, Hawkins TJ, Schubert D, Schmidt R, Smertenko A, Hussey PJ (2002) MOR1/GEM1 has an essential role in the plant-specific cytokinetic phragmoplast. Nat Cell Biol 4(9):711–714. https://doi.org/10.1038/ncb844
Vallee RB, Herskovits JS, Burgess CC (1993) Dynamin, a microtubule-activated GTPase involved in endocytosis. Springer, Berlin, Heidelberg
Verma DPS, Hong Z, Menzel D (2006) Dynamin-related proteins in plant endocytosis. In: Šamaj J, Baluška F, Menzel D (eds) Plant endocytosis. Springer, Berlin, Heidelberg, pp 217–232. https://doi.org/10.1007/7089_013
Whittington AT, Vugrek O, Wei KJ, Hasenbein NG, Sugimoto K, Rashbrooke MC, Wasteneys GO (2001) MOR1 is essential for organizing cortical microtubules in plants. Nature 411(6837):610–613. https://doi.org/10.1038/35079128
Wilson ZA, Song J, Taylor B, Yang C (2011) The final split: the regulation of anther dehiscence. J Exp Bot 62(5):1633–1649. https://doi.org/10.1093/jxb/err014
Acknowledgements
We thank Dr. Kezhen Yang and Dr. Yiqun Bao for providing seeds of mCherry-TUA5 and CLC-RFP transgenic plants, respectively. We thank Dr. De Ye for revising the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Authors declare they have no conflict of interest.
Additional information
Communicated by Dali Zeng.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Liu, X., Tan, X. Mutation in Arabidopsis MOR1 gene impairs endocytosis in stamen filament cells and results in anther indehiscence. Plant Growth Regul 96, 303–314 (2022). https://doi.org/10.1007/s10725-021-00777-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10725-021-00777-7