Abstract
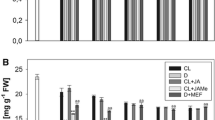
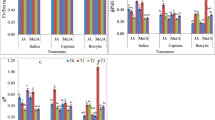
Methyl jasmonate (JA-Me) applied to the abaxial side of the middle of leaf blade on the beginning of September extremely induced leaf senescence in Ginkgo biloba; three weeks after the treatment the leaves disappeared chlorophylls and became yellow, while the control leaves remained still green. Contrarily JA-Me applied to the adaxial side of leaves affected little to induce senescence. When JA-Me was treated on the abaxial side of a half of leaf blade across to the vein, the yellowing widely took place both in acropetal and basipetal directions along vein, but no yellowing was visible in the non-treated half. On the other hand, when JA-Me was applied to the abaxial side of a half of leaf blade along to the vein, yellowing was observed only in both sides of a small area along to the applied area, and little yellowing was visible in the neighboring lateral tissues. JA-Me treatment on the abaxial side of leaf blade greatly increased the levels of JA-Me and jasmonic acid in leaf blade and petiole. Endogenous abscisic acid levels substantially increased during natural and JA-Me-induced leaf senescence, but 1-aminocyclopropane carboxylic acid levels did not change. The contents of cytokinins, gibberellins and auxins identified changed little during natural and JA-Me-induced leaf senescence. These suggest that a cooperative crosstalk between JAs and various hormonal signals, especially abscisic acid, occurs in regulation of G. biloba leaf senescence. Possible different action of JA-Me applied to the abaxial and the adaxial sides of the leaves on leaf senescence is discussed.








Similar content being viewed by others
References
Bruinsma J (1963) The quantitative analysis of chlorophylls a and b in plant extracts. Phytochem Phytobiol 2:241–249. https://doi.org/10.1111/j.1751-1097.1963.tb08220.x
Cheng SY, Xu F, Wang Y (2009) Advances in the study of flavonoids in Ginkgo biloba leaves. J Med Plants Res 3:1248–1252
Daszkowska-Golec A, Szarejko I (2013) Open or close the gate–stomata action under the control of phytohormones in drought stress conditions. Front Plant Sci 4:138. https://doi.org/10.3389/fpls.2013.00138
De Ollas C, Arbona V, Gómez-Cadenas A (2015) Jasmonic acid interacts with abscisic acid to regulate plant responses to water stress conditions. Plant Signal Behav 10:12. https://doi.org/10.1080/15592324.2015.1078953. e1078953.
Dmuchowski W, Brągoszewska P, Gozdowski D, Baczewska-Dabrowska AB, Chojnacki T, Jozwiak A, Swiezewska E, Gworek B, Suwara I (2019) Strategy of Ginkgo biloba L. in the mitigation of salt stress in the urban environment. Urban For Urban Green 38:223–231. https://doi.org/10.1016/j.ufug.2019.01.003
Du H, Liu H, Xiong L (2013) Endogenous auxin and jasmonic acid levels are differentially modulated by abiotic stresses in rice. Front Plant Sci 4:397. https://doi.org/10.3389/fpls/2013.00397
Dziurka M, Janeczko A, Juhász C, Gullner G, Oklestková J, Novák O, Saja D, Skoczowski A, Tóbiász J, Barna B (2016) Local and systemic hormonal responses in pepper leaves during compatible and incompatible pepper-tobamovirus interactions. Plant Physiol Biochem 109:355–364. https://doi.org/10.1016/j.plaphy.2016.10.013
Engvild KCV (1989) The death hormone hypothesis. Physiol Plant 77:282–285. https://doi.org/10.1111/j.1399-3054.1989.tb04982.x
Gachet MS, Schubert A, Calarco S, Boccardi J, Gertsch J (2017) Targeted metabolomics shows plasticity in the evolution of signaling lipids and uncovers old and new endocannabinoids in the plant kingdom. Sci Rep 7:41177. https://doi.org/10.1038/srep41177
Gülz P-G, Müller E, Schmitz K, Marner F-J, Güth S (1992) Chemical composition and surface structures of epicuticular leaf waxes of Ginkgo biloba, Magnolia grandiflora and Liliodendron tulipifera. Z Naturforsch 47:516–526. https://doi.org/10.1515/znc-1992-7-805
Hasler A (2005) Chemical constituents of Ginkgo biloba. In: Van Beek TA (ed) Ginkgo biloba. Harvard Academic Publishers, Switzerland, pp 124–164
He X, Fukushige H, Hildebrand D, Gan S (2002) Evidence supporting a role of jasmonic acid in Arabidopsis leaf senescence. Plant Physiol 128:876–884. https://doi.org/10.1104/pp.010843
Hentrich M, Büttcher C, Düchting P, Cheng Y, Zhao Y, Berkowitz O, Masle J, Medina J, Pollmann S (2013) The jasmonic acid signaling pathway is linked to auxin homeostasis through the modulation of YUCCA8 and YUCCA9 gene expression. Plant J 74:626–637. https://doi.org/10.1111/tpj.12152
Hossain MA, Munemasa S, Uraji M, Nakamura Y, Mori IC, Murata Y (2011) Involvement of endogenous abscisic acid in methyl jasmonate-induced stomatal closure in Arabidopsis. Plant Physiol 156:430–438. https://doi.org/10.1104/pp.111.172254
Ishimaru Y, Hayashi K, Suzuki T, Fukai H, Prusinska J, Meester C, Quareshy S, Egoshi S, Matsuura H, Takahashi K, Kato N, Kombrik E, Napier RM, Hayashi K, Ueda M (2018) Jasmonic acid inhibits auxin-induced lateral rooting independently of the CORONATINE INSENSITIVE1 receptor. Plant Physiol 177:1704–1716. https://doi.org/10.1104/pp.18.00357
Jiang Y, Ye J, Li S, Niinemets Ü (2017) Methyl jasmonate-induced emission of biogenic volatiles is biphasic in cucumber: a high-resolution analysis of dose dependence. J Exp Bot 68: 4679–4694. https://doi.org/10.1093/jxb/erx244
Jin J, Jiang H, Yu SQ, Zhou GM (2008) Sex-linked photosynthetic physiologic research and the evolutionary ecological analysis in living fossil plant, Ginkgo biloba L. Acta Ecol Sin 28:1128–1136. https://doi.org/10.1016/S1872-2032(08)60035-9
Kang S-M, Min J-Y, Kim Y-D, Park D-J, Jung H-N, Kim S-W, Choi M-S (2006) Effects of methyl jasmonate and salicylic acid on the production of bilobalide and ginkgolides in cell cultures of Ginkgo biloba. In Vitro Cell Dev Biol–Plant 42:44–49. https://doi.org/10.1079/IVP2005719
Kim EH, Kim SK, Park SH, Koo YJ, Choi YD, Chung Y-Y, Lee I-J, Kim J-K (2009) Methyl jasmonate reduces grain yield by mediating stress signals to alter spikelet development. Plant Physiol 149:1751–1760. https://doi.org/10.1104/pp.108.134684
Klimko M, Korszun S, Bykowska J (2015) Comparative morphology and anatomy of the leaves of Ginkgo biloba L. cultivars. Acta Sci Pol Hortorum Cult 14:169–189
Kondo S, Motoyama M, Michiyama H, Kim M (2002) Roles of jasmonic acid in the development of sweet cherries as measured from fruit or disc samples. Plant Growth Regul 37:37–44. https://link.springer.com/content/pdf/10.1023/A:1020362926829.pdf
Kramer EM, Ackelsberg EM (2015) Auxin metabolism rates and implications for plant development. Front Plant Sci 6:150. https://doi.org/10.3389/fpls.2015.00150
Lackman P, González-Guzmán M, Tilleman S, Carqueijeiro I, Pérez AC, Moses T, Seo M, Kannpo Y, Häkkinen ST, Van Montagu MCE, Thevelein JM, Maaheimo H, Oksman-Caldentey K-M, Rodriguez PL, Rischer H, Goossens A (2011) Jasmonate signaling involves the abscisic acid receptor PYL4 to regulate metabolic reprogramming in Arabidopsis and tobacco. Proc Natl Acad Sci USA 108:5891–5896. https://doi.org/10.1073/pnas.1103010108
Lépez-Carbonell M, Gabasa M, Jáuregui O (2009) Enhanced determination of abscisic acid (ABA) and its abscisic acid glucose ester (ABA-GE) in Cistus albidus plants by liquid chromatography-mass spectrometry in tandem mode. Plant Physiol Biochem 47:256–261. https://doi.org/10.1016/j.plaphy.2008.12.016
Li W, Yang S, Lu Z, He Z, Ye Y, Zhao B, Wang L, Jin B (2018) Cytological, physiological, and transcriptomics analyses of golden leaf coloration in Ginkgo biloba L. Hortic Res 5:12. https://doi.org/10.1038/s41438-018-0015-4
Ljung K (2013) Auxin metabolism and homeostasis during plant development. Development 140:943–950. https://doi.org/10.1242/dev.086363
Ludwig-Müller J (2011) Auxin conjugates: their role for plant development and in the evolution of land plants. J Exp Bot 62: 1757–1773. https://doi.org/10.1093/jxb/erq412
Matile P (1994) Fluorescent idioblasts in autumn leaves of Ginkgo biloba. Bot Helv 104:87–92
Matile P (2000) Biochemistry of Indian summer: physiology of autumnal leaf coloration. Exp Gerontol 35(2):145–158. https://doi.org/10.1016/S0531-5565(00)00081-4
Mueller-Roeber B, Balazadeh S (2014) Auxin and its role in plant senescence. J Plant Growth Regul 33:21–33. https://doi.org/10.1007/s00344-013-9398-5
Novák O, Hényková E, Sairanen I, Kowalczyk M, Pospíšil T, Ljung K (2012) Tissue-specific profiling of the Arabidopsis thaliana auxin metabolome. Plant J 72:523–536. https://doi.org/10.1111/j.1365-313X.2012.05085.x
Okazawa A, Tang L, Itoh Y, Fukusaki E, Kobayashi A (2006) Characterization and subcellular localization of chlorophyllase from Ginkgo biloba. Z Naturforsch C 61:111–117. https://doi.org/10.1515/znc-2006-1-220
Park J, Thimann KV (1990) Senescence and stomatal aperture as affected by antibiotics in darkness and light. Plant Physiol 92:696–702. https://doi.org/10.1104/pp.92.3.696
Pazmino DM, Rodriguez-Serrano M, Romero-Puertas MC, Sandalio LM (2014) Regulation of epinasty induced by 2,4-dichlorophenoxyacetic acid in pea and Arabidopsis plants. Plant Biol 16:809–818. https://doi.org/10.1111/plb.12128
Pěnčík A, Simonovik B, Petersson SV, Henyková E, Simon S, Greenham K, Zhang Y, Kowalczyk M, Estelle M, Zažímalová E, Novák O, Sandberg G, Ljung K (2013) Regulation of auxin homeostasis and gradients in Arabidopsis roots through the formation of the indole-3-acetic catabolite 2-oxindole-3-acetic acid. Plant Cell 25:3858–3870. https://doi.org/10.1105/tpc.113.114421
Pérez AC, Goossens A (2013) Jasmonate signaling: a copycat of auxin signaling? Plant Cell Environ 36:2071–2084. https://doi.org/10.1111/pce.12121
Płażek A, Dubert F, Kopeć P, Dziurka M, Kalandyk A, Pastuszak J, Wolko B (2018) Seed hydropriming and smoke water significantly improve low-temperature germination of Lupinus angustifolius L. Int J Mol Sci 19:992. https://doi.org/10.3390/ijms19040992
Reinecke DM, Bandurski RS (1981) Metabolic conversion of 14C-indole-3-acetic acid to 14C-oxindole-3-acetic acid. Biochem Biophys Res Commun 103:429–433. https://doi.org/10.1016/0006-291X(81)90470-8
Reinecke DM, Bandurski RS (1983) Oxindole-3-acetic acid, an indole-3-acetic acid catabolite in Zea mays. Plant Physiol 71:211–213. https://doi.org/10.1104/pp.71.1.211
Sabater-Jara AB, Souliman-Youssef S, Novo-Uzal E, Almagro L, Belchi-Navarro S, Pedreño MA (2013) Biotechnological approaches to enhance the biosynthesis of ginkgolides and bilobalide in Ginkgo biloba. Phytochem Rev 12:191–205. https://doi.org/10.1007/s11101-013-9275-7
Saniewski M, Ueda J, Miyamoto K (2002) Relationships between jasmonates and auxin in regulation of some physiological processes in higher plants. Acta Physiol Plant 24: 211–220. https://doi.org/10.1007/s11738-002-0013-9
Sato C, Aikawa K, Sugiyama S, Nabeta K, Masuta C, Matsuura H (2011) Distal transport of exogenously applied jasmonoyl-isoleucine with wounding stress. Plant Cell Physiol 52:509–517. https://doi.org/10.1093/pcp/pcr011
Schennen A, Hölzl J (1986) 6-Hydroxykyunurensäure, die erste N-haltige Verbindung aus den Blättern von Ginkgo biloba. Plant Med 52:235–236. https://doi.org/10.1055/s-2007-969133
Seo HS, Song JT, Cherong J-J, Lee Y-H, Lee Y-W, Hwang I, Lee JS, Choi YD (2001) Jasmonic acid carboxyl methyltransferase: A key enzyme for jasmonate-regulated plant responses. Proc Natl Acad Sci USA 98:4788–4793. https://doi.org/10.1073/pnas.081557298
Shi D, Wei X, Chen G, Xu Y (2012) Changes in photosynthetic characteristics and antioxidative protection in male and female Ginkgo during natural senescence. J Am Soc Hort Sci 137:349–360. https://doi.org/10.21273/JASHS.137.5.349
Singh B, Kaur P, Singh GRD, Ahuja PS (2008) Biology and chemistry of Ginkgo biloba. Fitoterapia 79:401–418. https://doi.org/10.1016/j.fitote.2008.05.007
Strømgaard K, Nakanishi K (2004) Chemistry and biology of terpene trilactones from Ginkgo biloba. Angew Chem Int Ed Eng 43:1640–1658. https://doi.org/10.1002/anie.200300601
Sukito A, Tachibana S (2016) Effect of methyl jasmonate and salicylic acid synergism on enhancement of bilobalide and ginkgolide production by immobilized cell cultures of Ginkgo biloba. Bioresour Bioprocess 3:24. https://doi.org/10.1186/s40643-016-0101-0
Szewczyk A (2008) Wpływ elicytacji jasmonianem metylu na akumulację kwasów fenolowych w kulturach zawiesinowych Ginkgo biloba L. Zesz Probl Postępów Nauk Roln 524:419–423
Tang L, Okazawa A, Itoh Y, Fukusaki E, Kobayashi A (2004) Expression of chlorophyllase is not induced during autumnal yellowing in Ginkgo biloba. Z Naturforsch C 59:415–420. https://doi.org/10.1515/znc-2004-5-623
Thimann KV (1977) Hormone action in the whole life of plants. The University of Massathusetts Press, Amherst
Thimann KV, Satler SO (1978) Relation between leaf senescence and stomatal closure: Senescence in light. Proc Natl Acad Sci USA 76(5):2295–2298. https://doi.org/10.1073/pnas.76.5.2295
Thorpe MR, Ferrieri AP, Herth MM, Ferrieri RA (2007) 11C-imaging: 11C-imaging: methyl jasmonate moves in both phloem and xylem, promotes transport of jasmonate, and of photoassimilate even after proton transport is decoupled. Planta 226:541–551. https://doi.org/10.1007/s00425-007-0503-5
Tomogami S, Noge K, Abe M, Agrawal GK, Rakwal R (2012) Methyl jasmonate is transported to distal leaves via vascular process metabolizing itself into JA-Ile and triggering VOCs emission as defensive metabolites. Plant Signal Behav 7:1378–1381. https://doi.org/10.4161/psb.21762
Ueda J, Kato J (1980) Isolation and identification of a senescence-promoting substance from wormwood (Artemisia absinthium L.). Plant Physiol 66:246–249. https://doi.org/10.1104/pp.66.2.246
Ueda J, Kato J (1982) Identification of jasmonic acid and abscisic acid as senescence-promoting substances from Cleyera ochnacea DC. Agric Biol Chem 46:1975–1976. https://doi.org/10.1271/bbb1961.46.1975
Van Beek TA (2002) Chemical analysis of Ginkgo biloba leaves and extracts. J Chromatogr A 967:21–55. https://doi.org/10.1016/S0021-9673(02)00172-3
Van Beek TA, Montoro P (2009) Chemical analysis and quality control of Ginkgo biloba leaves, extracts, and phytopharmaceuticals. J Chromatogr A 1216:2002–2032. https://doi.org/10.1016/j.chroma.2009.01.013
Van Staden J (1996) Changes in foliar cytokinins of Salix babylonica and Ginkgo biloba prior to and during leaf senescence. S Afr J Bot 62:1–10. https://doi.org/10.1016/S0254-6299(15)30570-6
Wasternack C (2007) Jasmonates: An update on biosynthesis, signal transduction and action in plant stress response, growth and development. Ann Bot 100:681–697
Wasternack C, Hause B (2013) Jasmonates: biosynthesis, perception, signal transduction and action in plant stress response, growth and development. An update to the 2007 review in Annals of Botany. Ann Bot 111:1021–1058. https://doi.org/10.1093/aob/mct067
Wei X-D, Shi D-W, Chen G-X (2013) Physiological, structural, and proteomic analysis of chloroplasts during natural senescence of Ginkgo leaves. Plant Growth Regul 69:191–201. https://doi.org/10.1007/s10725-012-9761-8
Wiszniewska A, Koźmińska A, Hanus-Fajerska E, Dziurka M, Dziurka K (2019) Insight into mechanisms of multiple stresses tolerance in a halophyte Aster tripolium subjected to salinity and heavy metal stress. Ecotoxicol Environ Saf 180:12–22. https://doi.org/10.1016/j.ecoenv.2019.04.059
Xu M, Dong J, Wang H, Huang L (2009) Complementary action of jasmonic acid on salicylic acid in mediating fungal elicitor-induced flavonol glycoside accumulation of Ginkgo biloba cells. Plant Cell Environ 32:960–967. https://doi.org/10.1111/j.1365-3040.2009.01976.x
Yang X-S, Chen G-X, Yuan Z-Y (2013) Photosynthetic decline in Ginkgo leaves during natural senescence. Pak J Bot 45:1537–1540
Acknowledgements
This work was supported by Polish Ministry of Science and Higher Education through statutory funds of the Research Institute of Horticulture, Skierniewice, Poland.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Saniewski, M., Dziurka, M., Dziurka, K. et al. Methyl jasmonate induces leaf senescence of Ginkgo biloba L.: relevance to endogenous levels of plant hormones. Plant Growth Regul 91, 383–396 (2020). https://doi.org/10.1007/s10725-020-00612-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10725-020-00612-5